Advanced Aromatization Strategy for High-Purity Arylalkyl Amine Intermediates
Advanced Aromatization Strategy for High-Purity Arylalkyl Amine Intermediates
The pharmaceutical and fine chemical industries are constantly seeking sustainable alternatives to petrochemical-derived intermediates, particularly for complex aromatic amines used in drug synthesis. Patent CN102382002B introduces a groundbreaking methodology for preparing 3,4-dihydroxy-5-alkylamino methyl benzoate compounds, utilizing 3-dehydroshikimic acid methyl ester as a renewable starting material. This innovation represents a significant paradigm shift from traditional transition-metal catalyzed cross-couplings to a biomimetic aromatization process driven by simple acid catalysis. By leveraging the unique structural features of shikimic acid derivatives, this route achieves high atom economy while operating under exceptionally mild conditions, typically between 20°C and 50°C. The resulting products serve as critical building blocks for potent diuretics like Bumetanide and local anesthetics, addressing the growing demand for reliable pharmaceutical intermediate supplier solutions that prioritize green chemistry principles without compromising on yield or purity standards.
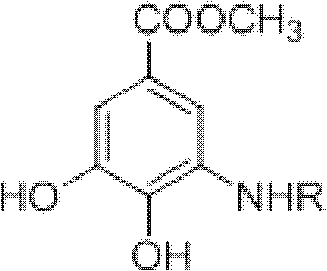
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of arylalkyl amine compounds has relied heavily on palladium or copper-catalyzed cross-coupling reactions between halogenated aromatics and aliphatic amides. These traditional pathways are fraught with significant operational and economic challenges that hinder efficient cost reduction in pharma manufacturing. For instance, palladium-catalyzed methods often require expensive and unstable organophosphorus ligands, introducing toxicity concerns and complicating downstream purification due to heavy metal residues. Alternatively, copper-catalyzed variants frequently necessitate harsh reaction temperatures ranging from 100°C to 200°C, along with excessive amounts of copper powder and highly polar, poisonous solvents. Furthermore, methods attempting to replace halohydrocarbons with alcohols often suffer from low reactivity, requiring complex transition metal catalysts based on ruthenium, iridium, or rhenium to facilitate C-N bond formation. These limitations collectively result in high production costs, substantial waste generation, and safety hazards that are increasingly unacceptable in modern regulatory environments.
The Novel Approach
In stark contrast to these legacy technologies, the novel approach disclosed in the patent utilizes 3-dehydroshikimic acid methyl ester, a derivative of the renewable non-grain biomass resource shikimic acid, to construct the aromatic core directly. This method employs a simple acid catalyst, such as p-toluenesulfonic acid or sulfuric acid, to drive a condensation-dehydrogenation reaction with primary amines, effectively aromatizing the six-membered ring framework. The process operates under normal pressure and mild thermal conditions, eliminating the need for expensive noble metals or extreme temperatures. By transforming a non-aromatic precursor into the desired aromatic system through an elegant cascade of nucleophilic addition and dehydration, this route significantly simplifies the synthetic workflow. This strategic pivot not only enhances the sustainability profile of the manufacturing process but also drastically reduces the complexity of impurity profiles, making it an ideal candidate for the commercial scale-up of complex pharmaceutical intermediates.
Mechanistic Insights into Acid-Catalyzed Condensation and Aromatization
The core of this transformative synthesis lies in the unique reactivity of the 3-dehydroshikimic acid methyl ester scaffold, which possesses a conjugated enone system primed for nucleophilic attack. When mixed with a primary amine (RNH2) in the presence of a Lewis or Brønsted acid catalyst, the carbonyl group of the ketone undergoes nucleophilic addition to form an imine intermediate, accompanied by the loss of a water molecule. This initial condensation step is followed by a crucial isomerization and dehydrogenation sequence that drives the thermodynamic stability of the final aromatic product. The acid catalyst plays a dual role: it activates the carbonyl electrophile for the initial amine attack and facilitates the subsequent elimination steps required to establish the aromatic sextet. This mechanism bypasses the high energy barriers associated with breaking strong C-Halogen bonds in traditional methods, allowing the reaction to proceed smoothly at ambient or slightly elevated temperatures.
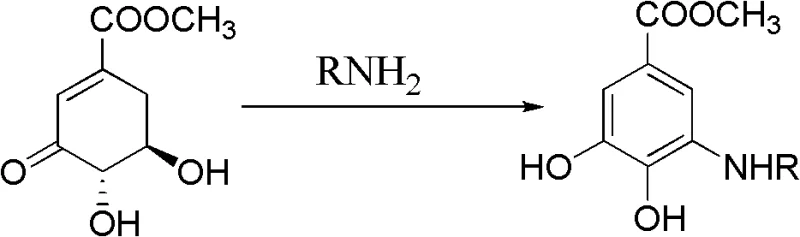
From an impurity control perspective, the mildness of this acid-catalyzed pathway offers distinct advantages over high-temperature metal-catalyzed processes. Harsh conditions often promote side reactions such as over-alkylation, polymerization, or decomposition of sensitive functional groups, leading to complex mixtures that are difficult to separate. In this novel route, the specificity of the condensation-dehydrogenation sequence ensures that the primary amine reacts selectively at the intended position on the ring. The use of common organic solvents like methanol, ethanol, or dichloromethane further aids in maintaining a homogeneous reaction environment, minimizing localized hot spots that could degrade product quality. Consequently, the crude product obtained after concentration and extraction typically exhibits high purity, reducing the burden on recrystallization steps and ensuring that the final high-purity OLED material or pharmaceutical intermediate meets stringent specifications with minimal effort.
