Advanced C(sp3)-C(sp2) Cross-Coupling Technology for Commercial Scale-Up of Complex Pharmaceutical Intermediates
Advanced C(sp3)-C(sp2) Cross-Coupling Technology for Commercial Scale-Up of Complex Pharmaceutical Intermediates
The development of efficient synthetic routes for alkyl-substituted heterocycles remains a critical challenge in modern organic chemistry, particularly for the production of high-value pharmaceutical intermediates and electronic materials. Patent CN109790079B introduces a groundbreaking methodology for C(sp3)-C(sp2) cross-coupling reactions that utilizes organozinc reagents and heterocyclic (pseudo)halides. This technology addresses the longstanding limitations associated with coupling sterically hindered branched alkyl chains to heteroaromatic rings, a transformation that is notoriously difficult using traditional Grignard or nickel-catalyzed protocols. By leveraging a palladium-catalyzed system with specifically activated zinc species, this invention enables the synthesis of complex structures with exceptional yields and purity profiles.
The significance of this patent lies in its ability to bypass the instability issues inherent to sp3-hybridized coupling partners. Conventional methods often suffer from beta-elimination or demetallation, leading to poor mass balance and difficult purification scenarios. The disclosed method employs a robust catalytic cycle involving Pd(dppf)Cl2 and a variety of polar aprotic solvents such as DMA or DMF, ensuring that even sensitive functional groups remain intact throughout the reaction. This represents a substantial leap forward for process chemists seeking reliable pathways to access diverse chemical space for drug candidates and functional materials.

In the context of industrial application, the ability to couple branched alkyl chains efficiently is paramount for tuning the physicochemical properties of active pharmaceutical ingredients (APIs) and organic semiconductors. The patent details a comprehensive scope that includes not only simple alkylations but also the incorporation of functionalized chains containing esters, nitriles, and halides. This versatility makes the technology a powerful tool for the reliable pharmaceutical intermediate supplier looking to expand their portfolio of accessible building blocks without compromising on quality or scalability.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the alkylation of heterocycles such as 3-bromothiophene has relied heavily on nickel-catalyzed Kumada couplings using Grignard reagents. However, this approach faces severe limitations when applied to branched alkyl chains. The preparation of Grignard reagents from branched alkyl halides is fraught with difficulties, including slow initiation, competing Wurtz coupling, and thermal instability. Furthermore, the subsequent cross-coupling step is often plagued by low conversion rates, with reported yields frequently hovering around 50% or lower. These inefficiencies are compounded by the formation of stubborn by-products, such as alkanes from self-coupling and alkenes from beta-elimination, which possess physical properties nearly identical to the desired product.
The purification of these reaction mixtures presents a significant bottleneck in manufacturing. Because the by-products often have large Rf values on silica gel and extremely high boiling points, they cannot be easily separated by standard vacuum distillation or flash chromatography. This results in prolonged processing times, increased solvent consumption, and ultimately, a higher cost of goods sold (COGS). For procurement managers, these inefficiencies translate into supply chain vulnerabilities, where batch-to-batch consistency is hard to maintain, and the risk of failing purity specifications is elevated. The reliance on nickel catalysts also introduces potential heavy metal contamination issues that require additional scavenging steps to meet regulatory standards for pharmaceutical applications.
The Novel Approach
The methodology disclosed in CN109790079B offers a transformative solution by replacing unstable Grignard reagents with more robust organozinc species. The core innovation involves the in situ generation of organozinc reagents using activated zinc powder and a metal iodide catalyst, such as potassium iodide (KI), in polar solvents like N,N-Dimethylacetamide (DMA). This activation strategy ensures rapid and complete insertion of zinc into the carbon-halogen bond, even for sterically demanding substrates like 9-(bromomethyl)nonadecane. The resulting organozinc reagents are sufficiently stable to be handled and transferred, yet reactive enough to undergo efficient transmetallation with the palladium catalyst.
