Scalable Industrial Preparation of Key DPP-IV Inhibitor Fragment Using Novel Dehydration
The pharmaceutical industry's relentless pursuit of effective diabetes treatments has placed significant focus on Dipeptidyl Peptidase-IV (DPP-IV) inhibitors, making the efficient synthesis of their key intermediates a critical supply chain priority. Patent CN102174007A introduces a transformative industrial preparation method for [2-(2-cyano-pyrrolidine-1-yl)-2-oxo-ethyl]-tert-butyl carbamate, a vital fragment in this therapeutic class. This technology addresses the longstanding bottlenecks of traditional synthesis by replacing hazardous and expensive reagents with safer, cost-effective alternatives like cyanuric chloride. For R&D directors and procurement managers alike, this patent represents a pivotal shift towards greener chemistry that does not compromise on yield or purity. By leveraging L-prolineamide as a starting material and optimizing the dehydration step, the process achieves high purity standards suitable for API manufacturing while drastically simplifying the operational workflow. This report analyzes the technical merits and commercial viability of this novel route, highlighting its potential to redefine cost structures in pharmaceutical intermediate production.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional synthetic routes for this cyano-pyrrolidine derivative have been plagued by severe operational and economic inefficiencies that hinder large-scale adoption. As illustrated in the prior art reaction scheme, conventional methods typically rely on expensive coupling agents like EDC (1-ethyl-(3-dimethylaminopropyl) phosphinylidyne diimine) and activators such as HOBt in excessive molar ratios, driving up raw material costs significantly. Furthermore, the dehydration step in these legacy processes often necessitates the use of phosphorus oxychloride in pyridine, a combination that requires cryogenic conditions around -35°C and poses serious corrosion risks to stainless steel reactors. The reliance on high-boiling solvents like DMF complicates downstream processing, requiring energy-intensive vacuum distillation, while the final purification frequently demands silica gel column chromatography, which is notoriously difficult to scale and generates substantial solid waste. These factors collectively create a high barrier to entry for cost-effective manufacturing, resulting in long production cycles and elevated environmental compliance burdens.
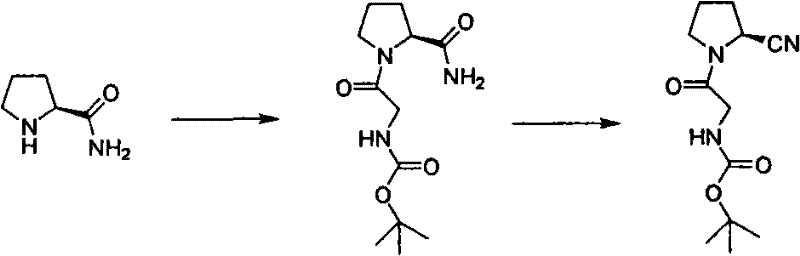
The Novel Approach
In stark contrast, the novel approach detailed in the patent utilizes a streamlined two-step sequence that prioritizes safety and scalability without sacrificing chemical efficiency. The process initiates with a condensation reaction using DCC (1,3-dicyclohexylcarbodiimide) and a catalytic amount of HOBt, where the molar ratios are tightly controlled to minimize waste while ensuring high conversion rates. The subsequent dehydration employs cyanuric chloride, a reagent that is not only inexpensive and readily available but also operates under much milder conditions, eliminating the need for extreme cooling and corrosive environments. This methodological shift allows for the use of lower boiling point solvents that are easier to recover and recycle, thereby reducing the overall energy footprint of the manufacturing process. Most critically, the crude product obtained from this route possesses sufficient purity to be upgraded to pharmaceutical grade through simple recrystallization, completely bypassing the need for column chromatography and enabling true batch production capabilities.
Mechanistic Insights into Cyanuric Chloride-Mediated Dehydration
The core innovation of this synthesis lies in the mechanistic efficiency of the dehydration step, where cyanuric chloride acts as a potent yet selective dehydrating agent for the primary amide group. Unlike phosphorus oxychloride, which proceeds through highly reactive and corrosive chlorophosphate intermediates, cyanuric chloride facilitates the elimination of water through a mechanism that generates soluble byproducts, simplifying the aqueous workup. The reaction is conducted in polar aprotic solvents like DMF or mixtures with dioxane, which stabilize the transition state and allow the reaction to proceed smoothly at temperatures near 0°C to 5°C. This thermal leniency is crucial for maintaining the stereochemical integrity of the chiral proline center, preventing racemization that could occur under harsher acidic or thermal conditions. Furthermore, the stoichiometry of the dehydrating agent is optimized to be less than equimolar in some embodiments, demonstrating a high atom economy that reduces the load on waste treatment facilities and lowers the cost of goods sold.
Impurity control in this novel pathway is achieved through a combination of selective reactivity and physical purification strategies that are inherently scalable. The initial condensation step produces dicyclohexylurea as a byproduct, which is insoluble in many organic solvents and can be removed quantitatively by simple filtration, preventing it from carrying over into the final product. In the dehydration stage, the specific choice of recrystallization solvents, such as mixtures of ethyl acetate and petroleum ether or methyl tert-butyl ether, is designed to selectively precipitate the target nitrile while keeping side products in solution. This reliance on crystallization rather than chromatography ensures that the process can be scaled from kilogram to multi-ton quantities without the linear increase in labor and silica costs associated with column purification. The result is a robust impurity profile that consistently meets stringent HPLC purity specifications, often exceeding 97%, which is essential for downstream API synthesis.
