Advanced Dopiperidine Amine Light Stabilizer: Technical Breakthroughs and Commercial Scalability
Advanced Dopiperidine Amine Light Stabilizer: Technical Breakthroughs and Commercial Scalability
The global demand for high-performance polymer additives continues to surge as manufacturers seek to extend the service life of plastic products in harsh environmental conditions. A significant technological advancement in this sector is detailed in patent CN116178346A, which discloses a novel dopiperidine amine light stabilizer, designated as Compound TP. This molecule represents a strategic evolution in stabilizer chemistry, moving beyond simple hindered amine light stabilizers (HALS) to a multifunctional architecture that combines triazine and resorcinol moieties with multiple piperidine units. For R&D directors and procurement specialists, understanding the synthesis and application of this compound is critical for developing next-generation polyolefins, polyesters, and engineering plastics that resist photo-oxidative degradation. The following analysis dissects the chemical innovation and supply chain implications of this technology.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional light stabilization strategies often rely on the physical blending of separate UV absorbers and hindered amine light stabilizers, a practice that introduces significant formulation complexities and performance inconsistencies. In many existing commercial products, the core active group, typically the piperidine amine moiety, is present in relatively low molar concentrations per unit of molecular weight, necessitating higher loading levels to achieve desired protection. Furthermore, the incompatibility between different additive types can lead to migration issues, where the stabilizer blooms to the surface of the plastic, causing haze and loss of efficacy over time. These limitations result in increased raw material costs and unpredictable mechanical failure in end-use applications such as automotive components and agricultural films, driving the urgent need for chemically integrated solutions.
The Novel Approach
The synthesis route described in the patent offers a transformative solution by covalently bonding multiple functional units into a single, high-molecular-weight entity. As illustrated in the reaction scheme below, the process begins with the nucleophilic substitution of cyanuric chloride, followed by the introduction of a resorcinol bridge and subsequent functionalization with additional piperidine derivatives. 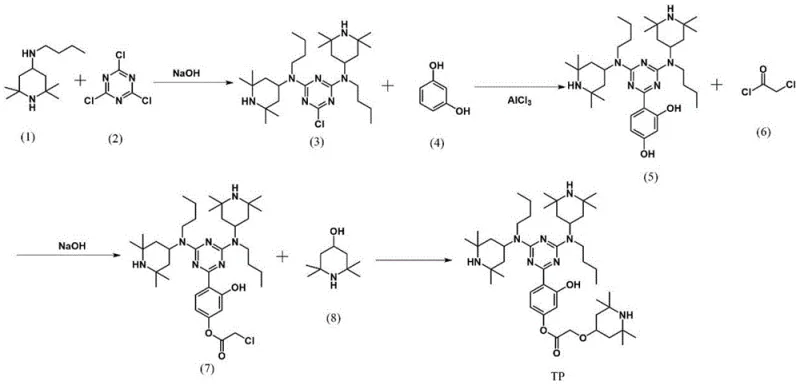 This integrated design ensures that every molecule delivers a triple dose of radical-scavenging capability alongside intrinsic UV absorption, eliminating the need for complex additive packages. The resulting compound exhibits superior compatibility with polymer matrices due to its tailored structure, significantly reducing migration risks and ensuring long-term stability. This approach not only simplifies the compounding process for manufacturers but also enhances the overall cost-efficiency of plastic production by maximizing the performance per gram of additive used.
This integrated design ensures that every molecule delivers a triple dose of radical-scavenging capability alongside intrinsic UV absorption, eliminating the need for complex additive packages. The resulting compound exhibits superior compatibility with polymer matrices due to its tailored structure, significantly reducing migration risks and ensuring long-term stability. This approach not only simplifies the compounding process for manufacturers but also enhances the overall cost-efficiency of plastic production by maximizing the performance per gram of additive used.
