Advanced Chiral Pincer Palladium and Nickel Complexes for Asymmetric Catalysis Manufacturing
The landscape of asymmetric catalysis is undergoing a significant transformation driven by the need for more efficient and selective synthetic routes in the pharmaceutical industry. Patent CN112142790B introduces a groundbreaking class of chiral pincer-like compounds, specifically focusing on palladium and nickel complexes that exhibit exceptional stereocontrol. These novel organometallic structures, characterized by a rigid PCP (phosphine-carbon-phosphine) backbone, address long-standing challenges in creating stable, high-performance catalysts for asymmetric Michael additions and allylic substitutions. By integrating a defined carbon chiral center directly into the pincer skeleton, this technology offers a robust alternative to traditional resolution methods, providing a reliable pharmaceutical intermediate supplier with the tools necessary to access high-value chiral building blocks with unprecedented purity and efficiency.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the production of chiral secondary phosphine compounds, which are vital precursors for many homogeneous catalysts, has been plagued by inefficiency and high costs. The conventional wisdom relied heavily on the resolution of racemic mixtures using high-performance liquid chromatography (HPLC) or classical crystallization techniques. These methods are inherently wasteful, as they theoretically discard half of the synthesized material to achieve optical purity, leading to substantial increases in raw material consumption and solvent waste. Furthermore, the separation processes are often slow, difficult to scale, and sensitive to minor variations in process parameters, resulting in inconsistent batch quality. For procurement managers, this translates to volatile pricing and extended lead times, while R&D teams struggle with the limited availability of diverse chiral ligands needed for method development.
The Novel Approach
The methodology outlined in the patent represents a paradigm shift by employing a direct, chiral-controlled synthesis strategy that bypasses the need for resolution entirely. As illustrated in the comprehensive reaction scheme below, the process begins with a readily available chalcone derivative and utilizes a specialized chiral palladium catalyst to induce asymmetry during the initial phosphine addition. 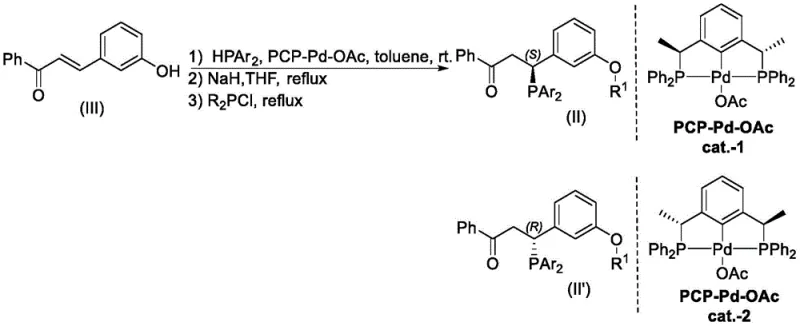 This streamlined approach not only maximizes atom economy by utilizing the entire starting material but also simplifies the downstream purification workflow. The ability to generate both (S) and (R) enantiomers selectively by simply switching between catalyst cat.-1 and cat.-2 provides unparalleled flexibility for process chemists. This direct route significantly reduces the number of unit operations required, thereby lowering the overall cost reduction in chiral catalyst manufacturing and ensuring a more consistent supply of high-purity reagents for complex synthetic sequences.
This streamlined approach not only maximizes atom economy by utilizing the entire starting material but also simplifies the downstream purification workflow. The ability to generate both (S) and (R) enantiomers selectively by simply switching between catalyst cat.-1 and cat.-2 provides unparalleled flexibility for process chemists. This direct route significantly reduces the number of unit operations required, thereby lowering the overall cost reduction in chiral catalyst manufacturing and ensuring a more consistent supply of high-purity reagents for complex synthetic sequences.