How to Synthesize [2-(2-cyano-pyrrolidine-1-yl)-2-oxo-ethyl]-tert-butyl carbamate Efficiently
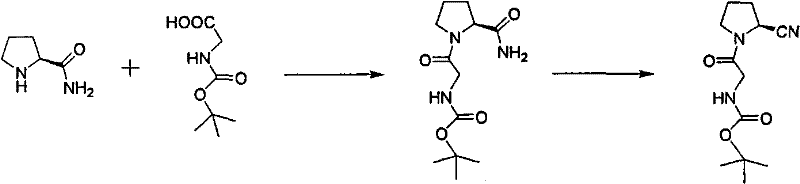
Implementing this synthesis requires precise control over reaction parameters to maximize yield and minimize the formation of side products. The process begins with the dissolution of L-prolineamide and Boc-glycine in a solvent like isopropanol or THF, followed by the controlled addition of DCC at low temperatures to manage the exotherm. Once the amide intermediate is isolated and dried, it is subjected to the dehydration conditions using cyanuric chloride, where temperature control remains critical to prevent over-reaction or degradation. The detailed standardized operating procedures for each step, including specific solvent volumes, stirring rates, and quenching protocols, are outlined in the structured guide below to ensure reproducibility across different manufacturing sites.
- Condense L-prolineamide with Boc-glycine using DCC and HOBt in a low-boiling solvent like isopropanol or THF at temperatures below 10°C.
- Filter the reaction mixture to remove dicyclohexylurea byproduct and concentrate the filtrate to obtain the amide intermediate.
- Dehydrate the intermediate using cyanuric chloride in DMF at controlled temperatures (≤5°C), followed by aqueous quenching and recrystallization.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this patented methodology offers profound advantages in terms of cost stability and operational reliability. By shifting away from volatile and expensive reagents like EDC and phosphorus oxychloride, manufacturers can secure a more predictable cost base that is less susceptible to market fluctuations in specialty chemicals. The elimination of column chromatography not only reduces the direct cost of silica gel and solvents but also dramatically shortens the production cycle time, allowing for faster turnover and improved responsiveness to market demand. Additionally, the use of non-corrosive reagents extends the lifespan of reactor vessels and reduces maintenance downtime, contributing to higher overall equipment effectiveness (OEE) in the production facility.
- Cost Reduction in Manufacturing: The substitution of expensive coupling agents and hazardous dehydrating reagents with cost-effective alternatives like cyanuric chloride leads to a substantial decrease in raw material expenditure. The ability to recycle solvents and the removal of energy-intensive distillation steps further compound these savings, resulting in a significantly lower cost of goods sold. Moreover, the simplified workup procedure reduces labor hours and utility consumption, providing a comprehensive economic benefit that enhances profit margins for high-volume production.
- Enhanced Supply Chain Reliability: The raw materials required for this process, such as L-prolineamide and Boc-glycine, are commodity chemicals with robust global supply chains, minimizing the risk of shortages. The simplified process flow reduces the number of unit operations, thereby decreasing the probability of batch failures and ensuring a consistent supply of high-quality intermediates. This reliability is crucial for maintaining uninterrupted API production schedules and meeting the strict delivery timelines demanded by pharmaceutical clients.
- Scalability and Environmental Compliance: The process is designed with inherent scalability, avoiding unit operations like column chromatography that act as bottlenecks in large-scale manufacturing. The reduced use of hazardous chemicals and the generation of less toxic waste streams simplify environmental compliance and lower the costs associated with waste disposal. This alignment with green chemistry principles not only mitigates regulatory risks but also enhances the corporate sustainability profile of the manufacturing organization.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis route. These answers are derived directly from the experimental data and beneficial effects described in the patent documentation, providing clarity on the practical aspects of the technology. Understanding these details is essential for evaluating the feasibility of adopting this method for commercial production.
Q: Why is cyanuric chloride preferred over phosphorus oxychloride for this dehydration?
A: Cyanuric chloride is significantly less corrosive to equipment and eliminates the need for hazardous pyridine solvents and extreme low temperatures (-35°C), reducing safety risks and maintenance costs.
Q: How does this process improve purity without column chromatography?
A: The optimized solvent system and specific molar ratios allow the crude product to be purified effectively through simple recrystallization, avoiding the time-consuming and costly silica gel column chromatography required in prior art.
Q: What are the scalability advantages of using DCC over EDC?
A: DCC is more cost-effective for large-scale production compared to EDC, and the resulting dicyclohexylurea byproduct is easily removed by filtration, simplifying the workup procedure for tonnage manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable [2-(2-cyano-pyrrolidine-1-yl)-2-oxo-ethyl]-tert-butyl carbamate Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of robust and scalable synthetic routes in the modern pharmaceutical supply chain. Our team of expert chemists has extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory bench to industrial reactor is seamless and efficient. We are committed to delivering high-purity intermediates that meet stringent purity specifications, supported by our rigorous QC labs and state-of-the-art analytical capabilities. By leveraging advanced technologies like the cyanuric chloride dehydration method, we can offer our partners a competitive edge through superior quality and consistent supply continuity.
We invite you to collaborate with us to optimize your supply chain for DPP-IV inhibitor production. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality standards. Please contact us to request specific COA data and route feasibility assessments, and let us demonstrate how our manufacturing expertise can drive value and efficiency in your drug development pipeline.
