Advanced Aqueous Synthesis of Acylated Enamines for Commercial Pharmaceutical Intermediates
Introduction to Green Catalytic Technology
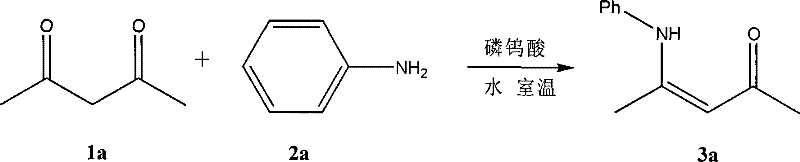
The pharmaceutical and fine chemical industries are currently undergoing a paradigm shift towards sustainable manufacturing processes, driven by both regulatory pressure and economic necessity. A pivotal development in this arena is documented in patent CN101314574B, which discloses a novel synthetic method for acylated enamines. This technology represents a significant departure from traditional protocols by utilizing pure water as the sole reaction medium and phosphotungstic acid as a highly efficient catalyst. For R&D directors and procurement strategists, this innovation offers a compelling pathway to produce high-purity pharmaceutical intermediates while simultaneously addressing critical environmental and safety concerns. The ability to conduct these condensations under mild thermal conditions without the need for hazardous organic solvents marks a substantial advancement in green chemistry, promising to redefine the cost structure and supply chain reliability for these essential building blocks.

The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the preparation of acylated enamines has relied heavily on the use of aromatic solvents to facilitate azeotropic dehydration, a process necessary to drive the equilibrium towards the desired product. As noted in prior art such as Synthesis 1983, 902, these conventional methods suffer from inherent deficiencies that pose significant challenges for modern manufacturing. The reliance on volatile organic compounds creates severe environmental liabilities, contributing to atmospheric pollution and necessitating complex waste treatment protocols. Furthermore, the flammability of these aromatic solvents introduces substantial safety hazards, requiring specialized explosion-proof infrastructure and rigorous operational controls that inflate capital expenditure. Beyond safety and environmental issues, these traditional routes often exhibit prolonged reaction times and inconsistent yields, leading to inefficient resource utilization and unpredictable production schedules that can disrupt downstream supply chains for active pharmaceutical ingredients.
The Novel Approach
In stark contrast, the methodology outlined in CN101314574B introduces a transformative approach by replacing toxic organic media with pure water, leveraging the unique properties of phosphotungstic acid to catalyze the condensation of dicarbonyl and amine compounds. This aqueous system operates effectively within a broad temperature window of 5°C to 60°C, demonstrating remarkable flexibility and robustness. The elimination of azeotropic distillation not only simplifies the reactor setup but also drastically reduces energy consumption, as there is no need to continuously remove water via high-boiling solvents. By employing a molar ratio of dicarbonyl compound to amine to catalyst of approximately 1:1.1:0.005-0.015, the process achieves high conversion rates with minimal catalyst loading. This shift to a water-based protocol fundamentally alters the economic and operational landscape, offering a cleaner, safer, and more cost-effective route for the commercial scale-up of complex pharmaceutical intermediates.
Mechanistic Insights into Phosphotungstic Acid-Catalyzed Condensation
The efficacy of this synthetic route is rooted in the unique catalytic properties of phosphotungstic acid, a heteropoly acid known for its strong Brønsted acidity and stability in aqueous environments. In the reaction mechanism, the phosphotungstic acid serves to activate the carbonyl groups of the dicarbonyl substrate through protonation, thereby increasing their electrophilicity and facilitating nucleophilic attack by the amine. The aqueous medium plays a dual role; it acts as a benign solvent that dissolves the polar transition states while potentially enhancing reaction rates through hydrophobic effects that concentrate organic reactants. This catalytic cycle proceeds smoothly without the generation of hazardous by-products, ensuring that the reaction mixture remains relatively clean throughout the process. The mild acidic conditions prevent the degradation of sensitive functional groups that might occur under harsher thermal or acidic regimes typically associated with traditional organic solvent systems, thus preserving the integrity of the molecular scaffold.
From an impurity control perspective, the use of water as a solvent provides a distinct advantage in managing the purity profile of the final acylated enamine. The high polarity of water ensures that many inorganic salts and polar by-products remain in the aqueous phase during the workup, allowing for efficient separation via simple extraction with ethyl acetate. The subsequent washing steps with saturated sodium carbonate and sodium chloride solutions further refine the organic layer, removing residual acid catalyst and trace impurities. This streamlined purification process minimizes the formation of complex impurity profiles often seen in organic solvent reactions, where side reactions such as polymerization or solvent incorporation can occur. Consequently, the resulting crude product requires less intensive purification, often yielding the target compound with high purity after standard column chromatography, which is critical for meeting the stringent specifications required for reliable pharmaceutical intermediate supplier standards.
How to Synthesize Acylated Enamine Efficiently
The practical implementation of this technology involves a straightforward sequence of mixing, reacting, and isolating, designed to maximize throughput while minimizing operational complexity. The process begins with the precise combination of the dicarbonyl and amine substrates in pure water, followed by the addition of the phosphotungstic acid catalyst. The reaction is then allowed to proceed under controlled thermal conditions, typically ranging from ambient temperature to moderate heating, depending on the specific reactivity of the substrates involved. This operational simplicity makes the method highly attractive for both laboratory optimization and large-scale manufacturing, as it does not require specialized anhydrous conditions or inert atmospheres. For detailed procedural specifics regarding stoichiometry, temperature profiling, and isolation techniques, please refer to the standardized synthesis guide below which outlines the critical parameters for reproducible results.
