Scalable Industrial Synthesis of DPP-IV Inhibitor Intermediates via Novel Dehydration
Scalable Industrial Synthesis of DPP-IV Inhibitor Intermediates via Novel Dehydration
The pharmaceutical industry constantly seeks robust manufacturing pathways for critical API intermediates, particularly for diabetes treatments targeting DPP-IV inhibition. Patent CN102174007A introduces a transformative industrial preparation method for [2-(2-cyano-pyrrolidin-1-yl)-2-oxo-ethyl]-tert-butyl carbamate, a pivotal fragment in this therapeutic class. This technology addresses longstanding inefficiencies in traditional synthesis by replacing hazardous and expensive reagents with cost-effective alternatives like cyanuric chloride. For R&D directors and procurement specialists, this patent represents a significant opportunity to optimize impurity profiles while drastically reducing raw material expenditures. The disclosed methodology ensures high yield and purity without relying on complex purification techniques, making it an ideal candidate for reliable pharmaceutical intermediate supplier partnerships aiming for large-scale commercial viability.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional synthetic routes for this cyano-pyrrolidine derivative suffer from severe economic and operational bottlenecks that hinder scalable production. Prior art methods typically rely on expensive coupling agents such as EDC and activators like HOBt in excessive molar ratios, often exceeding three times the mass of the starting material, which inflates the cost of goods sold significantly. Furthermore, the dehydration step in conventional processes frequently utilizes phosphorus oxychloride in pyridine, a combination that poses serious safety risks due to the corrosive nature of the reagents and the toxicity of the solvent. The high boiling point of pyridine necessitates energy-intensive vacuum distillation for removal, while the resulting product often requires silica gel column chromatography for purification. This reliance on chromatography not only extends the production cycle but also generates substantial solvent waste, creating environmental compliance challenges and increasing the overall manufacturing footprint.
The Novel Approach
The innovative process outlined in the patent data circumvents these issues by employing a streamlined two-step sequence starting from L-prolineamide. By utilizing cyanuric chloride as a dehydrating agent, the method eliminates the need for corrosive phosphorus oxychloride and toxic pyridine, thereby reducing equipment maintenance costs and safety hazards. The reaction conditions are mild, requiring temperature control only during the addition of the dehydrating agent, after which the system warms naturally, saving energy on heating and cooling infrastructure. Crucially, the final product can be purified through simple recrystallization rather than column chromatography, which drastically shortens the processing time and minimizes solvent consumption. This approach not only enhances the safety profile of the manufacturing plant but also aligns with green chemistry principles by reducing waste generation and improving atom economy.
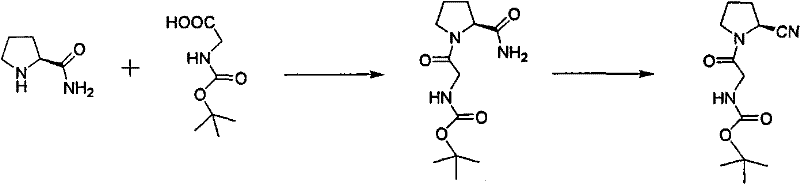
Mechanistic Insights into Cyanuric Chloride-Mediated Dehydration
The core chemical innovation lies in the efficient activation of the amide intermediate using cyanuric chloride under controlled thermal conditions. In the first step, L-prolineamide undergoes condensation with Boc-glycine using DCC and a catalytic amount of HOBt, forming the amide bond with high selectivity. The subsequent dehydration mechanism involves the activation of the primary amide by cyanuric chloride, which acts as a mild yet effective dehydrating agent to generate the nitrile functionality. Unlike harsh dehydrating agents that might cause racemization or side reactions, cyanuric chloride operates effectively at temperatures below 5°C during addition, preventing thermal degradation of the sensitive peptide backbone. The reaction mixture is then allowed to warm to room temperature naturally, driving the elimination of water to completion without the need for external heating sources, which preserves the stereochemical integrity of the chiral center essential for biological activity.
Impurity control is inherently built into this mechanistic pathway through the choice of solvents and workup procedures. The use of low-boiling solvents like isopropanol in the condensation step facilitates easy removal of urea byproducts formed from DCC, as they can be filtered off prior to concentration. In the dehydration step, the quenching of the reaction with ice water effectively hydrolyzes excess cyanuric chloride into water-soluble byproducts that are removed during the aqueous wash. This selective solubility profile ensures that the organic phase contains primarily the desired nitrile product, minimizing the formation of difficult-to-remove impurities. Consequently, the final recrystallization step yields a product with high HPLC purity, often exceeding 97%, without the need for further chromatographic separation, demonstrating the robustness of the chemical design.
