Revolutionizing Indoline Isochroman Production via Green Enzymatic Catalysis for Commercial Scale
Introduction to Advanced Enzymatic Synthesis Technologies
The landscape of fine chemical synthesis is undergoing a paradigm shift towards greener, more sustainable methodologies, particularly in the production of complex heterocyclic scaffolds essential for modern drug discovery. A pivotal development in this arena is detailed in patent CN113754673A, which discloses a novel enzymatic synthesis method for indoline isochroman derivatives. These compounds, characterized by their monoterpene alkaloid structures, are prevalent in bioactive natural products and pharmaceutical agents exhibiting antibacterial, anti-inflammatory, and anti-tumor properties. Traditionally, accessing these intricate molecular architectures has been a formidable challenge, often requiring harsh conditions and toxic reagents. However, this new technology leverages the power of multicopper oxidases, specifically CotA laccase from Bacillus licheniformis, to catalyze the oxidative coupling of 2,3-dihydroxybenzoic acid with various indole compounds. This approach not only streamlines the synthetic route but also aligns perfectly with the stringent environmental and safety standards demanded by today's global regulatory bodies, offering a robust pathway for the reliable pharmaceutical intermediate supplier seeking to modernize their portfolio.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of the indoline isochroman core has relied heavily on transition metal-catalyzed reactions, which present significant drawbacks for large-scale manufacturing. The reliance on precious or toxic metals introduces a critical contamination risk, necessitating expensive and time-consuming purification steps to meet the rigorous residual metal limits imposed by pharmacopoeias. Furthermore, traditional chemical oxidation often requires aggressive oxidants and extreme reaction conditions, such as high temperatures or strongly acidic environments, which can compromise the integrity of sensitive functional groups on the substrate. These factors collectively contribute to a lower overall process efficiency, increased waste generation, and a higher carbon footprint, making the conventional routes economically and environmentally unsustainable for the mass production of high-purity indoline isochroman derivatives needed for advanced therapeutic applications.
The Novel Approach
In stark contrast, the enzymatic strategy outlined in the patent utilizes a biomimetic approach that mimics nature's own biosynthetic pathways. By employing CotA laccase as a biocatalyst, the reaction proceeds through a highly selective oxidative coupling mechanism under remarkably mild conditions, typically at room temperature in a mixed solvent system of acetonitrile and water. This method effectively bypasses the need for transition metals, thereby eliminating the associated toxicity and purification burdens. The use of readily available starting materials, such as 2,3-dihydroxybenzoic acid and substituted indoles, combined with the high catalytic efficiency of the enzyme, results in superior yields and exceptional product purity. This green chemistry protocol represents a significant technological leap, providing a cost reduction in API manufacturing by simplifying the downstream processing and reducing the consumption of hazardous reagents.
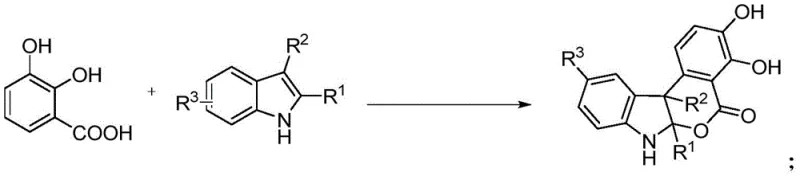
Mechanistic Insights into CotA-Catalyzed Oxidative Coupling
The core of this innovative synthesis lies in the unique catalytic cycle of the multicopper oxidase, CotA. Mechanistically, the enzyme facilitates the four-electron reduction of molecular oxygen to water while simultaneously oxidizing the phenolic substrate, 2,3-dihydroxybenzoic acid. This initial oxidation generates a highly reactive benzoquinone intermediate in situ. This electrophilic species then undergoes a regioselective nucleophilic attack by the electron-rich C3 position of the indole ring. The resulting adduct subsequently cyclizes to form the stable indoline isochroman framework. This cascade occurs with remarkable precision, driven by the enzyme's active site architecture which stabilizes the transition states and directs the stereochemical outcome. The presence of Type 1, Type 2, and Type 3 copper centers within the enzyme allows for efficient electron transfer, ensuring that the oxidation potential is perfectly matched to the substrate, minimizing side reactions and byproduct formation.
From an impurity control perspective, this enzymatic route offers distinct advantages over non-enzymatic methods. The high specificity of the biocatalyst ensures that oxidation occurs primarily at the desired phenolic hydroxyl groups, preventing over-oxidation or degradation of the indole moiety, which is a common issue with chemical oxidants. Furthermore, the mild pH and temperature conditions prevent thermal decomposition or polymerization of the reactive intermediates. The result is a cleaner reaction profile with fewer impurities, significantly reducing the burden on purification teams. For R&D directors focused on quality, this means a more predictable impurity profile and a smoother path to regulatory approval, as the absence of metal catalysts removes a major class of potential genotoxic impurities from the process.