How to Synthesize 3,4-Dihydroxy-5-Alkylamino Methyl Benzoate Efficiently
Implementing this synthesis requires careful attention to stoichiometry and solvent selection to maximize yield and operational efficiency. The patent outlines a robust protocol where 3-dehydroshikimic acid methyl ester is reacted with a slight excess of the primary amine (molar ratio 1:1.0 to 1.2) in the presence of a catalytic amount of acid (molar ratio 1:0.01 to 0.1). The reaction is typically conducted in solvents such as methanol, dichloromethane, or THF, with reaction times ranging from 2 to 6 hours depending on the specific amine substrate and temperature. Following the reaction, the workup involves standard unit operations including concentration, extraction with ethyl acetate, drying over anhydrous magnesium sulfate, and final recrystallization. For a comprehensive understanding of the specific parameters and optimization data, the detailed standardized synthesis steps are provided in the guide below.
- Mix 3-dehydroshikimic acid methyl ester with a fatty primary amine compound and an acid catalyst (such as p-toluenesulfonic acid) in an organic solvent like methanol or dichloromethane.
- Stir the reaction mixture under normal pressure at a mild temperature ranging from 20°C to 50°C for a duration of 2 to 6 hours to allow condensation and subsequent aromatization.
- Upon completion, concentrate the mixture, extract with ethyl acetate, dry the organic layer over anhydrous MgSO4, filter, and recrystallize to obtain the pure arylalkyl amine product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this biomass-based synthesis route offers compelling strategic benefits that extend beyond mere technical feasibility. By shifting away from petrochemical-derived halogenated aromatics and expensive transition metal catalysts, manufacturers can achieve substantial cost savings in raw material acquisition and waste disposal. The reliance on shikimic acid, a renewable resource with stable sources and large output potential, mitigates the volatility associated with fossil fuel pricing and supply chain disruptions. Furthermore, the elimination of heavy metals like palladium and copper removes the need for costly and time-consuming metal scavenging processes, streamlining the production timeline and reducing the overall environmental footprint of the facility.
- Cost Reduction in Manufacturing: The replacement of precious metal catalysts with inexpensive organic acids like p-toluenesulfonic acid results in a drastic reduction in catalyst costs. Additionally, the mild reaction conditions (20°C to 50°C) significantly lower energy consumption compared to traditional methods requiring heating to 100°C or higher. The simplified downstream processing, characterized by straightforward extraction and recrystallization rather than complex chromatography or metal removal, further contributes to operational expenditure optimization, ensuring a more competitive pricing structure for the final intermediates.
- Enhanced Supply Chain Reliability: Utilizing 3-dehydroshikimic acid methyl ester as a starting material diversifies the supply base away from petrochemical dependencies towards agricultural biomass, which is often more resilient to geopolitical fluctuations. The simplicity of the reaction setup, requiring only standard glass-lined or stainless steel reactors without specialized high-pressure or high-temperature capabilities, allows for flexible manufacturing across multiple sites. This flexibility ensures reducing lead time for high-purity pharmaceutical intermediates, as production can be scaled up rapidly in response to market demand without the bottleneck of specialized equipment availability.
- Scalability and Environmental Compliance: The process generates minimal hazardous waste, as it avoids the use of toxic phosphorus ligands and heavy metal salts that require special disposal protocols. The high atom economy of the condensation-dehydrogenation reaction means that a larger proportion of the starting mass is incorporated into the final product, reducing the volume of solvent and reagents needed per kilogram of output. This alignment with green chemistry principles facilitates easier regulatory approval and environmental compliance, making the commercial scale-up of complex polymer additives or drug intermediates smoother and more sustainable in the long term.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation and scope of this patented synthesis method. These answers are derived directly from the experimental data and mechanistic explanations provided in the patent documentation, ensuring accuracy and relevance for technical decision-makers. Understanding these nuances is critical for evaluating the feasibility of integrating this route into existing manufacturing portfolios.
Q: What are the advantages of using 3-dehydroshikimic acid methyl ester over traditional halogenated aromatics?
A: Using 3-dehydroshikimic acid methyl ester eliminates the need for toxic heavy metal catalysts like palladium or copper and avoids harsh reaction conditions (100-200°C). It utilizes renewable biomass resources, offering better atom economy and reduced environmental impact compared to traditional cross-coupling methods.
Q: What is the optimal temperature range for this aromatization reaction?
A: The reaction proceeds efficiently under mild conditions, specifically between 20°C and 50°C. Optimization data suggests that maintaining the temperature between 25°C and 30°C often yields the best balance between reaction rate and product purity.
Q: Can this method accommodate various alkyl amine substrates?
A: Yes, the method demonstrates broad substrate scope. It is compatible with straight-chain alkyl, branched-chain alkyl, cycloalkyl, and aryl-substituted amines, including methylamine, ethylamine, n-propylamine, isopropylamine, and benzylamine derivatives.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3,4-Dihydroxy-5-Alkylamino Methyl Benzoate Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of this biomass-derived aromatization technology for the next generation of pharmaceutical and fine chemical products. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from lab-scale discovery to industrial manufacturing is seamless and efficient. Our state-of-the-art facilities are equipped with rigorous QC labs capable of meeting stringent purity specifications, guaranteeing that every batch of 3,4-dihydroxy-5-alkylamino methyl benzoate delivered meets the highest international standards for quality and consistency.
We invite you to collaborate with our technical team to explore how this innovative synthesis route can optimize your specific supply chain requirements. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into the economic benefits of switching to this greener methodology. We encourage potential partners to contact our technical procurement team to索取 specific COA data and route feasibility assessments, allowing us to tailor a production strategy that aligns perfectly with your project timelines and budgetary goals.
