Advanced Regioselective Hydrogenation for Industrial Scale Isoindole Derivative Production
Introduction to Next-Generation Isoindole Synthesis
The pharmaceutical industry's relentless pursuit of potent antithrombotic agents has placed significant focus on the efficient synthesis of 2-iminopyrrolidine derivatives, particularly those serving as thrombin receptor antagonists. Patent CN101056853B introduces a groundbreaking methodology for preparing isoindole derivatives, which serve as critical precursors in this therapeutic class. The disclosed technology addresses long-standing inefficiencies in the construction of the 5,6-dialkoxy-7-fluoro-3H-isoindol-1-ylamine scaffold, a structural motif essential for high-affinity receptor binding. By leveraging a novel palladium-catalyzed regioselective hydrogenation strategy, this invention transforms a previously hazardous and low-yielding process into a robust, industrially viable pathway. For R&D directors and procurement specialists seeking a reliable pharmaceutical intermediate supplier, understanding the nuances of this patent is crucial for securing supply chains for next-generation cardiovascular therapeutics.
The significance of this technology extends beyond mere academic interest; it represents a tangible solution to the bottlenecks often encountered in the commercial scale-up of complex pharmaceutical intermediates. The traditional reliance on platinum-based catalysts has historically plagued manufacturers with issues regarding catalyst removal, product stability, and safety hazards associated with pyrophoric residues. In contrast, the methodology outlined in CN101056853B utilizes readily available palladium catalysts in conjunction with specific acidic promoters to achieve unprecedented levels of control over the reduction process. This shift not only enhances the purity profile of the resulting intermediates but also drastically simplifies the downstream processing requirements, thereby aligning perfectly with the modern demands for cost reduction in API manufacturing and environmental compliance.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Prior art methods, such as those described in WO 02/085855, typically relied on the use of platinum oxide (PtO2) for the reduction of dinitrile precursors. While chemically feasible on a small laboratory scale, these conventional approaches suffer from severe drawbacks when translated to industrial production. The primary issue lies in the strong adsorption of the product onto the platinum catalyst surface, which necessitates complex and costly purification techniques such as column chromatography to recover the target material. Furthermore, the residual platinum catalyst poses a significant safety risk due to its pyrophoric nature, requiring specialized handling procedures to prevent ignition upon exposure to air during filtration. These factors collectively contribute to low isolated yields, often reported around 20% after purification, and inflated production costs that render the process economically unviable for large-scale pharmaceutical applications.
The Novel Approach
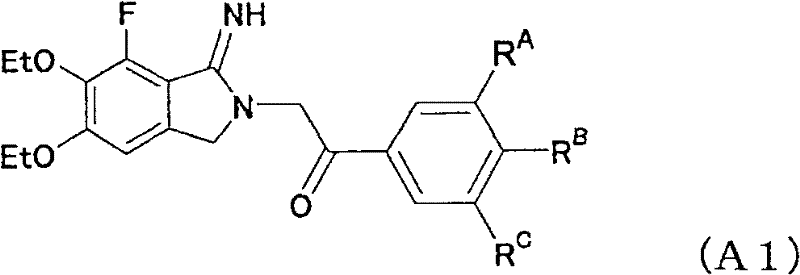
The innovative process disclosed in the patent circumvents these limitations by employing a palladium catalyst, specifically palladium hydroxide on carbon (Pd(OH)2/C) or palladium on carbon (Pd/C), in the presence of an organic or inorganic acid. This combination facilitates a highly regioselective hydrogenation where only the cyano group at the meta-position relative to the fluorine atom is reduced to an aminomethyl group, leaving the other cyano group intact. As demonstrated in the specific reaction conditions shown below, the use of methanesulfonic acid (MsOH) in 1,2-dimethoxyethane (DME) at controlled temperatures allows for the isolation of the intermediate amine salt with exceptional efficiency.
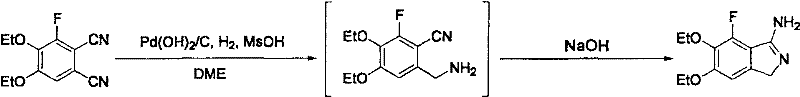
This novel approach enables the subsequent cyclization step to proceed with high yield, reportedly achieving up to 82% isolated yield in optimized examples, a substantial improvement over the prior art. The ability to purify the intermediate and final product through simple filtration, extraction, and crystallization eliminates the need for chromatographic separation, thereby reducing solvent consumption and processing time. For supply chain heads, this translates to a more predictable and reliable production timeline, while for procurement managers, it signals a potential for significant cost reduction in pharmaceutical intermediate manufacturing due to the elimination of expensive purification steps and the use of cheaper catalyst materials.
Mechanistic Insights into Acid-Promoted Regioselective Hydrogenation
The core scientific breakthrough of this patent lies in the mechanistic understanding of how acid promotion influences the selectivity of palladium-catalyzed hydrogenation on a substituted phthalonitrile substrate. In the absence of an acid, hydrogenation of dinitriles often leads to non-selective reduction, producing diamines or over-reduced species that are difficult to separate. However, the presence of an acid such as methanesulfonic acid or sulfuric acid protonates the nitrogen atoms of the cyano groups, altering their electronic interaction with the palladium surface. This protonation effect creates a steric and electronic environment that favors the reduction of the cyano group at the 1-position (meta to fluorine) over the 2-position. The reaction proceeds through a distinct intermediate, 6-aminomethyl-3,4-dialkoxy-2-fluorobenzonitrile, which is stabilized as a salt in the acidic reaction medium.