Mechanistic Insights into Triazine-Piperidine Synergistic Stabilization
The exceptional performance of Compound TP arises from its unique molecular architecture, which facilitates a synergistic mechanism of action rarely achieved in single-component stabilizers. The central 1,3,5-triazine ring acts as a rigid scaffold that supports three distinct piperidine amine groups, creating a high local concentration of nitrogen-centered radicals capable of intercepting propagating polymer radicals. Simultaneously, the incorporation of the 2,4-dihydroxyphenyl group via the resorcinol linkage introduces a potent UV-absorbing chromophore that dissipates incident photon energy as harmless heat through keto-enol tautomerism. This dual-functionality means the molecule actively protects the polymer matrix through both prevention (UV screening) and repair (radical trapping), addressing the root causes of photo-degradation more comprehensively than traditional additives. The intramolecular hydrogen bonding potential within the structure further stabilizes the molecule against thermal decomposition, ensuring it remains active throughout the processing and lifecycle of the plastic.
From a purity and impurity profile perspective, the stepwise synthesis allows for rigorous control over side reactions, which is paramount for high-clarity applications. The final structure, shown below, highlights the steric bulk provided by the tetramethyl groups on the piperidine rings, which shields the active nitrogen centers from premature deactivation while allowing access to smaller radical species. 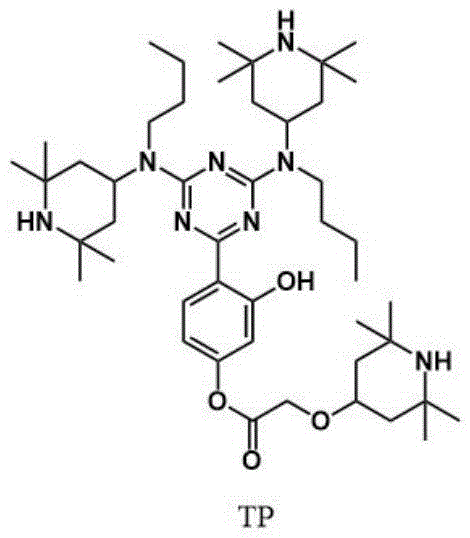 This steric protection is crucial for maintaining stabilizer activity during high-temperature extrusion processes where simpler amines might degrade. For quality control teams, the distinct NMR and mass spectrometry signatures of the intermediates provide clear analytical checkpoints, ensuring that the final product meets stringent specifications for color and ash content, which are critical for premium plastic grades used in packaging and consumer electronics.
This steric protection is crucial for maintaining stabilizer activity during high-temperature extrusion processes where simpler amines might degrade. For quality control teams, the distinct NMR and mass spectrometry signatures of the intermediates provide clear analytical checkpoints, ensuring that the final product meets stringent specifications for color and ash content, which are critical for premium plastic grades used in packaging and consumer electronics.
How to Synthesize Dopiperidine Amine Light Stabilizer Efficiently
The preparation of this advanced stabilizer involves a logical four-step sequence that leverages well-established organic transformations, making it highly accessible for scale-up. The process initiates with the controlled substitution of chlorine atoms on the triazine ring, followed by a Friedel-Crafts type alkylation to attach the phenolic component. Subsequent acylation and etherification steps install the final piperidine alcohol moiety, completing the multifunctional architecture. Each step is optimized for yield and purity, utilizing common reagents and moderate reaction conditions that minimize energy consumption and safety risks. Detailed standard operating procedures for each stage, including specific solvent systems and workup protocols, are essential for reproducing the high yields reported in the patent literature.
- Perform nucleophilic substitution of 2,4,6-trichloro-1,3,5-triazine with N-n-butyl-2,2,6,6-tetramethyl-4-piperidinamine at 70°C using NaOH catalyst.
- React the intermediate with resorcinol using anhydrous AlCl3 catalyst at 80°C to introduce the dihydroxyphenyl group.
- Conduct acylation with chloroacetyl chloride followed by substitution with 2,2,6,6-tetramethylpiperidinol using phase transfer catalysts.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the adoption of this novel stabilizer technology presents a compelling value proposition centered on operational efficiency and risk mitigation. By consolidating the functions of UV absorption and radical scavenging into a single molecule, manufacturers can drastically simplify their raw material inventory and reduce the complexity of their compounding formulations. This consolidation translates directly into cost reduction in polymer additive manufacturing, as it eliminates the logistical overhead of sourcing, storing, and handling multiple distinct stabilizer ingredients. Furthermore, the high efficiency of the dopiperidine structure means that lower loading levels may be required to achieve equivalent or superior protection compared to legacy blends, effectively stretching the purchasing power of the additive budget while maintaining product performance standards.
