Advanced Copper-Catalyzed Synthesis of Diarylanilines for Scalable Pharmaceutical Manufacturing
Introduction to Novel Diarylaniline Synthesis Technology
The pharmaceutical and agrochemical industries continuously demand efficient, scalable, and environmentally benign synthetic routes for constructing nitrogen-containing heterocycles and aromatic amines. Addressing this critical need, the technical disclosure found in patent CN103342651A introduces a groundbreaking methodology for the synthesis of diarylaniline compounds. This innovation represents a significant departure from traditional cross-coupling strategies by leveraging a copper-catalyzed system that directly couples nitrobenzene derivatives with arylboronic acids. The strategic advantage of this approach lies in its ability to bypass the necessity for pre-functionalized halogenated substrates, which are often associated with higher costs and greater environmental toxicity. By utilizing readily available nitro compounds and boronic acids, this process not only streamlines the synthetic pathway but also enhances the overall atom economy of the reaction. For R&D directors and process chemists, this technology offers a compelling alternative for generating high-purity intermediates essential for drug discovery and development pipelines.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of C-N bonds in aromatic systems has relied heavily on the Buchwald-Hartwig cross-coupling reaction. While effective, this conventional methodology presents substantial logistical and economic challenges for large-scale manufacturing. The primary bottleneck involves the reliance on expensive palladium catalysts and sophisticated, air-sensitive phosphine ligands, which drastically inflate the cost of goods sold (COGS). Furthermore, the requirement for aryl halides as starting materials introduces significant environmental liabilities due to the generation of halogenated waste streams that require complex remediation. Alternative copper-catalyzed methods, such as the classic Chan-Lam coupling, have attempted to mitigate some of these issues by using arylboronic acids; however, early iterations often suffered from moderate yields, narrow substrate scopes, and the necessity for stoichiometric amounts of copper or harsh oxidants. These limitations collectively restrict the commercial viability of existing technologies for the production of complex diarylaniline scaffolds.
The Novel Approach
In stark contrast to these legacy methods, the novel approach detailed in the patent data utilizes a highly optimized copper acetylacetonate (Cu(acac)2) catalytic system to drive the coupling of nitrobenzenes and arylboronic acids. This methodology is transformative because it expands the substrate scope to include nitro compounds, which are typically cheaper and more diverse than their halogenated counterparts. The reaction proceeds efficiently in the presence of a base, specifically alkali metal alkoxides like potassium tert-butoxide, within organic solvents such as 1,4-dioxane. This specific combination of reagents facilitates a smooth transformation under relatively mild thermal conditions, typically ranging from 80°C to 140°C. The result is a robust process capable of delivering target diarylanilines with exceptional yields and purity profiles, effectively overcoming the yield ceilings and substrate limitations observed in prior art techniques.

Mechanistic Insights into Cu(acac)2-Catalyzed C-N Coupling
The mechanistic underpinning of this synthesis relies on the unique redox properties of the copper acetylacetonate complex in conjunction with the strong base. Unlike simple copper salts which may exhibit poor solubility or inconsistent catalytic activity, Cu(acac)2 provides a stable coordination environment that facilitates the activation of the boronic acid species. The reaction likely proceeds through a transmetallation step where the aryl group from the boronic acid is transferred to the copper center, followed by the reduction of the nitro group and subsequent C-N bond formation. The choice of potassium tert-butoxide is critical, as experimental data confirms that other bases, including lithium or sodium tert-butoxide, fail to promote the reaction effectively, suggesting a specific cation effect or solubility requirement for the active catalytic species. This precise tuning of the catalytic cycle ensures that side reactions, such as homocoupling of the boronic acid or over-reduction of the nitro group, are minimized, thereby preserving the integrity of the final product.
From an impurity control perspective, this mechanism offers distinct advantages for pharmaceutical manufacturing. The high selectivity of the Cu(acac)2 system means that the crude reaction mixture contains fewer byproducts compared to palladium-catalyzed routes, simplifying downstream purification. The patent data highlights that products can be isolated with purity levels exceeding 98% via standard silica gel chromatography, indicating a clean reaction profile. This is particularly vital for regulatory compliance, where the identification and quantification of genotoxic impurities or heavy metal residues are stringent. By avoiding palladium entirely, the process eliminates the need for costly and time-consuming metal scavenging steps, further enhancing the operational efficiency. The robustness of the mechanism across various substituted nitrobenzenes and boronic acids demonstrates its versatility for synthesizing diverse libraries of bioactive molecules.
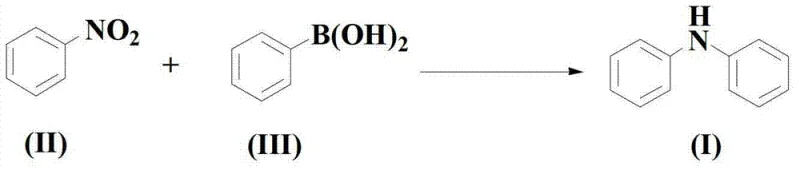
How to Synthesize Diarylaniline Compounds Efficiently
Implementing this synthesis route in a laboratory or pilot plant setting requires strict adherence to the optimized parameters identified in the patent to ensure reproducibility and safety. The process is designed to be operationally simple, involving the mixing of reagents in a specific order under an inert atmosphere to prevent oxidation of sensitive intermediates. The use of 1,4-dioxane as the solvent is preferred due to its ability to dissolve both organic substrates and the inorganic base while maintaining thermal stability at the required reaction temperatures. Operators must monitor the reaction progress, typically via TLC or HPLC, to determine the optimal endpoint, which generally falls between 10 to 14 hours depending on the specific steric and electronic nature of the substrates. Detailed standardized operating procedures for this synthesis are outlined below to guide technical teams in achieving consistent results.
