Advanced Electrochemical Bromination for High-Purity Pharmaceutical Intermediates and Commercial Scale-Up
Advanced Electrochemical Bromination for High-Purity Pharmaceutical Intermediates and Commercial Scale-Up
The pharmaceutical and fine chemical industries are constantly seeking greener, more efficient pathways to synthesize critical heterocyclic building blocks. Patent CN113235116B introduces a groundbreaking electrochemical synthesis method for bromopyridine derivatives, addressing the longstanding challenges of toxicity and waste associated with traditional halogenation. This technology leverages constant current electrolysis in an undivided cell to achieve selective bromination under mild conditions, eliminating the need for stoichiometric oxidants or transition metal catalysts. For R&D directors and procurement managers, this represents a significant shift towards sustainable manufacturing, offering a route to high-purity intermediates with a simplified safety profile. The method demonstrates exceptional versatility across various substituted pyridines, ensuring robust supply chain continuity for complex drug synthesis.
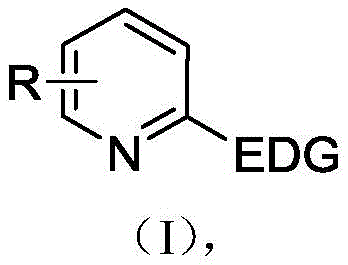
Traditional synthetic routes for aromatic bromides often rely heavily on hazardous reagents such as molecular bromine or N-bromosuccinimide (NBS), frequently requiring strong chemical oxidants like hydrogen peroxide or potassium persulfate to drive the reaction. These conventional methods invariably introduce stoichiometric amounts of metal catalysts, such as copper bromide, which complicate the purification process and raise concerns about heavy metal residues in the final active pharmaceutical ingredient (API). Furthermore, the use of unstable halogenating agents poses significant safety risks during storage and handling, while the generation of chemical waste necessitates costly disposal protocols. The inherent electron-deficient nature of pyridine rings makes them particularly resistant to electrophilic substitution, often forcing chemists to employ harsh conditions that degrade sensitive functional groups, thereby limiting the scope of accessible derivatives for drug discovery pipelines.
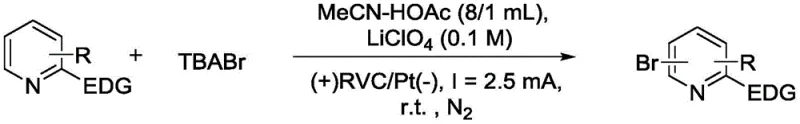
In stark contrast, the novel electrochemical approach described in the patent utilizes electricity as a clean reagent to generate reactive bromine species in situ from simple bromide salts like tetrabutylammonium bromide (TBABr). This method operates at room temperature (25°C) in a mixed solvent system of acetonitrile and acetic acid, providing a mild environment that preserves sensitive substituents on the pyridine ring. By employing common inert electrodes such as reticulated vitreous carbon (RVC) as the anode and platinum sheets as the cathode, the process avoids the complexities of electrode modification or the addition of exotic catalysts. The result is a streamlined reaction system that achieves high conversion rates with excellent atom economy, directly addressing the industry's demand for environmentally friendly and economically viable synthetic strategies for bulk chemical production.
Mechanistic Insights into Electrochemical Bromination of Pyridine Derivatives
The core mechanism of this transformation relies on the anodic oxidation of bromide ions to generate electrophilic bromine species, which subsequently attack the electron-rich positions of the pyridine ring activated by electron-donating groups. In the absence of external oxidants, the applied constant current (typically 1-10 mA) drives the regeneration of the active brominating agent at the anode surface, creating a catalytic cycle that sustains the reaction without consuming stoichiometric reagents. This electrochemical activation allows for precise control over the reaction kinetics, minimizing side reactions such as over-bromination or oxidative degradation of the substrate. The use of lithium perchlorate as the supporting electrolyte ensures sufficient conductivity in the organic solvent medium, facilitating efficient electron transfer while maintaining the stability of the reactive intermediates throughout the 6 to 20-hour reaction window.
From an impurity control perspective, the exclusion of metal catalysts is a critical advantage for pharmaceutical applications, as it inherently prevents the formation of organometallic byproducts that are difficult to remove. The reaction specificity is further enhanced by the choice of solvent and current density, which can be tuned to favor mono-bromination at the desired position, typically ortho or para to the activating amino or alkoxy groups. Post-reaction workup involves simple rotary evaporation followed by silica gel column chromatography, yielding products with high purity profiles suitable for direct use in subsequent coupling reactions. This mechanistic simplicity translates to a cleaner crude reaction mixture, reducing the burden on quality control laboratories and ensuring that the final bromopyridine derivatives meet stringent regulatory specifications for residual solvents and heavy metals.