- Cost Reduction in Manufacturing: The synthetic pathway utilizes readily available starting materials such as cyanuric chloride and resorcinol, which are produced on a massive global scale, ensuring stable pricing and supply security. The elimination of expensive transition metal catalysts in favor of Lewis acids and phase transfer catalysts reduces the burden on downstream purification processes, thereby lowering the overall cost of goods sold. Additionally, the high yield and selectivity of the reaction steps minimize waste generation, contributing to a leaner and more economically sustainable production model that aligns with modern green chemistry principles.
- Enhanced Supply Chain Reliability: The robustness of the synthesis route, which avoids extreme pressures or cryogenic temperatures, makes it highly resilient to disruptions and easily transferable between different manufacturing sites. This flexibility is crucial for maintaining supply continuity in a volatile global market, allowing for rapid capacity expansion to meet surging demand from the automotive and construction sectors. The use of standard solvents and reagents further mitigates the risk of supply bottlenecks, ensuring that production schedules can be maintained without reliance on niche or single-source specialty chemicals.
- Scalability and Environmental Compliance: The process is designed with scalability in mind, utilizing batch reactors and separation techniques that are standard in the fine chemical industry, facilitating a smooth transition from pilot plant to commercial scale. The reduced environmental footprint, characterized by lower solvent usage and the absence of heavy metal residues, simplifies regulatory compliance and waste disposal management. This alignment with increasingly strict environmental regulations enhances the marketability of the final plastic products, appealing to eco-conscious brands and consumers who prioritize sustainability in their supply chains.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this stabilizer technology in industrial settings. These insights are derived directly from the experimental data and claims presented in the patent documentation, providing a reliable foundation for decision-making. Understanding these aspects is vital for technical teams evaluating the feasibility of integrating Compound TP into existing product lines.
Q: What distinguishes this dopiperidine amine stabilizer from conventional HALS?
A: Unlike conventional hindered amine light stabilizers (HALS) which often suffer from low active group content, this molecule integrates three piperidine amine units into a single triazine-resorcinol scaffold. This architecture provides a significantly higher density of radical-scavenging sites per molecule, enhancing anti-aging efficiency while simultaneously offering UV absorption capabilities through the conjugated system.
Q: Is the synthesis process suitable for large-scale industrial production?
A: Yes, the patented method utilizes standard organic synthesis operations such as nucleophilic substitution and acylation under moderate temperatures (60-80°C). The use of common solvents like toluene and dichloromethane, along with easily separable byproducts, ensures that the process is robust, environmentally manageable, and readily scalable from pilot batches to multi-ton commercial manufacturing without requiring exotic high-pressure equipment.
Q: How does this additive improve the mechanical properties of plastics?
A: By effectively capturing free radicals generated during UV exposure and absorbing harmful ultraviolet energy, the stabilizer prevents polymer chain scission and cross-linking degradation. Experimental data indicates that films doped with this compound retain their tensile strength and elongation at break significantly longer than untreated controls, preventing surface cracking and maintaining structural integrity over extended aging periods.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Dopiperidine Amine Light Stabilizer Supplier
As the polymer industry evolves towards higher performance and sustainability standards, the ability to source advanced additives with proven efficacy becomes a critical competitive advantage. NINGBO INNO PHARMCHEM stands at the forefront of this transition, leveraging extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production to deliver consistent, high-quality stabilizer solutions. Our commitment to excellence is underpinned by stringent purity specifications and rigorous QC labs that ensure every batch meets the demanding requirements of global plastic manufacturers. We understand that the integrity of your final product depends on the reliability of your raw materials, and we are dedicated to providing the technical support and supply security necessary for your success.
We invite you to explore how our capabilities can enhance your formulation strategy and drive value across your organization. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific application needs. We are prepared to provide specific COA data and route feasibility assessments to demonstrate how our dopiperidine amine light stabilizers can optimize your production costs while delivering superior anti-aging performance for your plastic products.