Mechanistic Insights into PCP-Type Pincer Coordination Chemistry
The exceptional performance of these complexes stems from their unique structural architecture, which combines the stability of a tridentate pincer ligand with the precise stereochemical information encoded in the carbon backbone. The synthesis initiates with a conjugate addition where the chiral catalyst directs the attack of the phosphine nucleophile to the beta-position of the enone system. The subsequent basification and reaction with chlorophosphines lock this stereochemistry in place, creating a rigid framework that prevents racemization under typical reaction conditions. The resulting ligand coordinates to the metal center (Pd or Ni) through two phosphorus atoms and the central aryl carbon, forming a thermodynamically stable five-membered metallacycle. This rigid coordination sphere minimizes the degrees of freedom for the substrate, forcing it into a specific orientation that favors the formation of one enantiomer over the other during catalytic cycles.
Furthermore, the versatility of this system allows for fine-tuning of the electronic and steric properties through the selection of different substituents on the phosphorus atoms, such as tert-butyl, cyclohexyl, or phenyl groups. 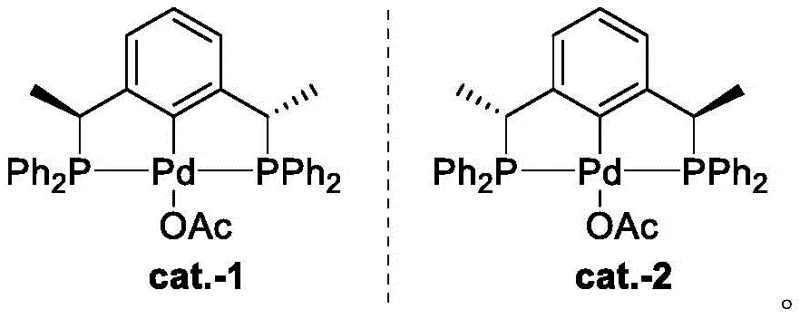 The patent also highlights an optional anion exchange step, where chloride ligands can be replaced with acetate groups. This modification is crucial for modulating the Lewis acidity of the metal center and improving solubility in various reaction media. Such tunability is essential for optimizing catalytic activity in diverse transformations, ranging from asymmetric Heck couplings to hydrogenation reactions. By understanding these mechanistic nuances, manufacturers can tailor the catalyst properties to specific substrate classes, ensuring high turnover frequencies and minimizing the formation of trace impurities that could complicate regulatory filings for active pharmaceutical ingredients.
The patent also highlights an optional anion exchange step, where chloride ligands can be replaced with acetate groups. This modification is crucial for modulating the Lewis acidity of the metal center and improving solubility in various reaction media. Such tunability is essential for optimizing catalytic activity in diverse transformations, ranging from asymmetric Heck couplings to hydrogenation reactions. By understanding these mechanistic nuances, manufacturers can tailor the catalyst properties to specific substrate classes, ensuring high turnover frequencies and minimizing the formation of trace impurities that could complicate regulatory filings for active pharmaceutical ingredients.
How to Synthesize Chiral Pincer Complexes Efficiently
The synthesis protocol described in the patent is designed for operational simplicity while maintaining rigorous control over stereochemical outcomes. The process typically involves a one-pot or telescoped sequence where the chiral addition, basification, and phosphorylation steps are conducted sequentially without isolating unstable intermediates. This approach minimizes exposure to air and moisture, which is critical for handling reactive phosphine species. Following the formation of the free ligand, coordination with divalent metal salts is performed under mild heating in common organic solvents like tetrahydrofuran or toluene. The detailed standardized synthesis steps see the guide below for specific molar ratios and temperature profiles optimized for maximum yield and enantiomeric excess.
- Perform a chiral-controlled addition reaction between a chalcone derivative and a diarylphosphine using a specialized chiral palladium catalyst (cat.-1 or cat.-2) in an inert organic solvent at low temperatures.
- Execute a basification step using strong alkali compounds such as sodium hydride or n-butyllithium to facilitate the formation of the carbon-phosphorus bond and establish the chiral center.
- Complete the synthesis by reacting the intermediate with chlorophosphines followed by coordination with divalent palladium or nickel salts, optionally performing anion exchange to tune catalytic properties.