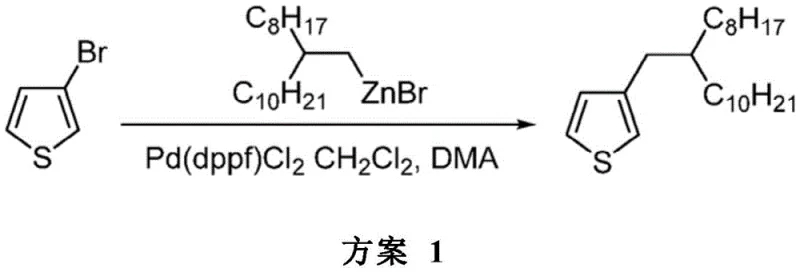
Experimental data within the patent demonstrates the superiority of this approach, with isolated yields consistently reaching 95% for model reactions such as the synthesis of 3-(2-octyldodecyl)thiophene. Unlike the messy mixtures produced by nickel catalysis, the palladium-catalyzed organozinc coupling generates minimal by-products, allowing for straightforward purification via simple filtration and column chromatography. This drastic simplification of the workup procedure not only reduces operational complexity but also significantly enhances the overall throughput of the manufacturing process. For supply chain heads, this means a more predictable production schedule and a reliable source of high-purity intermediates that meet stringent quality requirements.
Mechanistic Insights into Pd-Catalyzed Organozinc Cross-Coupling
The success of this reaction hinges on the precise interplay between the palladium catalyst, the ligand system, and the organozinc nucleophile. The catalytic cycle likely initiates with the oxidative addition of the heterocyclic halide to the Pd(0) species generated from the precatalyst Pd(dppf)Cl2. The use of the bidentate ligand dppf (1,1'-bis(diphenylphosphino)ferrocene) is critical, as it stabilizes the palladium center and facilitates the subsequent transmetallation step. Unlike monodentate ligands, dppf provides a favorable bite angle that accommodates the steric bulk of the branched alkyl-zinc species, preventing catalyst deactivation and promoting efficient bond formation.
Following oxidative addition, the transmetallation of the alkyl group from zinc to palladium occurs. This step is typically the rate-determining factor in sp3-sp2 couplings due to the slow kinetics of alkyl transfer. However, the presence of iodide ions from the metal iodide activator (e.g., KI, NaI) plays a crucial role in accelerating this process, potentially by forming more reactive halo-zincate complexes or by facilitating halogen exchange on the palladium center. The final reductive elimination step releases the coupled product and regenerates the active Pd(0) catalyst, completing the cycle. The high yields observed across a wide range of substrates suggest that this mechanism effectively suppresses competitive beta-hydride elimination, a common pathway for decomposition in alkyl-palladium intermediates.
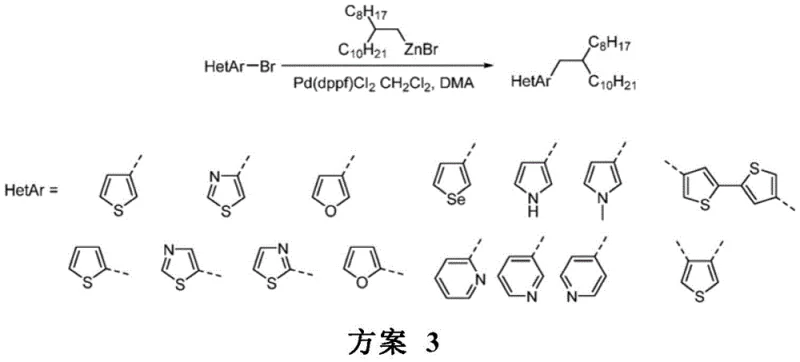
Impurity control is inherently built into this mechanistic framework. By avoiding the highly basic conditions associated with Grignard reagents, the process minimizes base-sensitive side reactions such as deprotonation of the heterocycle or hydrolysis of sensitive functional groups. Furthermore, the mild reaction conditions (typically 80°C in DMA) prevent thermal degradation of the product. The patent highlights the compatibility of various functional groups, including esters, nitriles, and halides, which would be incompatible with traditional organolithium or Grignard chemistries. This functional group tolerance allows for late-stage functionalization strategies, providing R&D directors with greater flexibility in designing synthetic routes for complex drug molecules.
How to Synthesize Alkylated Heterocycles Efficiently
The synthesis protocol outlined in the patent provides a clear roadmap for implementing this technology in a laboratory or pilot plant setting. The process begins with the activation of commercial zinc dust using dilute hydrochloric acid, followed by thorough washing and drying to remove surface oxides. This activated zinc is then reacted with the alkyl halide and a catalytic amount of potassium iodide in DMA at 80°C to generate the organozinc reagent. Once formed, this reagent is added dropwise to a solution of the heterocyclic bromide and the palladium catalyst. The detailed standardized synthesis steps for this procedure are provided in the guide below.
- Activate zinc powder by treating with acid, followed by filtering, washing, and drying to ensure high reactivity.
- Prepare the organozinc reagent by reacting the activated zinc with an alkyl halide and a metal iodide catalyst in a polar solvent like DMA.