- Mix the dicarbonyl compound and amine compound in pure water solvent, maintaining a ratio of approximately 6-10 mL of water per 5 mmol of dicarbonyl substrate.
- Add 0.005 to 0.015 equivalents of phosphotungstic acid catalyst to the mixture and stir at a controlled temperature between 5°C and 60°C for 2 to 12 hours.
- Upon completion, extract the reaction mixture with ethyl acetate, wash the organic layer with saturated sodium carbonate and sodium chloride solutions, dry, and purify via column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this aqueous catalytic technology translates into tangible strategic benefits that extend beyond mere technical feasibility. The primary driver for value creation lies in the drastic simplification of the manufacturing process, which directly impacts the total cost of ownership for these chemical intermediates. By eliminating the need for expensive, regulated aromatic solvents and the associated recovery or disposal systems, manufacturers can achieve substantial cost savings in raw material procurement and waste management. Furthermore, the non-flammable nature of the reaction medium significantly lowers insurance premiums and safety compliance costs, creating a more resilient and economically viable production model. This efficiency gain allows suppliers to offer more competitive pricing structures without compromising on quality, aligning perfectly with the goals of cost reduction in pharmaceutical intermediates manufacturing.
- Cost Reduction in Manufacturing: The transition to a water-based system removes the financial burden associated with purchasing, storing, and disposing of large volumes of hazardous organic solvents. Traditional methods often require significant investment in solvent recovery units or incineration facilities to handle aromatic waste, whereas the aqueous effluent from this process is far easier and cheaper to treat. Additionally, the high catalytic efficiency of phosphotungstic acid means that only minute quantities are required to drive the reaction to completion, reducing the cost of goods sold related to catalyst consumption. These cumulative savings create a leaner cost structure that enhances margin potential and provides a buffer against volatility in raw material markets.
- Enhanced Supply Chain Reliability: The robustness of this synthesis method contributes to greater supply chain stability by reducing the risk of production delays caused by safety incidents or regulatory bottlenecks. Since the process operates under mild conditions without flammable solvents, the likelihood of unplanned shutdowns due to safety violations or accidents is markedly diminished. Moreover, the raw materials required, such as simple dicarbonyl compounds and amines, are widely available commodity chemicals, ensuring a steady supply flow even during market fluctuations. This reliability is crucial for maintaining continuous production schedules and meeting the just-in-time delivery expectations of global pharmaceutical clients who depend on reducing lead time for high-purity pharmaceutical intermediates.
- Scalability and Environmental Compliance: Scaling this process from pilot batches to multi-ton commercial production is inherently safer and more straightforward due to the absence of exothermic risks associated with organic solvents. The use of water facilitates better heat transfer and temperature control in large reactors, allowing for consistent product quality regardless of batch size. From an environmental standpoint, the process aligns with increasingly strict global regulations regarding volatile organic compound (VOC) emissions, future-proofing the supply chain against tightening environmental laws. This compliance advantage ensures long-term operational continuity and protects the brand reputation of partners who prioritize sustainability in their sourcing strategies.
Frequently Asked Questions (FAQ)
To assist technical teams in evaluating the feasibility of integrating this technology into their existing workflows, we have compiled a set of frequently asked questions based on the specific technical details and beneficial effects described in the patent literature. These inquiries address common concerns regarding reaction conditions, substrate scope, and purification requirements, providing a clear overview of what to expect during technology transfer. Understanding these nuances is essential for R&D directors who need to assess the compatibility of this method with their current infrastructure and quality control protocols. The following answers are derived directly from the experimental data and claims of the referenced intellectual property, ensuring accuracy and relevance for decision-making processes.
Q: What are the primary advantages of using phosphotungstic acid in water for enamine synthesis?
A: The use of phosphotungstic acid in pure water eliminates the need for toxic aromatic solvents and azeotropic dehydration processes. This results in a significantly safer operation, reduced environmental impact, and simplified downstream processing compared to conventional methods.
Q: Can this synthesis method be scaled for industrial production?
A: Yes, the method is highly suitable for industrial scale-up. The reaction operates under mild temperatures (5°C to 60°C) and uses water as a non-flammable solvent, which drastically reduces safety risks and infrastructure costs associated with large-scale organic solvent handling.
Q: What is the typical yield range for this acylated enamine synthesis?
A: According to the patent data, yields can reach up to 91% under optimized conditions (e.g., 60°C for 2 hours). The process demonstrates robust performance across various temperatures and catalyst loadings, ensuring consistent output for commercial manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Acylated Enamine Supplier
The technological breakthroughs presented in CN101314574B highlight the immense potential for greener, more efficient chemical manufacturing, a vision that NINGBO INNO PHARMCHEM is fully equipped to realize for its global partners. As a premier CDMO expert, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovative laboratory methods are seamlessly translated into robust industrial processes. Our commitment to quality is underpinned by stringent purity specifications and rigorous QC labs that utilize state-of-the-art analytical instrumentation to verify every batch. We understand that in the pharmaceutical sector, consistency is paramount, and our advanced manufacturing capabilities are designed to deliver high-purity acylated enamines that meet the exacting standards of international regulatory bodies.
We invite forward-thinking organizations to collaborate with us to leverage these advanced synthetic routes for their next-generation drug candidates. By partnering with NINGBO INNO PHARMCHEM, you gain access to a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality targets. We encourage you to contact our technical procurement team today to request specific COA data and route feasibility assessments that demonstrate how we can optimize your supply chain. Let us help you navigate the complexities of modern chemical sourcing with solutions that balance economic efficiency, environmental responsibility, and uncompromising quality.
