Scalable Green Synthesis of 2-Amino-1,3,4-Oxadiazoles: A Technical Analysis for Global Supply Chains
The pharmaceutical and agrochemical industries continuously seek efficient pathways to access bioactive heterocyclic scaffolds, among which the 2-amino-1,3,4-oxadiazole motif stands out due to its profound biological significance. As detailed in the recent Chinese patent CN113307780B, a novel preparation method has been developed that addresses long-standing challenges in synthesizing these critical intermediates. This technology leverages potassium bisulfate (KHSO4) as a mild, cost-effective catalyst to drive the cyclization of benzoyl hydrazides and isothiocyanates, achieving yields up to 85% under remarkably benign conditions. Unlike traditional methods that rely on hazardous reagents or extreme thermal inputs, this approach prioritizes operational safety and environmental compliance without compromising chemical efficiency. For global supply chain leaders, this represents a pivotal opportunity to optimize the manufacturing of high-purity pharmaceutical intermediates while mitigating regulatory risks associated with corrosive waste streams.
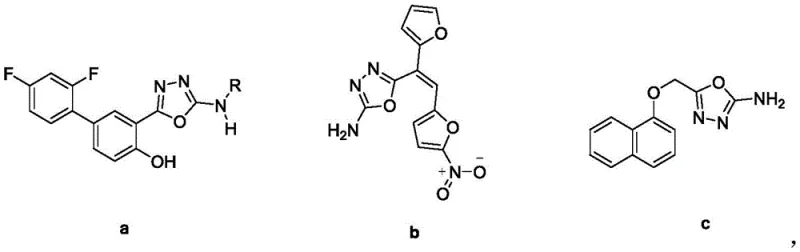
The structural versatility of the 2-amino-1,3,4-oxadiazole core is evident in its presence across a spectrum of therapeutic agents, ranging from muscle relaxants to potent antifungal compounds. The ability to construct this scaffold reliably is therefore not merely an academic exercise but a commercial imperative for reliable pharmaceutical intermediate supplier networks. By shifting away from legacy synthetic routes, manufacturers can unlock substantial cost savings in API manufacturing through reduced energy consumption and simplified downstream processing. The following analysis dissects the technical merits of this innovation, providing R&D directors and procurement managers with the insights needed to evaluate its integration into existing production portfolios.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of the 1,3,4-oxadiazole ring has been plagued by significant process safety and economic hurdles that hinder scalable production. Conventional protocols often necessitate the use of elemental sulfur powder to promote cyclization, a reagent that poses severe flammability and explosion risks, particularly when handled in bulk quantities at elevated temperatures. Alternative pathways utilizing ammonium thiocyanate frequently require concentrated hydrochloric acid as a catalyst, introducing highly corrosive conditions that demand specialized reactor linings and generate difficult-to-treat acidic wastewater. Furthermore, modern peptide coupling strategies employing reagents such as EDCI or TBTU, while effective on a milligram scale, are economically prohibitive for tonnage production due to the exorbitant cost of these activators and the generation of stoichiometric urea byproducts that complicate purification. Other oxidative methods relying on hypervalent iodine species like IBX or molecular iodine introduce additional layers of complexity, requiring strict microwave conditions or dealing with sublimation issues that lead to reactor fouling and inconsistent batch quality.
The Novel Approach
In stark contrast to these cumbersome legacy techniques, the methodology disclosed in CN113307780B introduces a streamlined catalytic cycle driven by potassium bisulfate. This solid acid salt provides sufficient proton activity to facilitate the dehydration and cyclization steps without the aggressive corrosivity of liquid mineral acids. The reaction proceeds efficiently in dimethyl sulfoxide (DMSO), a polar aprotic solvent that stabilizes the transition states involved in the nucleophilic attack of the hydrazide on the isothiocyanate. Crucially, the process operates effectively at temperatures ranging from 25°C to 50°C, eliminating the need for energy-intensive heating protocols often required to overcome activation barriers in non-catalyzed variants. The post-reaction workup is exceptionally straightforward, typically involving the addition of water to precipitate the product or facilitate extraction, thereby bypassing the need for complex chromatographic separations that are impractical in large-scale commercial settings. This combination of mild conditions, inexpensive catalysis, and simple isolation defines a new standard for cost reduction in pharmaceutical intermediate manufacturing.
