Advanced Electro-Oxidative Modification of Tryptophan and Indole Derivatives for Commercial Scale-Up
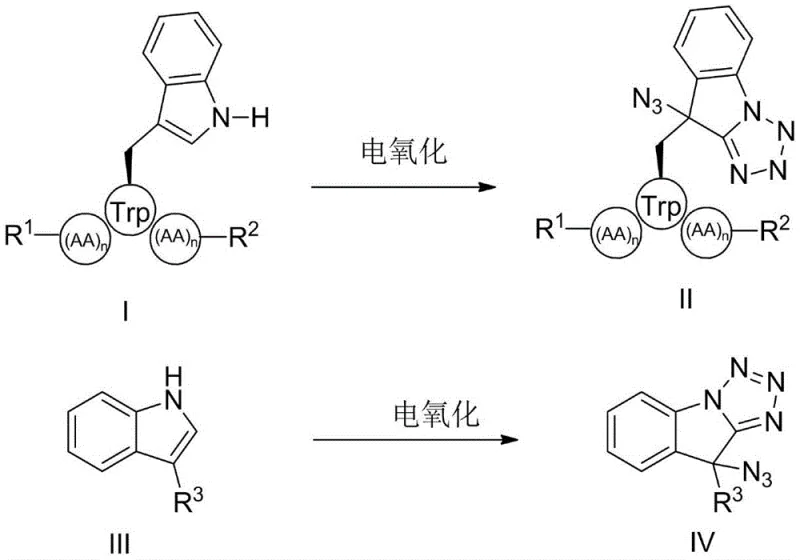
The pharmaceutical industry is constantly seeking more efficient and sustainable methods for the late-stage functionalization of bioactive molecules, particularly peptides and heterocycles. Patent CN115613061A introduces a groundbreaking approach for the modification of tryptophan and indole derivatives under electro-oxidation conditions. This technology enables the direct conversion of indole moieties into azide-substituted tetrazolo[1,5-a]indoles through a one-pot electrocatalytic process. By leveraging electricity as a clean reagent, this method circumvents the limitations of traditional stoichiometric oxidants, offering a pathway to high-purity pharmaceutical intermediates with exceptional atom economy. For R&D teams focused on peptide drug discovery, this represents a significant advancement in accessing novel chemical space around the tryptophan residue, a key amino acid found in nearly all proteins.
Tryptophan modification is a critical strategy in developing therapeutic peptides, yet conventional methods often suffer from poor selectivity and harsh reaction conditions. The disclosed invention addresses these challenges by utilizing a manganese-catalyzed electro-oxidative system that operates at room temperature. This allows for the precise installation of azide and tetrazole functionalities at the N-1, C-2, and C-3 positions of the indole ring. The resulting scaffolds are not only valuable as final active pharmaceutical ingredients but also serve as pivotal intermediates for generating diverse libraries of drug candidates through subsequent click chemistry or reduction reactions.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional C-H oxidation and redox reactions typically rely on the use of large amounts of expensive metal or non-metal oxidants, such as hypervalent iodine reagents or heavy metal salts. These conventional protocols often necessitate elevated reaction temperatures and extended reaction times, which can lead to the degradation of sensitive peptide backbones and the formation of complex impurity profiles. Furthermore, the stoichiometric generation of chemical waste from these oxidants poses significant environmental and disposal challenges, increasing the overall cost of goods sold (COGS) for manufacturing processes. The lack of site-selectivity in many traditional electrophilic substitutions also complicates the purification process, requiring extensive chromatographic separation to isolate the desired regioisomer.
The Novel Approach
In stark contrast, the electro-oxidative method described in the patent utilizes electrons as the primary oxidant, effectively replacing hazardous chemical reagents with a controllable electrical current. This approach allows the reaction to proceed under mild conditions, typically at 25°C to 30°C, preserving the integrity of complex polypeptide structures. The use of a catalytic amount of manganese species combined with a suitable ligand system ensures high reaction selectivity, targeting the tryptophan indole ring specifically even in the presence of other reactive amino acid side chains. This green chemistry protocol significantly simplifies the workup procedure, as the reaction mixture can be directly quenched and extracted, reducing the burden on downstream processing facilities.
