Scalable Catalyst-Free Synthesis of Pyrazole Heterocycles for Commercial Pharmaceutical Manufacturing
Scalable Catalyst-Free Synthesis of Pyrazole Heterocycles for Commercial Pharmaceutical Manufacturing
The pharmaceutical and fine chemical industries are constantly seeking more efficient, environmentally benign, and cost-effective synthetic routes for critical heterocyclic scaffolds. A significant breakthrough in this domain is detailed in Chinese Patent CN101372476B, which discloses a novel method for synthesizing pyrazole heterocycles under strictly catalyst-free and solvent-free conditions. This technology represents a paradigm shift from traditional Lewis acid-catalyzed processes, offering a greener alternative that aligns perfectly with modern green chemistry principles and stringent regulatory requirements for API intermediates. By eliminating the need for external catalysts and organic solvents, this method not only simplifies the downstream processing but also drastically reduces the environmental footprint associated with volatile organic compound (VOC) emissions and hazardous waste disposal. For procurement and supply chain leaders, this innovation signals a potential for substantial cost reductions and enhanced supply continuity, as the reliance on specialized catalytic reagents is completely removed from the manufacturing equation.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the preparation of pyrazole heterocycles has predominantly relied on the condensation of dicarbonyl compounds with hydrazine derivatives in the presence of Lewis acid catalysts, as documented in literature such as Tetrahedron Letters 2008, 49, 397-400. These conventional methodologies suffer from several inherent drawbacks that pose significant challenges for large-scale commercial manufacturing. Firstly, the dependence on Lewis acid catalysts introduces the risk of metal contamination in the final product, necessitating rigorous and often expensive purification steps to meet the strict residual solvent and heavy metal guidelines set by pharmacopoeias. Secondly, the use of organic solvents in these traditional routes contributes to high operational costs related to solvent purchase, recovery, and disposal, while also raising safety concerns regarding flammability and toxicity. Furthermore, the work-up procedures for catalyst removal can be cumbersome, leading to lower overall throughput and extended production cycles, which ultimately impacts the reliability of the supply chain for time-sensitive pharmaceutical projects.
The Novel Approach
In stark contrast, the methodology described in Patent CN101372476B offers a streamlined solution by conducting the condensation reaction under neat conditions, meaning no solvent is used, and without any added catalyst. The process involves simply mixing the dicarbonyl compound and the hydrazine compound in a molar ratio ranging from 1:1 to 1:2 and stirring the mixture at temperatures between 0°C and 100°C. This approach leverages the intrinsic reactivity of the substrates, allowing the reaction to proceed efficiently through thermal activation alone. The absence of solvent not only maximizes the concentration of reactants, thereby potentially accelerating the reaction rate, but also eliminates the need for solvent recovery infrastructure. The result is a process with superior atom economy, where the majority of the mass of the starting materials is incorporated into the final product, minimizing waste generation. This simplicity translates directly into operational efficiency, making it an highly attractive option for manufacturers looking to optimize their production lines for pyrazole-based intermediates.
Mechanistic Insights into Catalyst-Free Condensation Cyclization
The core of this technology lies in the direct nucleophilic attack of the hydrazine nitrogen on the carbonyl carbon of the dicarbonyl substrate, followed by cyclization and dehydration to form the pyrazole ring. Without the assistance of a Lewis acid to activate the carbonyl group, the reaction relies on the electronic properties of the substituents (R1, R2, R3, R4) and thermal energy to drive the equilibrium forward. The patent data indicates that the reaction is highly sensitive to temperature and substrate structure; for example, aliphatic dicarbonyls may react rapidly at moderate temperatures, while aromatic substrates might require higher thermal input to overcome activation barriers. Understanding these mechanistic nuances is crucial for R&D directors aiming to adapt this protocol for novel derivatives, as slight modifications in the electronic nature of the aryl or alkyl groups can influence both the reaction kinetics and the regioselectivity of the cyclization.
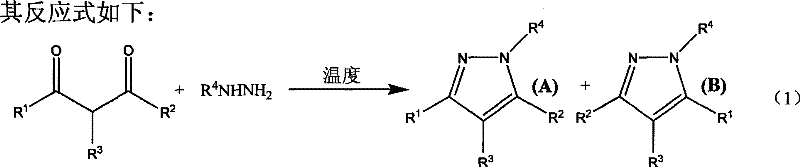
Furthermore, impurity control is inherently improved in this solvent-free system. In traditional catalytic methods, side reactions such as polymerization or over-alkylation can be promoted by the catalyst or the solvent medium. By removing these variables, the reaction profile becomes cleaner, leading to a crude product that is easier to purify. The patent highlights that the products exhibit good stereoselectivity, which is vital for pharmaceutical applications where specific isomers are required for biological activity. For instance, when unsymmetrical dicarbonyl compounds are used, the formation of regioisomers can be managed by controlling the reaction conditions, ensuring that the desired isomer is produced in high purity. This level of control reduces the burden on analytical teams and minimizes the loss of material during chromatographic separation, thereby enhancing the overall yield and economic viability of the process.
