Advanced Asymmetric Hydrogenation Technology for Commercial Scale Production of Chiral Diols
The pharmaceutical and fine chemical industries are constantly seeking more efficient pathways to access high-value chiral building blocks, particularly those serving as critical precursors for life-saving antibiotics. A groundbreaking development in this sector is detailed in Chinese Patent CN112538095B, which discloses a highly efficient method for preparing (R)-(-)-1,3-butanediol using a novel chiral tetradentate ligand and a ruthenium complex. This technology represents a significant leap forward in asymmetric catalysis, addressing long-standing challenges related to catalyst cost, recovery, and product separation that have plagued traditional synthesis routes. By enabling the direct asymmetric hydrogenation of methyl acetoacetate with exceptional stereocontrol, this innovation provides a robust foundation for the commercial scale-up of complex pharmaceutical intermediates. For R&D directors and procurement managers alike, understanding the mechanistic advantages and economic implications of this patent is crucial for optimizing supply chains and reducing manufacturing overheads in the production of carbapenem antibiotics and other fine chemicals.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of (R)-(-)-1,3-butanediol has been fraught with inefficiencies that hinder large-scale industrial application. Traditional chemical methods often rely on the direct resolution of racemic 1,3-butanediol, a process that inherently suffers from a maximum theoretical yield of only 50% due to the discard of the unwanted enantiomer, leading to substantial raw material waste and poor economic viability. Alternatively, microbiological fermentation routes, while capable of high enantioselectivity, are often limited by low space-time yields, lengthy reaction times, and complex downstream purification processes required to separate the product from the biological matrix. Furthermore, existing chemical hydrogenation methods using standard catalytic systems frequently require high catalyst loadings and struggle with the simultaneous reduction of both ketone and ester functionalities without compromising enantiopurity. These limitations result in elevated production costs and supply chain vulnerabilities, making the reliable sourcing of high-purity pharmaceutical intermediates a persistent challenge for global manufacturers.
The Novel Approach
In stark contrast to these legacy methods, the technology described in Patent CN112538095B introduces a streamlined one-pot synthesis strategy that dramatically simplifies the production workflow. This novel approach utilizes a specialized chiral ruthenium complex coordinated with a tetradentate P,P,P,N ligand to catalyze the asymmetric hydrogenation of methyl acetoacetate directly to (R)-(-)-1,3-butanediol. The reaction is promoted by a Bronsted acid additive, which enhances catalytic activity and allows for the simultaneous reduction of the carbonyl and ester groups in a single step.  This eliminates the need for multi-step protection and deprotection sequences or kinetic resolutions, thereby drastically reducing process time and solvent consumption. The ability to achieve yields and ee values exceeding 98% with minimal catalyst loading positions this method as a superior alternative for cost reduction in pharmaceutical intermediate manufacturing, offering a clear pathway to more sustainable and economical production.
This eliminates the need for multi-step protection and deprotection sequences or kinetic resolutions, thereby drastically reducing process time and solvent consumption. The ability to achieve yields and ee values exceeding 98% with minimal catalyst loading positions this method as a superior alternative for cost reduction in pharmaceutical intermediate manufacturing, offering a clear pathway to more sustainable and economical production.