- Perform the cross-coupling reaction by adding the organozinc reagent to a mixture of the heterocyclic halide and Pd(dppf)Cl2 catalyst, then purify via column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
Adopting this patented cross-coupling technology offers substantial strategic benefits for procurement and supply chain management, primarily driven by the simplification of the manufacturing process and the improvement in material efficiency. The shift from nickel-catalyzed Grignard couplings to palladium-catalyzed organozinc couplings eliminates the need for cryogenic conditions and the handling of pyrophoric reagents, thereby reducing safety risks and infrastructure costs. The high selectivity of the reaction means that raw materials are converted into the desired product with minimal waste, directly impacting the cost of goods sold through improved atom economy.
- Cost Reduction in Manufacturing: The elimination of difficult-to-prepare Grignard reagents and the avoidance of complex purification steps like vacuum distillation lead to significant operational savings. The process utilizes readily available starting materials such as alkyl bromides and heterocyclic bromides, which are commodity chemicals with stable pricing. Furthermore, the high yields (often exceeding 90%) reduce the amount of starting material required per kilogram of product, effectively lowering the material cost basis. The simplified workup, involving basic filtration and extraction, reduces solvent usage and labor hours, contributing to a leaner and more cost-effective production model.
- Enhanced Supply Chain Reliability: The robustness of the organozinc reagents allows for more flexible scheduling and inventory management. Unlike Grignard reagents which must be prepared immediately before use, the organozinc species in this protocol demonstrate sufficient stability to be generated and utilized in a controlled manner. This reliability ensures consistent batch quality and reduces the risk of production delays caused by reagent failure. Additionally, the use of common solvents like DMA and DMF ensures that supply chain disruptions for specialized reagents are minimized, securing a steady flow of materials for continuous manufacturing operations.
- Scalability and Environmental Compliance: The process is designed with scalability in mind, utilizing standard reactor equipment and moderate temperatures that are easily managed on a multi-ton scale. The reduction in hazardous by-products and the avoidance of heavy metal contaminants like nickel simplify waste treatment and disposal, aligning with increasingly strict environmental regulations. The ability to produce high-purity intermediates without extensive chromatographic purification on a large scale makes this technology ideal for commercial manufacturing, ensuring that supply can meet demand without compromising on environmental stewardship or regulatory compliance.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this cross-coupling technology. These answers are derived directly from the experimental data and scope defined in the patent documentation, providing clarity on substrate compatibility, reaction conditions, and product quality. Understanding these details is essential for technical teams evaluating the feasibility of integrating this method into existing synthetic workflows.
Q: Why are organozinc reagents preferred over Grignard reagents for branched alkyl chains?
A: Grignard reagents derived from branched alkyl chains are extremely difficult to synthesize and handle due to steric hindrance and instability. In contrast, the organozinc reagents described in this patent are more stable and tolerate the steric bulk of branched chains, leading to significantly higher yields (up to 95%) compared to traditional nickel-catalyzed methods which often struggle to exceed 50%.
Q: What types of heterocyclic substrates are compatible with this coupling method?
A: The method demonstrates broad substrate scope, successfully coupling with various heterocyclic halides including thiophenes, thiazoles, furans, selenophenes, and pyrroles. It is particularly effective for 3-bromothiophene and 4-bromothiazole derivatives, which are common building blocks in drug discovery and organic electronics.
Q: How does this process improve purification compared to conventional methods?
A: Conventional methods often produce by-products with similar Rf values and high boiling points, making vacuum distillation difficult. This novel approach minimizes side reactions like beta-elimination and self-coupling, resulting in a cleaner crude product that can be easily purified by standard flash column chromatography, drastically simplifying downstream processing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Alkylated Heterocycles Supplier
The technological advancements described in CN109790079B represent a significant opportunity for the pharmaceutical and fine chemical industries to access high-quality alkylated heterocycles more efficiently. At NINGBO INNO PHARMCHEM, we possess the technical expertise and infrastructure to leverage this cutting-edge chemistry for your specific project needs. Our team has extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that we can support your requirements from early-stage development through to full-scale manufacturing. We adhere to stringent purity specifications and operate rigorous QC labs to guarantee that every batch meets the highest industry standards.
We invite you to collaborate with us to optimize your supply chain and reduce costs through the adoption of this superior synthetic route. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your target molecules. We are ready to provide specific COA data and route feasibility assessments to demonstrate how our capabilities can accelerate your project timelines and enhance your competitive advantage in the market.