How to Synthesize Pyrazole Heterocycles Efficiently
To implement this synthesis effectively, operators must carefully monitor the reaction temperature and time, as these parameters dictate the conversion rate and product distribution. The patent provides specific examples demonstrating that lower temperatures (e.g., 0°C) may require extended reaction times (up to 12 hours) to achieve moderate yields, whereas elevating the temperature to 50°C can drastically reduce the reaction time to just 0.5 hours while significantly improving the yield. This flexibility allows manufacturers to tune the process based on their specific equipment capabilities and throughput requirements. The following guide outlines the standardized steps derived from the patent examples to ensure reproducible results.
- Mix the dicarbonyl compound and hydrazine compound in a molar ratio of 1: 1 to 1:2 under neat conditions.
- Stir the reaction mixture at a controlled temperature ranging from 0°C to 100°C for a duration of 0.5 to 12 hours depending on substrate reactivity.
- Upon completion, separate the crude product mixture using column chromatography to isolate the pure pyrazole heterocycle.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this catalyst-free technology offers compelling strategic advantages that go beyond mere technical feasibility. The elimination of catalysts and solvents fundamentally alters the cost structure of the manufacturing process, removing entire categories of raw material expenses and waste management fees. This simplification of the bill of materials (BOM) makes the supply chain more resilient to fluctuations in the prices of specialized reagents or solvents. Moreover, the operational simplicity reduces the risk of batch failures due to catalyst deactivation or solvent quality issues, ensuring a more consistent and reliable supply of high-purity intermediates for downstream API synthesis.
- Cost Reduction in Manufacturing: The most significant economic benefit stems from the complete removal of Lewis acid catalysts and organic solvents from the process. In traditional manufacturing, the cost of purchasing high-purity catalysts and the subsequent expense of removing trace metal residues to meet regulatory standards can be substantial. By eliminating these steps, the process achieves a drastic reduction in raw material costs and processing time. Additionally, the absence of solvents means there is no need for expensive solvent recovery systems or the disposal of hazardous liquid waste, leading to significant savings in environmental compliance and waste treatment overheads. This lean manufacturing approach directly contributes to a lower cost of goods sold (COGS), providing a competitive edge in the pricing of pharmaceutical intermediates.
- Enhanced Supply Chain Reliability: The robustness of this synthetic route enhances supply chain security by reducing dependency on complex reagent supply lines. Since the method utilizes readily available dicarbonyl compounds and hydrazines, sourcing is simplified, and the risk of supply disruption due to niche catalyst shortages is mitigated. The mild reaction conditions (0°C to 100°C) and the lack of sensitive catalytic species make the process less prone to variability, ensuring consistent batch-to-batch quality. This reliability is critical for long-term contracts with pharmaceutical companies, where supply continuity is often as important as price. The ability to produce high-purity products with minimal purification steps also shortens the manufacturing lead time, allowing for faster response to market demand changes.
- Scalability and Environmental Compliance: From a scalability perspective, the solvent-free nature of this reaction makes it ideal for transition from laboratory to industrial scale. Solvent-free reactions often have higher volumetric productivity since the reactor volume is not occupied by diluents, allowing for greater output per batch. Furthermore, the process aligns perfectly with increasingly stringent environmental regulations regarding VOC emissions and hazardous waste generation. By adopting this atom-economical and eco-friendly method, manufacturers can demonstrate a strong commitment to sustainability, which is becoming a key differentiator in the global chemical market. The simplified waste stream, primarily consisting of solid by-products rather than liquid solvent waste, facilitates easier and cheaper disposal, further enhancing the environmental and economic profile of the operation.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this catalyst-free pyrazole synthesis. These answers are derived directly from the experimental data and beneficial effects reported in the patent documentation, providing clarity on the practical application of this technology.
Q: What are the primary advantages of this catalyst-free method over traditional Lewis acid catalysis?
A: The primary advantage is the elimination of catalyst dependency, which removes the need for expensive metal removal steps and reduces heavy metal impurities in the final API. Additionally, the absence of solvents significantly lowers waste treatment costs and improves atom economy.
Q: How does temperature affect the yield and reaction time in this synthesis?
A: Temperature plays a critical role in kinetics; for instance, reacting acetylacetone with phenylhydrazine at 0°C requires 12 hours for 60% yield, whereas increasing the temperature to 50°C reduces the time to 0.5 hours while boosting yield to 93%.
Q: Is this process suitable for large-scale industrial production?
A: Yes, the patent explicitly states the method is suitable for industrial scale production due to its simple operation, readily available raw materials, and the absence of complex solvent recovery systems.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Pyrazole Heterocycles Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of the catalyst-free synthesis method described in Patent CN101372476B for the production of high-value pyrazole intermediates. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of this green chemistry approach are fully realized in a GMP-compliant manufacturing environment. Our state-of-the-art facilities are equipped to handle solvent-free and neat reactions safely and efficiently, while our rigorous QC labs enforce stringent purity specifications to guarantee that every batch meets the exacting standards required by the global pharmaceutical industry.
We invite you to leverage our technical expertise to optimize your supply chain for pyrazole-based APIs. By partnering with us, you gain access to a Customized Cost-Saving Analysis that quantifies the specific economic benefits of switching to this catalyst-free route for your specific molecule. We encourage you to contact our technical procurement team today to request specific COA data and route feasibility assessments, allowing us to demonstrate how our advanced manufacturing capabilities can deliver superior quality and value for your next project.