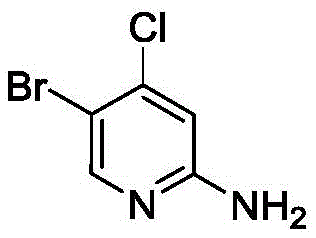
How to Synthesize 5-Bromo-2-aminopyridine Derivatives Efficiently
The synthesis protocol outlined in the patent provides a robust framework for producing a wide array of brominated pyridines, starting from readily available commercial substrates. The procedure involves charging an undivided electrolytic cell with the pyridine derivative, a bromine source like TBABr, and a supporting electrolyte in a mixed solvent system, followed by stirring under a nitrogen atmosphere to prevent moisture interference. The reaction is monitored via TLC until the starting material is consumed, after which the solvent is removed, and the residue is purified using standard flash chromatography techniques. This straightforward operational sequence minimizes the need for specialized equipment, making it accessible for both laboratory-scale optimization and pilot-plant trials.
- Prepare an undivided electrolytic cell by adding solvent (acetonitrile/acetic acid), electrolyte (LiClO4), and bromine source (TBABr).
- Add the pyridine derivative substrate and insert inert electrodes (RVC anode, Pt cathode), then stir under constant current (1-10 mA) at room temperature.
- Upon completion, remove solvent via rotary evaporation and purify the crude residue using silica gel column chromatography to isolate the target bromopyridine.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this electrochemical technology offers substantial strategic benefits by fundamentally altering the cost structure and risk profile of intermediate manufacturing. The elimination of expensive and toxic metal catalysts removes the necessity for complex scavenging steps and rigorous heavy metal testing, which are often bottlenecks in the release of pharmaceutical batches. Additionally, the use of electricity as the primary driving force reduces reliance on volatile chemical oxidants, leading to safer storage conditions and lower insurance premiums for chemical inventory. The mild reaction conditions also imply lower energy consumption for heating or cooling, contributing to a reduced carbon footprint and aligning with corporate sustainability goals without compromising on yield or throughput capabilities.
- Cost Reduction in Manufacturing: The process significantly lowers raw material costs by replacing stoichiometric oxidants and precious metal catalysts with inexpensive bromide salts and electricity. By avoiding the purchase of hazardous reagents like NBS or molecular bromine, facilities can reduce procurement expenses and minimize the costs associated with hazardous waste disposal and regulatory compliance. The simplified purification workflow further decreases operational expenditures by reducing solvent usage and labor hours required for chromatography, ultimately delivering a more cost-effective product to the market.
- Enhanced Supply Chain Reliability: Utilizing common inert electrodes and commercially available electrolytes ensures that the supply chain is not vulnerable to shortages of specialized catalysts or reagents. The robustness of the electrochemical method allows for consistent production quality regardless of batch size, mitigating the risk of supply disruptions caused by variable reaction outcomes. This reliability is crucial for maintaining continuous manufacturing lines for critical drug substances, ensuring that downstream customers receive their orders on time without unexpected delays due to synthesis failures.
- Scalability and Environmental Compliance: The undivided cell design and room temperature operation make this process highly amenable to scale-up from gram to ton quantities without significant engineering modifications. The absence of toxic byproducts and heavy metal waste simplifies environmental compliance, reducing the burden on wastewater treatment facilities and lowering the overall environmental impact of the manufacturing site. This scalability ensures that the technology can meet growing market demand for bromopyridine intermediates while adhering to increasingly strict global environmental regulations.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this electrochemical synthesis method in industrial settings. These answers are derived directly from the experimental data and beneficial effects reported in the patent documentation, providing clarity on the practical aspects of adoption. Understanding these details helps stakeholders evaluate the feasibility of integrating this green chemistry approach into their existing production portfolios.
Q: What are the primary advantages of this electrochemical bromination method over traditional chemical oxidation?
A: This method eliminates the need for stoichiometric chemical oxidants and toxic metal catalysts, significantly reducing hazardous waste generation and simplifying downstream purification processes for pharmaceutical grade intermediates.
Q: Is this electrochemical process suitable for large-scale industrial production?
A: Yes, the process utilizes common inert electrodes like reticulated vitreous carbon and operates under mild room temperature conditions, making it highly scalable and safe for commercial manufacturing without complex pressure or temperature controls.
Q: Does this method require expensive or specialized catalysts?
A: No, the reaction proceeds without any additional metal catalysts or electrode modification, relying solely on electrochemical potential to activate the bromine source, which drastically lowers raw material costs.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Bromopyridine Derivatives Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of electrochemical synthesis in modernizing the production of high-value pharmaceutical intermediates. As a dedicated CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovative laboratory methods are successfully translated into robust industrial processes. Our state-of-the-art facilities are equipped with rigorous QC labs capable of meeting stringent purity specifications, guaranteeing that every batch of bromopyridine derivatives delivered meets the highest standards of quality and consistency required by global regulatory bodies.
We invite you to collaborate with our technical procurement team to explore how this electrochemical technology can optimize your supply chain and reduce overall manufacturing costs. Please contact us to request a Customized Cost-Saving Analysis tailored to your specific project needs. We are ready to provide specific COA data and comprehensive route feasibility assessments to demonstrate how our advanced synthesis capabilities can support your long-term strategic goals in the competitive pharmaceutical landscape.