- Mix nitrobenzene derivative, arylboronic acid, Cu(acac)2 catalyst, and potassium tert-butoxide base in 1,4-dioxane solvent.
- Heat the reaction mixture to 80-140°C under nitrogen protection for 10-14 hours with stirring.
- Filter the mixture, remove solvent via rotary evaporation, and purify the residue using silica gel column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the adoption of this copper-catalyzed technology presents a compelling value proposition centered on cost optimization and supply security. The shift away from precious metal catalysts like palladium to abundant copper salts fundamentally alters the cost structure of the manufacturing process. Copper acetylacetonate is significantly less expensive than palladium complexes, and the elimination of proprietary ligands removes a major cost driver often subject to volatile pricing and supply constraints. Furthermore, the ability to use nitrobenzenes instead of aryl halides allows sourcing teams to tap into a broader, more competitive global market for raw materials. This diversification of the supply base reduces the risk of production stoppages due to single-source dependencies, ensuring a more resilient supply chain for critical pharmaceutical intermediates.
- Cost Reduction in Manufacturing: The economic benefits of this process are driven by the substitution of high-cost inputs with commodity chemicals. By eliminating the need for expensive palladium catalysts and specialized ligands, the direct material costs are substantially lowered. Additionally, the simplified workup procedure, which avoids complex metal removal steps, reduces the consumption of auxiliary materials and processing time. The high yields reported in the patent data mean that less raw material is wasted per unit of product, maximizing the return on investment for every batch produced. These factors combine to create a leaner, more cost-effective manufacturing model that improves overall margin potential.
- Enhanced Supply Chain Reliability: The reliance on widely available starting materials such as nitrobenzenes and arylboronic acids ensures a stable and continuous supply of feedstock. Unlike specialized halogenated intermediates which may have limited suppliers, these commodities are produced at scale by multiple chemical manufacturers globally. This abundance mitigates the risk of shortages and allows for more flexible procurement strategies, including just-in-time inventory management. The robustness of the reaction conditions also means that the process is less sensitive to minor variations in raw material quality, further stabilizing the supply chain against upstream fluctuations.
- Scalability and Environmental Compliance: From an environmental, health, and safety (EHS) perspective, this method offers significant advantages that facilitate easier regulatory approval and scale-up. The avoidance of halogenated waste streams reduces the burden on waste treatment facilities and lowers disposal costs. The use of copper, a less toxic metal compared to palladium or nickel, simplifies effluent management and aligns with green chemistry principles. The process operates at moderate temperatures and pressures, making it amenable to scaling in standard stainless steel reactors without requiring exotic metallurgy. This ease of scale-up accelerates the transition from clinical supply to commercial production, reducing time-to-market for new drug candidates.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These answers are derived directly from the experimental data and technical specifications provided in the patent documentation, ensuring accuracy and relevance for decision-makers evaluating this process for their own manufacturing portfolios. Understanding these nuances is crucial for assessing the feasibility of integrating this method into existing production lines or new product development projects.
Q: What is the primary catalyst used in this synthesis method?
A: The method utilizes copper acetylacetonate (Cu(acac)2) as the specific catalyst, which demonstrates superior catalytic specificity compared to other copper salts like CuSO4 or CuCO3.
Q: How does this method compare to traditional Buchwald-Hartwig coupling?
A: Unlike Buchwald-Hartwig coupling which often requires expensive palladium catalysts and complex phosphine ligands, this method uses inexpensive copper catalysts and directly utilizes nitrobenzenes, significantly reducing raw material costs and environmental pollution from halogenated hydrocarbons.
Q: What yields and purity levels can be expected?
A: Experimental data indicates high efficiency, with specific examples achieving yields up to 98.2% and HPLC purity exceeding 98.9%, demonstrating robust process control suitable for industrial applications.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Diarylaniline Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of advanced catalytic technologies like the Cu(acac)2-mediated coupling described in patent CN103342651A. As a leading CDMO partner, we possess the technical expertise and infrastructure to translate such innovative laboratory methods into robust, commercial-scale manufacturing processes. Our team has extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the high purity and yield demonstrated in the lab are maintained at an industrial level. We operate stringent purity specifications and utilize rigorous QC labs to guarantee that every batch of diarylaniline intermediate meets the exacting standards required by the global pharmaceutical industry.
We invite you to collaborate with us to leverage this cost-effective and environmentally friendly synthesis route for your next project. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and target specifications. Please contact us today to request specific COA data for our existing catalog or to discuss route feasibility assessments for your custom synthesis needs. Let us help you optimize your supply chain and accelerate your drug development timeline with our reliable diarylaniline supply solutions.
