Advanced One-Pot Palladium Catalysis for High-Purity Isoindolinone Pharmaceutical Intermediates
Advanced One-Pot Palladium Catalysis for High-Purity Isoindolinone Pharmaceutical Intermediates
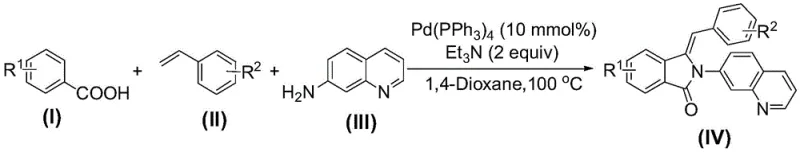
The pharmaceutical industry is constantly seeking more efficient pathways to synthesize complex heterocyclic scaffolds that serve as critical building blocks for next-generation therapeutics. Patent CN110194760B introduces a groundbreaking methodology for the preparation of 3-benzylidene-2-(7'-quinoline)-2,3-dihydro-isoindol-1-one compounds, a class of molecules with significant potential in oncology and asymmetric synthesis. This innovation leverages a transition metal-catalyzed multicomponent reaction that merges benzoic acid derivatives, styrene compounds, and 7-aminoquinoline in a single operational step. By utilizing a palladium catalyst system under mild thermal conditions, this process addresses long-standing inefficiencies in heterocycle construction, offering a streamlined route that minimizes waste and maximizes atom economy. For R&D directors and procurement specialists, this technology represents a pivotal shift towards more sustainable and cost-effective manufacturing of high-value pharmaceutical intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of complex isoindolinone derivatives has relied heavily on classical cross-coupling strategies such as the Stille or Suzuki-Miyaura reactions. While effective, these traditional pathways often necessitate the pre-functionalization of starting materials, requiring the synthesis of organotin or organoboron intermediates which are both costly and environmentally hazardous. Furthermore, these multi-step sequences typically suffer from lower overall yields due to cumulative losses at each isolation stage, and they generate substantial quantities of toxic heavy metal waste that complicates downstream purification and regulatory compliance. The requirement for stringent anhydrous conditions and expensive ligands in many of these legacy protocols further exacerbates the operational expenditure, making the commercial scale-up of complex polymer additives or drug candidates economically challenging for many manufacturers.
The Novel Approach
In stark contrast, the methodology disclosed in CN110194760B employs a direct, one-pot annulation strategy that bypasses the need for pre-activated coupling partners. By directly utilizing readily available benzoic acids and styrenes in the presence of a palladium catalyst and an organic base, the reaction achieves rapid construction of the isoindolinone core with exceptional efficiency. As illustrated in the reaction scheme, this approach tolerates a wide variety of substituents on both the aromatic acid and the styrene components, allowing for the rapid generation of diverse chemical libraries. The use of 1,4-dioxane as a solvent and triethylamine as a promoter ensures that the reaction proceeds under relatively mild conditions (100 °C), significantly reducing energy consumption compared to high-temperature pyrolysis methods often seen in older literature.
Mechanistic Insights into Pd-Catalyzed Multicomponent Cyclization
The core of this technological breakthrough lies in the sophisticated catalytic cycle mediated by tetrakis(triphenylphosphine)palladium(0). The mechanism likely initiates with the oxidative addition of the benzoic acid derivative to the Pd(0) center, followed by coordination and insertion of the styrene double bond. This sequence facilitates a cascade of C-H activation and C-N bond formation events that ultimately close the five-membered lactam ring. The presence of the 7-aminoquinoline moiety acts not only as a nucleophile but potentially assists in directing the metal center, ensuring high regioselectivity during the cyclization process. Understanding this mechanistic pathway is crucial for process chemists aiming to optimize reaction parameters, as it highlights the delicate balance between catalyst loading (10 mmol%) and base equivalents required to drive the equilibrium towards the desired product without forming oligomeric byproducts.
From an impurity control perspective, the mild nature of this catalytic system is paramount. Traditional high-energy methods often lead to thermal degradation of sensitive functional groups, resulting in complex impurity profiles that are difficult to separate. However, operating at 100 °C in a polar aprotic solvent like 1,4-dioxane minimizes side reactions such as polymerization of the styrene component or decarboxylation of the acid. The subsequent workup involving extraction with ethyl acetate and saturation with sodium chloride effectively removes inorganic salts and residual catalyst species. This results in a crude product that is amenable to straightforward purification via flash column chromatography, yielding final materials with purity levels that meet the stringent specifications required for clinical trial materials and commercial API production.