Commercial Advantages for Procurement and Supply Chain Teams
For supply chain leaders and procurement specialists, the adoption of this technology offers tangible benefits that extend beyond mere technical performance. The elimination of resolution steps fundamentally alters the cost structure of producing chiral catalysts, removing the expensive and time-consuming chromatographic separations that traditionally bottleneck production. This efficiency gain allows for a significant reduction in solvent usage and waste disposal costs, aligning with modern green chemistry initiatives and environmental compliance standards. Moreover, the robustness of the synthesis conditions means that production can be scaled up rapidly without the need for specialized cryogenic equipment or exotic reagents, ensuring a stable and continuous supply of critical catalytic materials.
- Cost Reduction in Manufacturing: The direct asymmetric synthesis route drastically simplifies the production workflow by consolidating multiple steps into a streamlined sequence. By avoiding the 50% theoretical loss associated with racemic resolution, the process effectively doubles the yield from the same amount of starting material. This inherent efficiency, combined with the use of commodity chemicals like chalcones and simple phosphines, leads to substantial cost savings in the final product price, making high-performance chiral catalysis economically viable for large-scale industrial applications.
- Enhanced Supply Chain Reliability: The reliance on readily available starting materials and standard solvents mitigates the risk of supply disruptions often associated with specialized chiral pool reagents. The synthetic route is tolerant to minor variations in reaction conditions, which enhances batch-to-batch consistency and reduces the rate of failed productions. This reliability is crucial for maintaining uninterrupted manufacturing schedules for downstream pharmaceutical clients who depend on just-in-time delivery of high-purity intermediates for their own synthesis campaigns.
- Scalability and Environmental Compliance: The process utilizes common organic solvents such as THF, toluene, and dichloromethane, which are well-understood in terms of recovery and recycling infrastructure. The workup procedures involve standard operations like filtration, washing, and recrystallization, which are easily adaptable from laboratory glassware to multi-ton reactor systems. This scalability ensures that the technology can meet growing market demand without compromising on purity or environmental safety, facilitating the commercial scale-up of complex polymer additives and fine chemical intermediates with a reduced carbon footprint.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this chiral pincer technology. These insights are derived directly from the experimental data and structural analysis provided in the patent documentation, offering clarity on how these catalysts can be integrated into existing manufacturing workflows. Understanding these details is essential for technical teams evaluating the feasibility of adopting this new class of organometallic reagents for their specific synthetic challenges.
Q: What distinguishes these chiral pincer complexes from traditional phosphine ligands?
A: Unlike traditional monodentate phosphines, these PCP-type pincer complexes feature a rigid tridentate architecture that locks the metal center in a specific geometry. This structural rigidity, combined with the introduced carbon chirality, provides superior thermal stability and significantly higher enantioselectivity in asymmetric transformations like Michael additions.
Q: Can these catalysts be scaled for industrial production of API intermediates?
A: Yes, the synthesis pathway described in patent CN112142790B utilizes robust reaction conditions and commercially available solvents like THF and toluene. The purification involves standard operations such as filtration and recrystallization, making the process highly amenable to commercial scale-up from kilogram to multi-ton quantities without requiring exotic equipment.
Q: How does the anion exchange modification affect catalyst performance?
A: The patent details an optional anion exchange step where chloride ligands are replaced with acetate groups. This modification alters the electronic environment around the metal center, often enhancing solubility in polar media and modifying the Lewis acidity, which can be critical for optimizing turnover numbers in specific asymmetric coupling reactions.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Chiral Pincer Complexes Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of the chiral pincer complexes described in patent CN112142790B for advancing asymmetric synthesis in the pharmaceutical sector. As a dedicated CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from benchtop discovery to industrial manufacturing is seamless and efficient. Our state-of-the-art facilities are equipped with rigorous QC labs capable of verifying stringent purity specifications, including precise enantiomeric excess measurements, to guarantee that every batch meets the highest international standards for quality and performance.
We invite you to collaborate with our technical procurement team to explore how these advanced catalysts can optimize your specific synthetic routes. By requesting a Customized Cost-Saving Analysis, you can gain a deeper understanding of the economic benefits tailored to your production volume. We encourage you to contact us today to obtain specific COA data and route feasibility assessments, allowing us to demonstrate our commitment to delivering high-quality chiral solutions that drive innovation and efficiency in your supply chain.