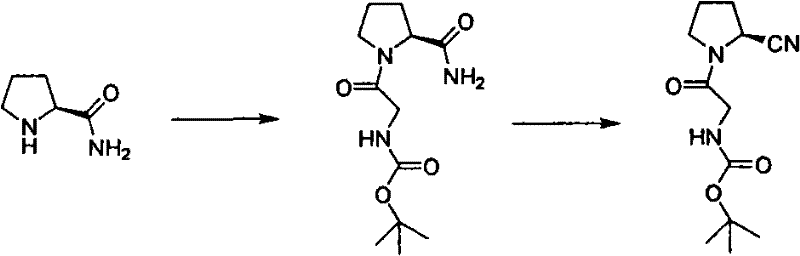
How to Synthesize 2-(2-cyano-pyrrolidin-1-yl)-2-oxo-ethyl-tert-butyl carbamate Efficiently
Implementing this synthesis requires precise adherence to the stoichiometric ratios and temperature profiles defined in the patent to ensure reproducibility and safety. The process begins with the dissolution of L-prolineamide and Boc-glycine in a suitable alcohol solvent, followed by the controlled addition of the coupling agents to manage exothermicity. Following the isolation of the amide intermediate, the dehydration step must be conducted under nitrogen protection to prevent moisture interference, with careful batch-wise addition of cyanuric chloride to maintain the reaction temperature below 5°C.
- Condense L-prolineamide with Boc-glycine using DCC and HOBt in a low-boiling solvent like isopropanol at controlled temperatures.
- Perform dehydration of the resulting amide intermediate using cyanuric chloride in DMF at low temperature followed by natural warming.
- Purify the final crude product through recrystallization using mixed solvents such as ethyl acetate and petroleum ether to achieve high purity.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, this patented methodology offers a compelling value proposition by addressing key cost drivers and logistical risks associated with traditional manufacturing. The substitution of expensive coupling reagents and hazardous solvents with commodity chemicals directly translates to a lower bill of materials, enhancing margin potential for high-volume production runs. Additionally, the elimination of column chromatography significantly reduces the demand for high-purity solvents and silica gel, which are often subject to price volatility and supply constraints. The simplified workup procedure also means faster batch turnover times, allowing manufacturing facilities to increase throughput without requiring additional capital investment in new reactor trains or purification units.
- Cost Reduction in Manufacturing: The replacement of costly activators like EDC and HOBt with optimized, lower-loading equivalents, combined with the use of inexpensive cyanuric chloride, leads to substantial raw material savings. Furthermore, the ability to recover and recycle low-boiling solvents like ethyl acetate and isopropanol reduces waste disposal costs and lowers the overall solvent purchase volume. By avoiding the use of pyridine and phosphorus oxychloride, the process also mitigates the hidden costs associated with specialized waste treatment and corrosion-resistant equipment maintenance.
- Enhanced Supply Chain Reliability: The raw materials utilized in this process, such as L-prolineamide and cyanuric chloride, are commercially available in bulk quantities from multiple global suppliers, reducing the risk of single-source dependency. The robustness of the reaction conditions means that the process is less sensitive to minor variations in raw material quality, ensuring consistent output even when sourcing from different vendors. This flexibility allows supply chain managers to negotiate better pricing and secure long-term contracts without compromising on production schedules or product quality.
- Scalability and Environmental Compliance: The absence of column chromatography and the use of standard extraction and recrystallization techniques make this process highly scalable from pilot plant to multi-ton commercial production. The reduced generation of hazardous waste and the elimination of toxic solvents align with increasingly stringent environmental regulations, simplifying the permitting process for new manufacturing lines. This environmental compatibility not only future-proofs the supply chain against regulatory changes but also enhances the corporate sustainability profile of the manufacturing partner.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this industrial preparation method. These insights are derived directly from the comparative data and experimental examples provided in the patent documentation, offering clarity on process feasibility and advantages.
Q: Why is cyanuric chloride preferred over phosphorus oxychloride for this dehydration?
A: Cyanuric chloride is significantly cheaper, less corrosive to equipment, and safer to handle than phosphorus oxychloride, eliminating the need for specialized corrosion-resistant reactors and reducing environmental hazards associated with toxic byproducts.
Q: How does this method avoid the need for silica gel column chromatography?
A:
Q: What are the solvent advantages in this new protocol?
A: The protocol utilizes low-boiling point solvents like isopropanol and ethyl acetate which are easier to recover and recycle compared to high-boiling solvents like DMF or pyridine, leading to lower energy consumption during concentration steps.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-(2-cyano-pyrrolidin-1-yl)-2-oxo-ethyl-tert-butyl carbamate Supplier
At NINGBO INNO PHARMCHEM, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that complex synthetic routes like the one described in CN102174007A are executed with precision. Our facility is equipped with rigorous QC labs capable of meeting stringent purity specifications, guaranteeing that every batch of this critical DPP-IV inhibitor intermediate meets the highest quality standards required by global pharmaceutical clients. We understand the critical nature of supply continuity and have optimized our operations to deliver consistent quality and reliability for long-term projects.
We invite you to contact our technical procurement team to discuss how we can support your specific project needs with a Customized Cost-Saving Analysis. By leveraging our expertise in process optimization, we can help you evaluate the feasibility of this route for your supply chain and provide specific COA data and route feasibility assessments tailored to your requirements. Partner with us to secure a stable, cost-effective source of high-quality pharmaceutical intermediates for your next generation of therapies.