How to Synthesize 3-Benzylidene-2-(7'-quinoline)-2,3-dihydro-isoindol-1-one Efficiently
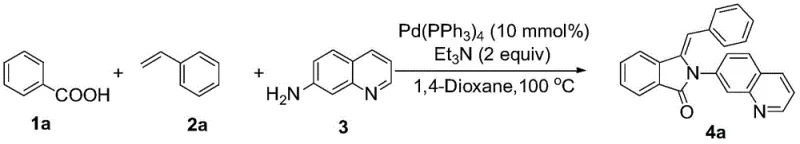
To implement this synthesis in a laboratory or pilot plant setting, operators must adhere to precise stoichiometric ratios and environmental controls to ensure reproducibility. The protocol dictates a molar ratio of benzoic acid to styrene to 7-aminoquinoline of approximately 1:1:1.2 to 1:1:1.5, ensuring that the amine component is slightly in excess to drive the amidation step to completion. The reaction is conducted under a nitrogen atmosphere to prevent oxidation of the palladium catalyst, which is critical for maintaining catalytic turnover throughout the 8-hour reaction period. Detailed standardized synthetic steps for the preparation of specific analogues, such as compound 4a, are outlined below to guide technical teams in replicating these high-yield results.
- Combine benzoic acid derivative, styrene derivative, and 7-aminoquinoline in a reaction vessel with Pd(PPh3)4 catalyst and triethylamine base.
- Add 1,4-dioxane as the solvent and heat the mixture to 100 °C under nitrogen protection for 8 hours.
- Cool the reaction, extract with ethyl acetate and saturated sodium chloride, dry over sodium sulfate, and purify via silica gel column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this patented methodology offers transformative benefits that extend far beyond simple yield improvements. By consolidating what was previously a multi-step synthesis into a single pot, the process drastically reduces the man-hours required for production and minimizes the inventory of intermediate materials that must be stored and managed. This simplification of the manufacturing workflow translates directly into substantial cost savings, as fewer unit operations mean lower utility costs, reduced solvent consumption, and decreased labor overhead. Furthermore, the reliance on commodity chemicals like benzoic acid and styrene derivatives ensures a robust and resilient supply chain, mitigating the risks associated with sourcing exotic or custom-synthesized reagents that often plague the pharmaceutical sector.
- Cost Reduction in Manufacturing: The elimination of expensive organometallic reagents and the reduction in reaction steps significantly lower the raw material costs per kilogram of finished product. Additionally, the use of a recoverable solvent system and the avoidance of toxic tin or boron byproducts reduce waste disposal fees, contributing to a leaner and more profitable production model that enhances overall margin potential for high-volume contracts.
- Enhanced Supply Chain Reliability: Since the starting materials are bulk commodities available from multiple global suppliers, the risk of supply disruption is minimized. This availability allows for flexible sourcing strategies and better negotiation leverage with vendors, ensuring that production schedules can be maintained even during periods of market volatility or logistical constraints affecting specialized chemical distributors.
- Scalability and Environmental Compliance: The reaction operates at atmospheric pressure and moderate temperatures, removing the need for specialized high-pressure reactors or cryogenic cooling systems. This ease of scale-up facilitates the transition from gram-scale R&D to tonne-scale commercial production with minimal engineering modifications, while the greener profile of the waste stream aligns perfectly with increasingly strict environmental regulations and corporate sustainability goals.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this palladium-catalyzed synthesis route. These insights are derived directly from the experimental data and process descriptions found in the patent documentation, providing a clear understanding of the operational realities and potential hurdles. By addressing these points proactively, we aim to facilitate smoother technology transfer and faster decision-making for our partners looking to integrate this chemistry into their pipelines.
Q: What are the key advantages of this Pd-catalyzed method over traditional Stille or Suzuki couplings?
A: This method eliminates the need for pre-functionalized organometallic reagents (like tin or boron species), reducing step count, waste generation, and raw material costs significantly while achieving yields up to 96%.
Q: Is this process scalable for industrial production of oncology intermediates?
A: Yes, the reaction operates at normal pressure and uses common solvents like 1,4-dioxane, making it highly suitable for commercial scale-up without requiring specialized high-pressure equipment.
Q: What is the typical purity profile of the resulting isoindolinone derivatives?
A: The process utilizes mild conditions and efficient column chromatography purification, consistently delivering high-purity solids suitable for downstream pharmaceutical applications.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3-Benzylidene-2-(7'-quinoline)-2,3-dihydro-isoindol-1-one Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of reliable supply chains for complex pharmaceutical intermediates. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project needs are met with precision and consistency. We are committed to delivering high-purity isoindolinone derivatives that adhere to stringent purity specifications, supported by our rigorous QC labs equipped with state-of-the-art analytical instrumentation to verify every batch against the highest industry standards.
We invite you to collaborate with us to leverage this advanced synthetic technology for your upcoming projects. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements. Please contact us today to request specific COA data and route feasibility assessments, and let us demonstrate how our expertise can accelerate your development timeline while optimizing your overall production costs.