Mechanistic Insights into Ru-Catalyzed Asymmetric Hydrogenation
The core of this technological breakthrough lies in the unique architecture of the chiral tetradentate ligand, which forms a highly stable chelate with the ruthenium center. Unlike traditional bidentate ligands that may dissociate under harsh reaction conditions, the P,P,P,N coordination sphere creates a rigid chiral environment that effectively controls the stereochemical outcome of the hydrogen transfer. 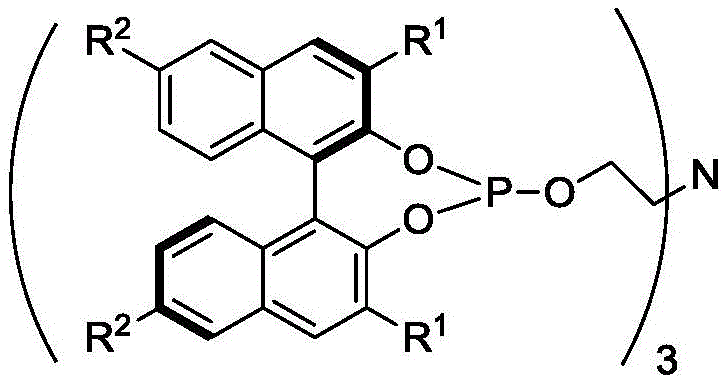 The ligand design incorporates bulky substituents on the binaphthyl backbone, such as tert-butyl or phenyl groups, which further enhance steric differentiation between the prochiral faces of the substrate. Mechanistically, the reaction proceeds via the heterolytic cleavage of hydrogen molecules to generate active Ru-H species, followed by the coordination of the substrate and subsequent hydride transfer. The presence of the Bronsted acid plays a pivotal role in facilitating proton transfer steps, thereby accelerating the overall reaction rate without sacrificing enantioselectivity. This synergistic interaction between the metal center, the tetradentate ligand, and the acid promoter ensures that the catalytic cycle remains robust even at extremely low metal-to-substrate ratios.
The ligand design incorporates bulky substituents on the binaphthyl backbone, such as tert-butyl or phenyl groups, which further enhance steric differentiation between the prochiral faces of the substrate. Mechanistically, the reaction proceeds via the heterolytic cleavage of hydrogen molecules to generate active Ru-H species, followed by the coordination of the substrate and subsequent hydride transfer. The presence of the Bronsted acid plays a pivotal role in facilitating proton transfer steps, thereby accelerating the overall reaction rate without sacrificing enantioselectivity. This synergistic interaction between the metal center, the tetradentate ligand, and the acid promoter ensures that the catalytic cycle remains robust even at extremely low metal-to-substrate ratios.
From an impurity control perspective, this catalytic system offers distinct advantages by minimizing side reactions that typically plague ester hydrogenations. The high stability of the ruthenium complex prevents the formation of metallic ruthenium particles or inactive clusters that could lead to non-selective background hydrogenation. Consequently, the formation of by-products such as (R)-3-hydroxybutyric acid methyl ester is significantly suppressed, as evidenced by comparative examples where traditional systems showed poor selectivity. The rigorous control over the reaction pathway ensures that the final product stream is exceptionally clean, simplifying the purification process and reducing the burden on quality control laboratories. For R&D teams focused on impurity profiling, this means a more predictable and manageable impurity spectrum, facilitating faster regulatory approval and smoother technology transfer to commercial manufacturing sites.
How to Synthesize (R)-(-)-1,3-Butanediol Efficiently
The implementation of this synthesis route involves a straightforward sequence of operations that can be readily adapted to existing high-pressure hydrogenation infrastructure. The process begins with the preparation of the chiral ligand through the condensation of chiral binaphthol derivatives with phosphorus trichloride and triethanolamine, followed by the in situ generation of the active catalyst. Detailed operational parameters regarding temperature, pressure, and stoichiometry are critical to achieving the reported performance metrics. The following guide outlines the standardized synthetic steps derived from the patent data to ensure reproducibility and optimal results.
- Preparation of the chiral tetradentate ligand by reacting chiral binaphthol derivatives with phosphorus trichloride and triethanolamine under basic conditions.
- In situ formation of the active chiral ruthenium complex by coordinating the ligand with a ruthenium metal precursor such as RuCl3.
- Catalytic asymmetric hydrogenation of methyl acetoacetate in the presence of a Bronsted acid promoter under high pressure to yield (R)-(-)-1,3-butanediol.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this catalytic technology translates into tangible strategic benefits that extend beyond simple yield improvements. The primary advantage lies in the drastic reduction of catalyst consumption, which directly impacts the bill of materials and reduces dependency on expensive precious metals. By operating at catalyst-to-substrate ratios as low as 1:600,000, manufacturers can achieve substantial cost savings per kilogram of product, making the process economically competitive even against bulk commodity chemicals. Furthermore, the simplicity of the reaction workup, which primarily involves distillation, minimizes the need for complex chromatographic separations or extensive aqueous washes, thereby lowering utility costs and waste treatment expenses. This efficiency gain is critical for maintaining margin integrity in a market characterized by fluctuating raw material prices and increasing regulatory pressures.