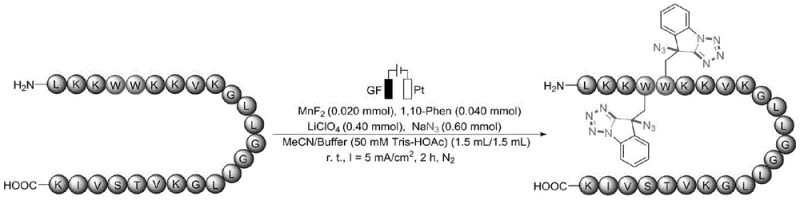
The core of this innovation lies in the synergistic interaction between the electrochemical cell setup and the homogeneous manganese catalyst. The process employs a divided or undivided cell configuration with electrode systems such as Graphite Felt (GF) as the anode and Platinum (Pt) as the cathode. The reaction medium is a sophisticated mixture of a buffer-acid system, typically Tris-HOAc adjusted to pH 2-4, and an organic co-solvent like acetonitrile. This biphasic-like environment stabilizes the reactive intermediates generated at the anode surface while maintaining the solubility of the peptide substrates. The manganese catalyst, often used at a loading of 20 mol%, cycles between oxidation states to mediate the electron transfer, facilitating the formation of the nitrogen-nitrogen bonds required for the tetrazole ring closure without the need for external chemical oxidants.
Impurity control is inherently superior in this electrochemical system due to the precise control over the oxidation potential via the applied current. By maintaining a constant current of 2 mA to 15 mA, the generation of highly reactive radical species is regulated, minimizing over-oxidation or non-specific degradation of the substrate. The patent data indicates that this method tolerates a wide range of protecting groups on the N-terminus and C-terminus of the peptides, including Acetyl, Boc, and Fmoc groups. This robustness ensures that the synthesized intermediates maintain high chemical purity, which is a critical parameter for regulatory compliance in API manufacturing. The ability to synthesize these complex heterocycles in a single pot also reduces the number of isolation steps, thereby minimizing material loss and improving the overall process mass intensity (PMI).
Mechanistic Insights into Mn-Catalyzed Electro-Oxidative Cyclization
The mechanistic pathway involves the anodic oxidation of the manganese catalyst to a high-valent species, which then activates the azide reagent and the indole substrate. The electrochemical generation of the active oxidant occurs at the electrode interface, where the manganese center facilitates the insertion of the azide group into the indole framework. This is followed by an intramolecular cyclization to form the fused tetrazolo[1,5-a]indole system. The use of ligands such as 1,10-phenanthroline is crucial for stabilizing the manganese center and tuning its redox potential to match the oxidation requirements of the specific substrate. This fine-tuning prevents the decomposition of the catalyst and ensures a sustained turnover number throughout the electrolysis period, which can last from 2 to 24 hours depending on the substrate complexity.
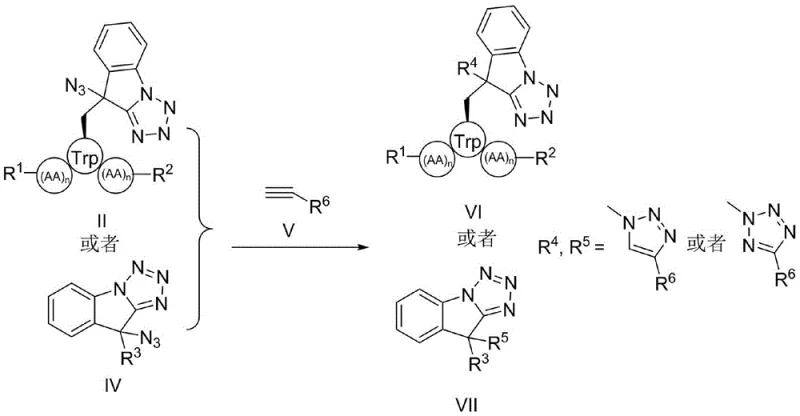
Furthermore, the versatility of the resulting azide-substituted intermediates opens up extensive opportunities for diversification. The azide functionality serves as a powerful handle for bioorthogonal chemistry, specifically the copper-catalyzed azide-alkyne cycloaddition (CuAAC). As demonstrated in the patent examples, these intermediates can react with various alkynes to form stable triazole linkages, effectively conjugating the peptide with other pharmacophores or labeling agents. Alternatively, the azide group can be reduced to a primary amine using palladium catalysts under hydrogen atmosphere, providing access to amino-functionalized indole derivatives. This modularity makes the electro-oxidative method a platform technology for generating diverse compound libraries for high-throughput screening.
How to Synthesize Azide-Substituted Tetrazolo[1,5-a]indole Efficiently
To implement this synthesis effectively, precise control over the electrochemical parameters and reagent stoichiometry is essential. The protocol requires a dry and clean reaction vessel to prevent side reactions with moisture, although the buffer system introduces a controlled amount of water. The concentration of the substrate in the reaction solvent should be maintained between 0.003 mmol/mL and 0.1 mmol/mL to ensure efficient mass transfer at the electrode surface. Detailed standard operating procedures regarding the specific assembly of the electrolytic cell and the preparation of the buffer-acid system are critical for reproducibility. For a comprehensive guide on the exact experimental steps and workup procedures, please refer to the standardized synthesis instructions below.
