Advanced Synthesis of Chalcone Pyridinium Salts for Complex Nitrogen Heterocycle Manufacturing
The pharmaceutical and fine chemical industries are constantly seeking robust methodologies to access complex nitrogen-containing scaffolds, which serve as the backbone for numerous bioactive molecules. Patent CN110423215B, published in July 2020, introduces a groundbreaking advancement in this domain by disclosing a novel class of chalcone pyridinium salts and their efficient preparation. This technology addresses the longstanding challenge of pyridine dearomatization, a critical step in synthesizing diverse heterocyclic libraries. Unlike traditional approaches that rely on harsh activation conditions, this invention leverages a unique N-alkylation strategy using a bromomethyl chalcone precursor. The resulting pyridinium salts are not merely stable intermediates but function as versatile platforms for constructing intricate nitrogen-bridged ring systems through cascade cyclization. For R&D directors and process chemists, this represents a significant leap forward in accessing chemical space that was previously difficult or expensive to explore, offering a pathway to high-value intermediates with improved atom economy and operational simplicity.
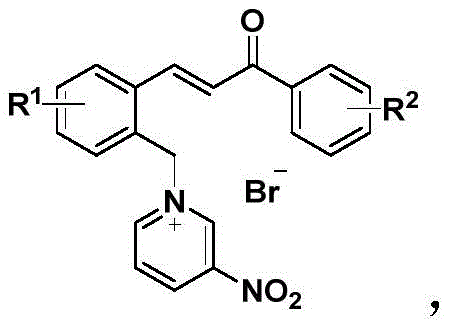
Historically, the functionalization of pyridine has been fraught with difficulties due to its inherent aromatic stability, which renders it resistant to nucleophilic attack and other transformative reactions. Conventional activation methods, such as N-acylation, N-oxidation, or coordination with Lewis acids, often necessitate the use of hazardous reagents, stringent anhydrous conditions, or cryogenic temperatures. These legacy processes frequently suffer from poor atom economy, generate substantial toxic waste, and require tedious purification steps like column chromatography to isolate the activated species. Furthermore, many traditional activated pyridine derivatives lack the necessary functionality to participate in subsequent bond-forming events without additional synthetic manipulation, leading to longer linear synthetic sequences. In contrast, the novel approach detailed in the patent circumvents these limitations by employing a bromomethyl chalcone as the alkylating agent. This strategy not only effectively breaks the aromaticity of the pyridine ring through quaternization but simultaneously installs a chalcone motif. This dual-functionality design transforms the pyridine into a highly reactive electrophile while providing a built-in handle for downstream diversification, thereby streamlining the synthesis of complex architectures.
The mechanistic elegance of this technology lies in the strategic design of the chalcone pyridinium cation, which serves as a potent electrophilic trigger for cascade reactions. Upon formation, the positive charge on the nitrogen atom significantly enhances the electron-withdrawing character of the attached chalcone system, specifically activating the β-carbon of the enone moiety towards nucleophilic attack. This activation allows for seamless engagement with double nucleophiles, such as enaminones, in a tandem sequence. The reaction proceeds through an initial Michael addition followed by intramolecular cyclization, ultimately yielding complex nitrogen-containing bridged ring compounds with high diastereoselectivity. From a purity and impurity profile perspective, this mechanism is highly favorable; the reaction conditions are mild (typically 60°C to 80°C in acetonitrile), minimizing the formation of thermal degradation byproducts or polymerization side reactions that often plague high-temperature processes. The use of 3-nitropyridine further modulates the electronic properties of the ring, stabilizing the intermediate while maintaining sufficient reactivity for the subsequent transformation. This precise control over reactivity ensures that the final bridged products are obtained with excellent structural fidelity, reducing the burden on downstream purification teams.
How to Synthesize Chalcone Pyridinium Salts Efficiently
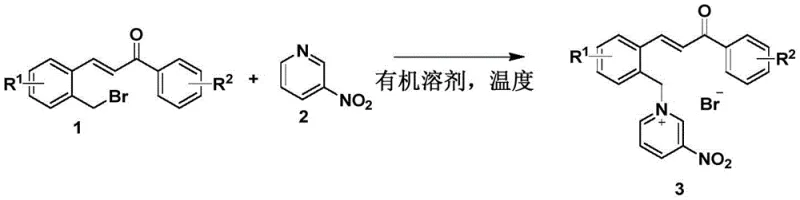
The preparation of these valuable intermediates is remarkably straightforward, designed for scalability and ease of execution in both laboratory and pilot plant settings. The protocol involves a simple nucleophilic substitution reaction where bromomethyl chalcone and 3-nitropyridine are combined in acetonitrile. The process operates at a moderate temperature of 60°C, avoiding the need for specialized heating or cooling equipment. A key advantage highlighted in the patent data is the isolation method; upon completion of the reaction, the target chalcone pyridinium salt precipitates as a yellow solid. This physical property allows for direct isolation via simple filtration through a Buchner funnel, completely bypassing the need for solvent evaporation or chromatographic separation. This operational simplicity translates directly into reduced processing time and lower solvent consumption. For detailed standard operating procedures and specific stoichiometric ratios optimized for different substituents, please refer to the standardized synthesis guide below.