How to Synthesize Indoline Isochroman Derivatives Efficiently
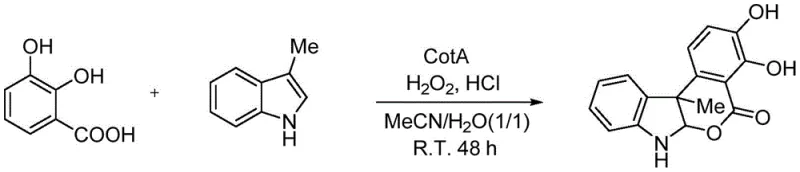
Implementing this enzymatic protocol in a laboratory or pilot plant setting is straightforward and relies on standard chemical engineering equipment. The process begins with the preparation of a reaction medium consisting of an organic solvent, preferably acetonitrile, mixed with water and a mild acid like hydrochloric acid to optimize enzyme activity and substrate solubility. Into this system, the key substrates—2,3-dihydroxybenzoic acid and the chosen indole derivative—are introduced along with the biocatalyst, CotA laccase, which can be sourced from fermented E. coli cultures. An auxiliary oxidant, such as hydrogen peroxide, is added to sustain the catalytic turnover. The reaction is allowed to proceed with stirring at ambient temperature for approximately 48 hours, ensuring complete conversion of the starting materials into the desired cyclic product.
- Prepare the reaction mixture by sequentially adding 2,3-dihydroxybenzoic acid, specific indole compounds, and Bacillus licheniformis laccase (CotA) into a solvent system comprising acetonitrile, water, and hydrochloric acid.
- Introduce an auxiliary oxidizing agent such as 30% aqueous hydrogen peroxide to facilitate the catalytic cycle and maintain the oxidation state of the enzyme active site.
- Stir the mixture at room temperature (approx. 25°C) for 48 hours to ensure complete substrate conversion, followed by extraction with ethyl acetate and purification via column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this enzymatic technology translates into tangible strategic benefits beyond mere technical novelty. The shift from precious metal catalysts to fermented enzymes fundamentally alters the cost structure of the synthesis. Enzymes like CotA can be produced via fermentation at a fraction of the cost of palladium or rhodium complexes, and they do not require the complex ligand systems that drive up reagent costs. Moreover, the elimination of heavy metal scavengers and the associated waste disposal fees leads to substantial cost savings in the overall manufacturing budget. The simplicity of the workup procedure, involving basic extraction and drying, further reduces operational expenditures related to labor and utility consumption, making the process economically attractive for high-volume production.
- Cost Reduction in Manufacturing: The replacement of expensive transition metal catalysts with biologically derived CotA laccase drastically lowers the raw material costs associated with the catalytic system. Since the enzyme is produced via fermentation, the supply is stable and scalable, avoiding the price volatility often seen with precious metals. Additionally, the mild reaction conditions reduce energy consumption for heating or cooling, and the simplified purification process minimizes the usage of expensive chromatography media or specialized scavenging resins, leading to a leaner and more cost-effective production model.
- Enhanced Supply Chain Reliability: The starting materials for this synthesis, including 2,3-dihydroxybenzoic acid and various substituted indoles, are commodity chemicals that are widely available from multiple global suppliers. This diversity in sourcing mitigates the risk of supply chain disruptions that can occur with specialized or proprietary reagents. Furthermore, the robustness of the enzymatic process allows for flexible manufacturing schedules, as the reaction is not sensitive to minor fluctuations in temperature or pressure, ensuring consistent output and reliable delivery timelines for critical pharmaceutical intermediates.
- Scalability and Environmental Compliance: Scaling this process from gram to ton scale is facilitated by the aqueous nature of the reaction medium and the absence of hazardous reagents. The process generates minimal hazardous waste, primarily consisting of aqueous streams that are easier to treat than those containing heavy metals. This aligns with increasingly strict environmental regulations and corporate sustainability goals. The ability to run the reaction at room temperature also simplifies reactor design and safety protocols, allowing for rapid scale-up without the need for specialized high-pressure or high-temperature equipment, thus accelerating time-to-market for new drug candidates.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this enzymatic synthesis platform. These insights are derived directly from the experimental data and technical specifications provided in the patent documentation, aiming to clarify the feasibility and advantages of adopting this green chemistry approach for industrial applications. Understanding these details is crucial for stakeholders evaluating the integration of biocatalysis into their existing manufacturing workflows.
Q: What are the advantages of using CotA laccase over traditional transition metal catalysts?
A: Using CotA laccase eliminates the risk of heavy metal residue in the final product, which is critical for pharmaceutical applications. It also operates under mild conditions (room temperature, aqueous/organic mix) compared to the harsh conditions often required for transition metal catalysis, reducing energy costs and safety hazards.
Q: What is the typical yield range for this enzymatic synthesis method?
A: According to the experimental data in the patent, the isolation yields for various indoline isochroman derivatives range significantly high, typically between 64% and 87%, depending on the specific substitution pattern of the indole substrate used.
Q: Is this process scalable for industrial production of API intermediates?
A: Yes, the process is highly scalable. The use of fermented enzymes (CotA produced in E. coli) ensures a consistent and cost-effective supply of the biocatalyst. The mild reaction conditions and simple workup procedures (extraction and chromatography) facilitate easy scale-up from laboratory to commercial manufacturing volumes.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Indoline Isochroman Derivative Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of the enzymatic synthesis technologies described in patent CN113754673A for the production of high-value pharmaceutical intermediates. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that our clients receive a consistent and reliable supply of critical building blocks. Our state-of-the-art facilities are equipped with rigorous QC labs capable of verifying stringent purity specifications, guaranteeing that every batch of indoline isochroman derivatives meets the highest industry standards for safety and efficacy, ready for immediate integration into your drug development pipelines.
We invite forward-thinking pharmaceutical companies to collaborate with us to leverage this cutting-edge green synthesis route. By partnering with our technical procurement team, you can access a Customized Cost-Saving Analysis tailored to your specific volume requirements and project timelines. We encourage you to reach out today to request specific COA data and comprehensive route feasibility assessments, allowing us to demonstrate how our expertise in biocatalysis can accelerate your project milestones while optimizing your overall manufacturing costs.