Mechanistic Insights into KHSO4-Catalyzed Cyclization
The efficacy of potassium bisulfate in this transformation stems from its unique ability to act as a solid-supported proton source that activates the electrophilic center of the isothiocyanate moiety. Upon mixing the benzoyl hydrazide and the isothiocyanate in DMSO, the KHSO4 dissociates to release protons which protonate the sulfur atom of the isothiocyanate group, increasing its electrophilicity and rendering it more susceptible to nucleophilic attack by the terminal nitrogen of the hydrazide. This initial addition forms a thiosemicarbazide intermediate, which subsequently undergoes an intramolecular cyclization. The acidic environment provided by the bisulfate ion facilitates the elimination of hydrogen sulfide or related sulfur species, driving the equilibrium towards the formation of the stable 1,3,4-oxadiazole ring. Unlike Lewis acids such as ferric chloride which failed to promote the reaction in experimental trials, the Brønsted acidity of KHSO4 is perfectly tuned to activate the substrates without causing decomposition of the sensitive heterocyclic product or the starting materials.
From an impurity control perspective, the mildness of the catalyst plays a pivotal role in ensuring high product purity. Harsh acidic conditions often lead to side reactions such as hydrolysis of the amide bond or polymerization of the reactive isothiocyanate, generating complex impurity profiles that are difficult to purge. By maintaining the reaction temperature below 50°C and utilizing a non-oxidative catalyst, the formation of colored byproducts and tars is significantly minimized. The patent data indicates that raising the temperature above 90°C leads to product darkening and decreased yields, confirming that the kinetic window for optimal selectivity lies within the ambient to mild heating range. This mechanistic understanding allows process chemists to define tight control parameters for commercial scale-up, ensuring that the critical quality attributes of the high-purity pharmaceutical intermediates are consistently met across different batch sizes.
How to Synthesize 2-Amino-1,3,4-Oxadiazole Efficiently
The practical implementation of this synthesis route is designed for ease of execution in standard chemical manufacturing facilities. The protocol begins with the dissolution of the benzoyl hydrazide and isothiocyanate substrates in DMSO, followed by the controlled addition of the potassium bisulfate catalyst. The molar ratio of the catalyst to the hydrazide substrate is a critical parameter, with optimization studies suggesting a ratio between 1:5 and 1:7 yields the best balance between reaction rate and economic efficiency. Maintaining the reaction mixture at 25-30°C for a duration of 5 to 6 hours typically ensures complete conversion of the starting materials. Following the reaction period, the mixture is quenched by pouring into water, which induces precipitation of the crude product. This solid can then be collected via filtration and dried, or further purified by recrystallization if higher purity specifications are required for specific drug applications. The detailed standardized synthesis steps see the guide below.
- Mix benzoyl hydrazide and isothiocyanate compounds in dimethyl sulfoxide (DMSO) solvent at room temperature.
- Add potassium bisulfate (KHSO4) catalyst at a molar ratio of 1: 6 relative to the hydrazide substrate.
- Stir the reaction mixture at 25-50°C for 4-8 hours, then quench with water and filter to isolate the product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this KHSO4-catalyzed methodology offers tangible strategic benefits that extend beyond simple yield improvements. The primary advantage lies in the drastic simplification of the supply chain for raw materials; potassium bisulfate is a commodity chemical available in bulk quantities at a fraction of the cost of specialized coupling reagents like EDCI or oxidants like IBX. This substitution directly translates to significant cost reduction in API manufacturing by lowering the bill of materials and reducing the dependency on single-source specialty chemical vendors. Furthermore, the elimination of hazardous reagents such as elemental sulfur or concentrated hydrochloric acid reduces the regulatory burden associated with storage, handling, and transportation, thereby enhancing overall supply chain reliability and reducing insurance and compliance overheads.
- Cost Reduction in Manufacturing: The economic impact of switching to this catalytic system is profound, primarily driven by the replacement of expensive stoichiometric reagents with a cheap, recyclable inorganic salt. Traditional methods often require excess amounts of costly activators to drive reactions to completion, whereas this method utilizes a catalytic amount of KHSO4 which is inherently low-cost. Additionally, the simplified workup procedure, which relies on water precipitation rather than extensive solvent exchanges or chromatography, significantly reduces solvent consumption and waste disposal costs. The avoidance of heavy metal catalysts also removes the need for expensive scavenging steps to meet residual metal limits in final drug substances, further streamlining the production budget and improving the overall margin profile for commercial scale-up of complex pharmaceutical intermediates.