- Combine bromomethyl chalcone and 3-nitropyridine in an acetonitrile solvent with a molar ratio ranging from 1: 1 to 1.5:1.
- Heat the reaction mixture to 60°C and stir for approximately 16 hours, monitoring progress via thin layer chromatography.
- Upon completion, isolate the target yellow solid product through simple filtration using a Buchner funnel, eliminating the need for complex purification.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the adoption of this synthetic route offers compelling economic and logistical benefits derived from its inherent process intensification. The elimination of complex purification steps, such as column chromatography, drastically reduces the consumption of silica gel and large volumes of elution solvents, which are significant cost drivers in fine chemical manufacturing. Furthermore, the high yields reported across a broad substrate scope (ranging from 47% to 88% for various derivatives) indicate a robust and forgiving process that maximizes raw material utilization. This efficiency minimizes the generation of chemical waste, aligning with increasingly stringent environmental regulations and reducing the costs associated with waste disposal and treatment. The use of acetonitrile, a common and readily available industrial solvent, further simplifies supply chain logistics, ensuring that raw material sourcing remains stable and cost-effective even during market fluctuations.
- Cost Reduction in Manufacturing: The process achieves significant cost optimization by removing the need for expensive transition metal catalysts or exotic activating reagents often required in traditional pyridine functionalization. The reliance on simple thermal activation in a standard solvent system lowers energy consumption and capital expenditure on specialized reactor hardware. Additionally, the direct filtration isolation method reduces labor hours and equipment occupancy time, allowing for higher throughput in existing manufacturing facilities without the need for major infrastructure upgrades.
- Enhanced Supply Chain Reliability: The starting materials, including substituted bromomethyl chalcones and nitropyridines, are commercially accessible and can be synthesized from bulk commodity chemicals. This reduces dependency on single-source suppliers for niche reagents, mitigating the risk of supply disruptions. The robustness of the reaction conditions, which tolerate a variety of electronic substituents (halogens, alkyl groups), ensures consistent production quality even if minor variations occur in raw material specifications, thereby enhancing the overall reliability of the supply chain for critical pharmaceutical intermediates.
- Scalability and Environmental Compliance: The absence of hazardous reagents and the use of mild temperatures make this process inherently safer and easier to scale from gram to ton quantities. The simplified work-up procedure reduces the volume of organic waste generated per kilogram of product, supporting sustainability goals and lowering the environmental footprint of the manufacturing process. This compliance with green chemistry principles not only reduces regulatory burdens but also enhances the marketability of the final products to environmentally conscious downstream partners in the pharmaceutical and agrochemical sectors.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this chalcone pyridinium salt technology. These insights are derived directly from the experimental data and beneficial effects described in the patent literature, providing a clear understanding of the process capabilities and limitations. Understanding these details is crucial for evaluating the feasibility of integrating this methodology into your existing production workflows or R&D pipelines.
Q: Why is direct dearomatization of pyridine challenging in organic synthesis?
A: Pyridine possesses a highly stable aromatic structure that resists direct chemical modification. Conventional methods require harsh pre-activation strategies such as N-acylation or N-oxidation, which often involve toxic reagents, extreme temperatures, or complex multi-step sequences that reduce overall process efficiency and increase waste generation.
Q: What are the primary commercial advantages of this new chalcone pyridinium salt method?
A: The patented method utilizes mild reaction conditions (60°C in acetonitrile) and achieves high yields (up to 88%) with exceptional ease of isolation. The product precipitates directly from the reaction mixture, allowing for simple filtration rather than energy-intensive column chromatography, which significantly lowers production costs and simplifies scale-up operations.
Q: How does this intermediate facilitate the construction of complex molecules?
A: Beyond activating the pyridine ring, the chalcone skeleton introduces a critical electrophilic site at the carbonyl β-position. This unique structural feature enables tandem cascade reactions with double nucleophiles like enaminones, allowing for the rapid and diastereoselective assembly of complex nitrogen-containing bridged ring systems found in advanced pharmaceutical candidates.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Chalcone Pyridinium Salt Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of this novel synthetic methodology for accessing complex nitrogen heterocycles. As a dedicated CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from bench-scale discovery to full-scale manufacturing is seamless. Our state-of-the-art facilities are equipped to handle the specific requirements of this chemistry, including rigorous QC labs that enforce stringent purity specifications to meet the demanding standards of the global pharmaceutical industry. We are committed to delivering high-quality intermediates that enable your drug discovery programs to advance without supply chain bottlenecks.
We invite you to collaborate with our technical team to explore how this technology can optimize your specific project needs. By contacting our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your volume requirements. We encourage potential partners to reach out for specific COA data and route feasibility assessments, allowing us to demonstrate how our expertise in fine chemical synthesis can drive efficiency and innovation in your supply chain.