- Combine indole or tryptophan-containing polypeptide with azide reagent (e.g., NaN3), manganese catalyst (20 mol%), ligand (1,10-Phen), and electrolyte (LiClO4) in a buffer-acid/acetonitrile solvent system.
- Install an electrode system (e.g., Graphite Felt anode, Platinum cathode) and perform electro-oxidation at room temperature (25°C) under constant current (5 mA) for 2-24 hours.
- Quench the reaction with water, extract with dichloromethane, concentrate under reduced pressure, and purify the crude product via silica gel column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
From a procurement and supply chain perspective, this electro-oxidative technology offers substantial strategic benefits by fundamentally altering the cost structure of peptide intermediate manufacturing. The elimination of stoichiometric chemical oxidants removes a major cost driver and significantly reduces the volume of hazardous waste generated per kilogram of product. This reduction in waste disposal fees and the associated environmental compliance costs translates directly into improved margin potential for large-scale production campaigns. Additionally, the mild reaction conditions reduce the energy consumption associated with heating and cooling, further contributing to operational expenditure savings without compromising on reaction efficiency or yield.
- Cost Reduction in Manufacturing: The replacement of expensive oxidizing reagents with electricity drastically lowers the raw material costs associated with the oxidation step. Since the manganese catalyst is used in catalytic quantities and can potentially be recovered or recycled, the recurring cost of metals is minimized. The simplified workup procedure, which avoids complex quenching of dangerous oxidants, reduces labor hours and solvent consumption during the isolation phase. These factors combine to create a more economically viable process for producing high-value peptide intermediates, making it attractive for cost-sensitive generic drug programs.
- Enhanced Supply Chain Reliability: The reagents required for this process, such as sodium azide, manganese salts, and common electrolytes, are commodity chemicals with robust global supply chains. This reduces the risk of supply disruptions that are often associated with specialized or proprietary oxidizing agents. The ability to run the reaction at room temperature also lowers the infrastructure requirements for the manufacturing facility, allowing for production in a wider range of CDMO sites. This flexibility ensures a more resilient supply chain capable of meeting fluctuating demand schedules for clinical and commercial materials.
- Scalability and Environmental Compliance: Electrochemical reactions are inherently scalable, particularly when transitioning from batch to continuous flow electrochemical reactors. This scalability supports the seamless transition from gram-scale laboratory synthesis to multi-kilogram commercial production. The green nature of the process aligns perfectly with increasingly stringent environmental regulations regarding solvent use and waste discharge. By adopting this technology, manufacturers can achieve a lower E-factor and demonstrate a commitment to sustainable chemistry practices, which is a growing requirement for partnerships with major pharmaceutical companies.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this electro-oxidative modification technology. These answers are derived directly from the experimental data and scope defined in the patent literature to provide accurate guidance for process development teams. Understanding these nuances is essential for evaluating the feasibility of integrating this method into existing manufacturing workflows.
Q: What are the advantages of using electro-oxidation over traditional chemical oxidants for indole modification?
A: Electro-oxidation eliminates the need for expensive and toxic stoichiometric metal or non-metal oxidants, operates under milder conditions (room temperature), and offers higher atom economy and step efficiency compared to traditional C-H redox reactions.
Q: Can this method be applied to complex polypeptide sequences?
A: Yes, the method demonstrates high selectivity and compatibility with various protecting groups and amino acid sequences, successfully modifying tryptophan residues within complex polypeptides like octreotide without degrading the backbone.
Q: What downstream modifications are possible with the azide-substituted intermediates?
A: The resulting azide-substituted tetrazolo[1,5-a]indoles serve as versatile handles for further functionalization, including copper-catalyzed dipolar cycloaddition (Click Chemistry) to form triazoles or reductive amination to generate amine derivatives.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Tetrazolo[1,5-a]indole Supplier
NINGBO INNO PHARMCHEM stands at the forefront of adopting advanced synthetic methodologies to deliver high-quality pharmaceutical intermediates. Our technical team has extensively evaluated the electro-oxidative protocols described in CN115613061A and possesses the expertise to scale these diverse pathways from 100 kgs to 100 MT/annual commercial production. We understand that consistency is key in API manufacturing, which is why our facilities are equipped with rigorous QC labs and stringent purity specifications to ensure every batch meets the highest international standards. Our experience in handling complex peptide modifications ensures that we can navigate the intricacies of electrochemical scale-up safely and efficiently.
We invite you to collaborate with us to leverage this cutting-edge technology for your next drug development project. Our team is ready to provide a Customized Cost-Saving Analysis tailored to your specific molecule, demonstrating how this green chemistry approach can optimize your budget. Please contact our technical procurement team today to request specific COA data and route feasibility assessments. Let us help you accelerate your timeline to market with reliable, cost-effective, and sustainable synthesis solutions.