- Cost Reduction in Manufacturing: The economic impact of this technology is driven by the exceptional turnover number of the catalyst, which allows for a significant decrease in the unit cost of the active pharmaceutical ingredient precursor. Since the catalyst can be recycled multiple times without significant loss of activity, the effective cost of the ruthenium complex is amortized over a much larger volume of product. Additionally, the use of readily available starting materials like methyl acetoacetate and substituted binaphthols ensures a stable supply base, mitigating risks associated with specialty reagent shortages. The elimination of resolution steps further contributes to cost efficiency by doubling the theoretical yield compared to racemic resolution methods, effectively halving the raw material requirement for the same output.
- Enhanced Supply Chain Reliability: Implementing a robust chemical synthesis route reduces reliance on biological fermentation processes, which can be susceptible to contamination and batch-to-batch variability. The chemical method described offers consistent reaction times and predictable output, allowing for more accurate production planning and inventory management. The ability to source key ligand precursors from established chemical suppliers ensures continuity of supply, while the modular nature of the synthesis allows for flexible scaling from pilot plant to full commercial production. This reliability is essential for meeting the just-in-time delivery requirements of major pharmaceutical clients who demand uninterrupted supply of critical intermediates.
- Scalability and Environmental Compliance: The process is designed with industrial scalability in mind, utilizing standard high-pressure reactors and common solvents that are easily handled in large-scale facilities. The high atom economy of the hydrogenation reaction aligns with green chemistry principles by minimizing waste generation and reducing the environmental footprint of the manufacturing process. The simplified downstream processing reduces the volume of organic waste streams, lowering disposal costs and easing compliance with increasingly stringent environmental regulations. This sustainability profile not only enhances the corporate image but also future-proofs the supply chain against potential regulatory changes regarding solvent use and heavy metal discharge.
Frequently Asked Questions (FAQ)
To assist technical decision-makers in evaluating the feasibility of this technology for their specific applications, we have compiled a set of frequently asked questions based on the detailed experimental data provided in the patent literature. These answers address common concerns regarding catalyst performance, product quality, and process robustness, providing a clear overview of what can be expected during technology adoption. Understanding these technical nuances is vital for conducting accurate risk assessments and feasibility studies prior to committing to large-scale production campaigns.
Q: What are the advantages of the new P,P,P,N ligand system over traditional BINAP catalysts?
A: The novel tetradentate P,P,P,N ligand system offers superior stability and allows for extremely low catalyst loading ratios up to 1:600,000, significantly reducing heavy metal residue concerns and overall production costs compared to traditional bidentate systems.
Q: Can this catalytic system be recycled for industrial batch processing?
A: Yes, the patent data demonstrates that the chiral ruthenium complex can be successfully recycled and reused for at least five consecutive batches while maintaining high enantioselectivity and yield, ensuring consistent product quality.
Q: What is the expected enantiomeric excess (ee) for the final product?
A: The optimized process consistently achieves an enantiomeric excess (ee) value of greater than 98%, often reaching up to 99%, which meets the stringent purity requirements for pharmaceutical intermediates like carbapenem antibiotics.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable (R)-(-)-1,3-Butanediol Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of accessing advanced catalytic technologies to maintain a competitive edge in the global pharmaceutical market. Our team of expert chemists possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovative laboratory processes like the one described in CN112538095B can be seamlessly translated into efficient manufacturing operations. We are committed to delivering high-purity pharmaceutical intermediates that meet stringent purity specifications, supported by our rigorous QC labs and state-of-the-art analytical capabilities. Our dedication to quality and process optimization makes us an ideal partner for companies seeking to secure a stable supply of chiral building blocks for antibiotic synthesis.
We invite you to collaborate with us to explore how this advanced asymmetric hydrogenation technology can be integrated into your supply chain to drive efficiency and reduce costs. Please contact our technical procurement team to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are ready to provide specific COA data and route feasibility assessments to demonstrate how our expertise can support your project goals and accelerate your time to market.