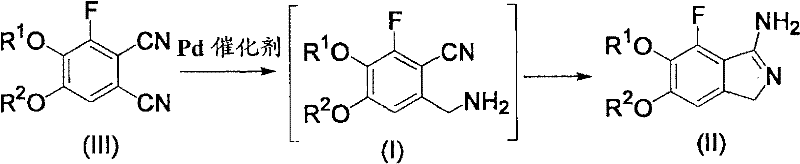
Following the selective hydrogenation, the intermediate undergoes an intramolecular cyclization in the presence of a base. This step involves the nucleophilic attack of the newly formed primary amine on the remaining nitrile group, closing the five-membered isoindole ring. The patent details that this cyclization can be quantitatively achieved using bases like sodium hydroxide or triethylamine, depending on the desired reaction temperature and kinetics. The mechanistic clarity provided by this patent allows process chemists to fine-tune reaction parameters such as hydrogen pressure (0.1 to 10 MPa) and temperature (-25 to 30°C) to maximize the formation of the desired regioisomer while minimizing impurities. This level of control is essential for maintaining stringent purity specifications required for GMP manufacturing of active pharmaceutical ingredients.
How to Synthesize 5,6-diethoxy-7-fluoro-3H-isoindol-1-ylamine Efficiently
Implementing this synthesis route requires careful attention to the stoichiometry of the acid promoter and the choice of solvent to ensure optimal regioselectivity. The process begins with the hydrogenation of 4,5-diethoxy-3-fluorophthalonitrile in a solvent system like DME, using a wet palladium catalyst to prevent runaway exotherms. The detailed standardized synthetic steps for replicating this high-yield transformation are provided in the guide below, which outlines the precise workup procedures necessary to isolate the intermediate salt before the final cyclization.
- Hydrogenate 4,5-diethoxy-3-fluorophthalonitrile using Pd(OH)2/C and methanesulfonic acid in DME at 7°C to selectively reduce the meta-cyano group.
- Filter the catalyst and adjust the pH of the filtrate to isolate the intermediate aminomethyl benzonitrile salt.
- Treat the intermediate with a base such as sodium hydroxide or triethylamine to effect cyclization and crystallize the final isoindole derivative.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this patented methodology offers transformative advantages for organizations managing the supply of complex heterocyclic intermediates. The shift from platinum to palladium catalysis is not merely a chemical substitution but a strategic operational improvement that impacts the entire value chain. By eliminating the reliance on expensive platinum oxide and the associated hazardous waste streams, manufacturers can achieve a leaner production model. The simplified purification protocol, which relies on crystallization rather than chromatography, significantly reduces the volume of organic solvents required, aligning with green chemistry principles and reducing disposal costs. These factors collectively contribute to a more sustainable and economically attractive manufacturing process.
- Cost Reduction in Manufacturing: The replacement of platinum catalysts with palladium-on-carbon variants results in direct material cost savings, as palladium is generally more cost-effective and easier to recover than platinum. Furthermore, the ability to isolate products via crystallization avoids the high operational expenses associated with large-scale chromatography, such as silica gel consumption and extensive solvent recovery. This streamlined workflow ensures that the overall cost of goods sold (COGS) for the isoindole intermediate is substantially lower compared to legacy processes, providing a competitive edge in pricing negotiations.
- Enhanced Supply Chain Reliability: The robustness of the palladium-catalyzed process under mild conditions (e.g., 7°C to 25°C) ensures consistent batch-to-batch quality, which is critical for maintaining uninterrupted supply to downstream API manufacturers. The use of stable, commercially available reagents like methanesulfonic acid and DME mitigates the risk of raw material shortages. Additionally, the improved safety profile, characterized by the absence of pyrophoric residues, reduces the likelihood of production stoppages due to safety incidents, thereby enhancing the overall reliability of the supply chain for high-purity pharmaceutical intermediates.
- Scalability and Environmental Compliance: The process is inherently scalable, having been demonstrated to work effectively with standard hydrogenation equipment found in most multipurpose chemical plants. The reduction in solvent usage and the elimination of heavy metal contamination risks simplify the environmental permitting process and waste management protocols. This ease of scale-up facilitates the transition from pilot plant quantities to commercial tonnage without the need for significant capital investment in specialized infrastructure, ensuring that the technology can meet growing market demand efficiently.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this isoindole synthesis technology. These answers are derived directly from the experimental data and claims presented in the patent documentation, providing a clear understanding of the process capabilities and limitations for potential partners and licensees.
Q: Why is the palladium-catalyzed method superior to the platinum oxide method?
A: The palladium method offers higher regioselectivity, eliminates the fire hazard associated with pyrophoric platinum residues, and allows for simpler purification via crystallization rather than chromatography.
Q: What is the key challenge in synthesizing this isoindole scaffold?
A: The primary challenge is achieving selective reduction of only one cyano group in the presence of another without over-reduction or forming complex by-products, which this patent solves using acid-promoted Pd catalysis.
Q: Can this process be scaled for commercial API production?
A: Yes, the use of robust heterogeneous catalysts like Pd/C and standard solvents like DME, combined with crystallization-based purification, makes this route highly suitable for multi-kilogram to ton-scale manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 5,6-diethoxy-7-fluoro-3H-isoindol-1-ylamine Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the development of life-saving antithrombotic therapies. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that we can meet the rigorous demands of global pharmaceutical clients. We are committed to delivering products with stringent purity specifications, supported by our rigorous QC labs that utilize advanced analytical techniques to verify every batch against the highest industry standards. Our facility is equipped to handle the specific hydrogenation and cyclization steps required by CN101056853B, guaranteeing a secure and compliant supply source for your project needs.
We invite you to collaborate with us to optimize your supply chain for isoindole derivatives. Our experts are ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements, demonstrating how our implementation of this patented technology can reduce your overall procurement costs. Please contact our technical procurement team today to request specific COA data and route feasibility assessments, and let us partner with you to accelerate the development of your next-generation thrombin receptor antagonists.