- Enhanced Supply Chain Reliability: Supply continuity is a critical metric for any reliable pharmaceutical intermediate supplier, and this method bolsters reliability by utilizing widely available, non-proprietary starting materials. Benzoyl hydrazides and isothiocyanates are common building blocks with established global supply chains, minimizing the risk of shortages that can plague niche reagents. The robustness of the reaction conditions, which tolerate a wide range of temperatures and do not require inert atmospheres or anhydrous conditions as strictly as other methods, means that production is less susceptible to disruptions caused by equipment failures or utility fluctuations. This resilience ensures reducing lead time for high-purity pharmaceutical intermediates, allowing manufacturers to respond more agilely to market demands and inventory requirements from downstream clients.
- Scalability and Environmental Compliance: Scaling chemical processes from the laboratory to the plant floor often reveals hidden bottlenecks related to heat transfer and safety, but this protocol is inherently designed for scalability. The exotherm of the reaction is manageable at ambient temperatures, removing the need for complex cooling systems required for highly exothermic acid additions. From an environmental standpoint, the use of DMSO, while requiring recovery, is preferable to chlorinated solvents often used in alternative routes, and the aqueous waste stream generated during workup is easier to treat than those containing heavy metals or strong mineral acids. This alignment with green chemistry principles facilitates smoother regulatory approvals and supports corporate sustainability goals, making it an attractive option for companies aiming to reduce their environmental footprint while maintaining high production volumes.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These answers are derived directly from the experimental data and claims presented in the patent documentation, providing a factual basis for decision-making. Understanding these nuances is essential for technical teams evaluating the feasibility of adopting this route for their specific product pipelines. The responses cover catalyst loading, substrate scope, and purification strategies to ensure a comprehensive understanding of the process capabilities.
Q: What are the advantages of using KHSO4 over traditional catalysts for oxadiazole synthesis?
A: Potassium bisulfate (KHSO4) offers a mild acidic environment that avoids the corrosive hazards of concentrated sulfuric acid or hydrochloric acid. It eliminates the need for expensive coupling reagents like EDCI or oxidants like IBX, significantly reducing raw material costs and simplifying waste treatment.
Q: Is this synthesis method suitable for large-scale industrial production?
A: Yes, the method operates at mild temperatures (25-50°C) and uses inexpensive, stable reagents. The workup involves simple water addition and filtration, which is highly scalable compared to methods requiring complex chromatographic purification or hazardous gas evolution.
Q: What is the typical yield range for this catalytic system?
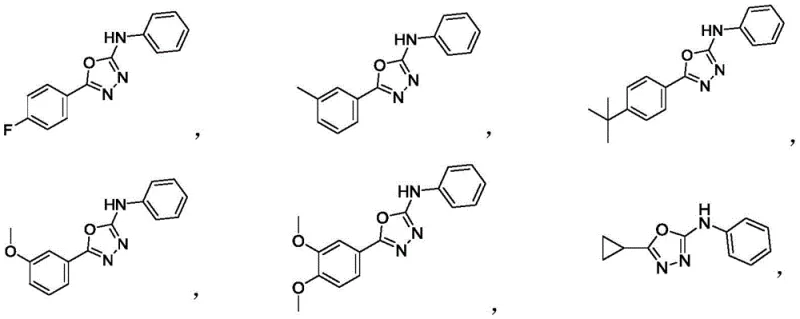
A: According to the patent data, yields can reach up to 85% under optimized conditions. Various substituted derivatives consistently show yields between 60% and 86%, demonstrating robust substrate tolerance.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-Amino-1,3,4-Oxadiazole Supplier
As the global demand for bioactive heterocycles continues to surge, partnering with a manufacturer that possesses deep technical expertise in advanced catalytic methodologies is essential for maintaining a competitive edge. NINGBO INNO PHARMCHEM stands at the forefront of this industry, leveraging cutting-edge process chemistry to deliver high-value intermediates with unmatched consistency. Our team has extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from pilot scale to full manufacturing is seamless and risk-mitigated. We adhere to stringent purity specifications and operate rigorous QC labs equipped with state-of-the-art analytical instrumentation to guarantee that every batch meets the exacting standards required by top-tier pharmaceutical companies worldwide.
We invite you to collaborate with us to explore how this innovative KHSO4-catalyzed synthesis can be tailored to your specific project needs. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis that quantifies the potential economic benefits of switching to this greener, more efficient route for your supply chain. Please contact us today to request specific COA data for our existing library of oxadiazole derivatives or to discuss route feasibility assessments for your proprietary targets. Together, we can drive efficiency and innovation in the development of next-generation therapeutic agents.
