Advanced Synthesis of Barosavir Intermediate: Enhancing Safety and Scalability for Global Pharma Supply Chains
Advanced Synthesis of Barosavir Intermediate: Enhancing Safety and Scalability for Global Pharma Supply Chains
The pharmaceutical industry continuously seeks robust manufacturing pathways for critical antiviral agents, particularly as the demand for influenza treatments like Baloxavir marboxil grows. Patent CN112851508A introduces a transformative preparation method for a key Barosavir intermediate, specifically 3,4-difluoro-2-methylbenzoic acid, addressing long-standing safety and scalability challenges in its production. This innovation shifts the paradigm from hazardous organometallic chemistry to a safer, palladium-catalyzed carbonylation strategy, offering a compelling value proposition for reliable pharmaceutical intermediate suppliers aiming to secure their supply chains. By leveraging carbon monoxide insertion under pressurized conditions, the disclosed method achieves high yields while operating under significantly milder thermal conditions compared to legacy processes. This technical breakthrough not only enhances process safety but also aligns with modern green chemistry principles, making it an attractive option for cost reduction in API manufacturing where operational efficiency and hazard mitigation are paramount.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 3,4-difluoro-2-methylbenzoic acid has relied heavily on aggressive organometallic transformations that pose severe risks for industrial application. As illustrated in the prior art, one common route involves the formation of a Grignard reagent followed by carbon dioxide quenching, a process depicted in the reaction scheme below. This approach necessitates the use of highly active magnesium reagents which are sensitive to moisture and oxygen, requiring stringent anhydrous conditions that complicate reactor operations and increase utility costs.
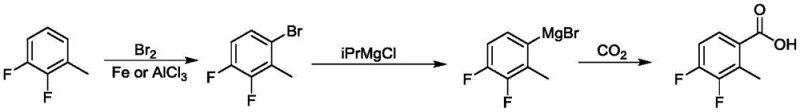
Furthermore, alternative legacy methods utilize n-butyllithium at cryogenic temperatures of -78°C to effect lithium-halogen exchange before carboxylation. Such extreme low-temperature requirements demand specialized refrigeration equipment and substantial energy consumption, creating a bottleneck for commercial scale-up of complex pharmaceutical intermediates. The inherent instability of these organolithium species increases the risk of thermal runaway incidents, while the generation of stoichiometric metal salt waste complicates downstream waste treatment and environmental compliance. These factors collectively render traditional routes economically inefficient and operationally hazardous for large-scale production facilities.
The Novel Approach
In stark contrast, the novel methodology disclosed in the patent utilizes a palladium-catalyzed carbonylation of 2-bromo-3,4-difluorotoluene, offering a streamlined and safer alternative. The general reaction scheme demonstrates the conversion of the aryl bromide directly into the corresponding ester using carbon monoxide and an alcohol solvent, mediated by a palladium catalyst and a base. This transition metal-catalyzed approach operates at moderate temperatures ranging from 65°C to 105°C, completely eliminating the need for cryogenic cooling or pyrophoric reagents.
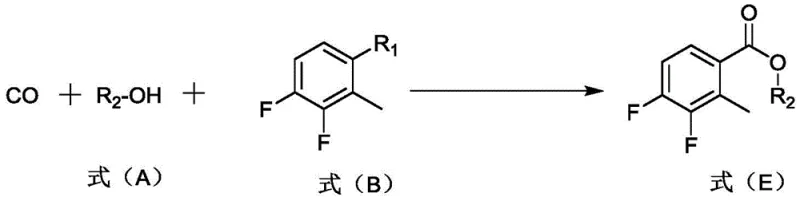
The versatility of this new route is further enhanced by the ability to use various alcohols, such as methanol or ethanol, as both solvents and reactants, simplifying the material inventory required for production. By avoiding the generation of large quantities of inorganic salts typical of Grignard or lithiation workups, the process significantly reduces the environmental footprint and simplifies the isolation of the crude product. This methodological shift represents a strategic advancement for high-purity pharmaceutical intermediates, enabling manufacturers to achieve consistent quality with reduced operational complexity and improved safety profiles for plant personnel.
Mechanistic Insights into Palladium-Catalyzed Carbonylation
The core of this innovative synthesis lies in the efficient catalytic cycle driven by palladium complexes, such as [1,1'-bis(diphenylphosphino)ferrocene]dichloropalladium (Pd(dppf)Cl2). The mechanism initiates with the oxidative addition of the aryl bromide substrate to the active Pd(0) species, forming an aryl-palladium(II) intermediate. This step is crucial as it activates the carbon-bromine bond for subsequent insertion reactions. Following oxidative addition, carbon monoxide coordinates to the metal center and inserts into the palladium-carbon bond, generating an acyl-palladium complex. This carbonyl insertion is the defining step that builds the carboxylic acid framework directly onto the aromatic ring.
Subsequently, the alcohol solvent (e.g., methanol), activated by the presence of a base like triethylamine or DIPEA, attacks the acyl-palladium species. This nucleophilic attack facilitates the reductive elimination step, releasing the desired ester product and regenerating the active Pd(0) catalyst to continue the cycle. The choice of ligand, such as dppf, plays a pivotal role in stabilizing the palladium center and promoting the reductive elimination step, thereby ensuring high turnover numbers and minimizing catalyst loading. This mechanistic elegance allows the reaction to proceed with high atom economy, as the carbon monoxide serves as the sole source of the carbonyl carbon, avoiding the stoichiometric waste associated with carboxylation using dry ice or similar reagents.
Impurity control in this system is inherently superior due to the specificity of the catalytic cycle. Unlike organometallic routes where side reactions like Wurtz coupling or protonation of the organometallic species can lead to significant byproduct formation, the palladium-catalyzed pathway is highly selective for the carbonylation product. The mild reaction conditions prevent thermal degradation of the sensitive difluoro-substituted aromatic ring, preserving the structural integrity of the molecule. Furthermore, the use of pressurized carbon monoxide ensures that the concentration of CO at the catalytic site remains high, suppressing competing pathways such as direct reduction of the aryl halide. This results in a cleaner reaction profile, simplifying the purification process and ensuring that the final high-purity pharmaceutical intermediates meet stringent regulatory specifications for residual metals and organic impurities.
How to Synthesize 3,4-Difluoro-2-methylbenzoic Acid Efficiently
The practical implementation of this synthesis involves a two-stage sequence: first, the carbonylation to form the ester intermediate, and second, the hydrolysis to yield the free acid. The patent provides detailed experimental protocols demonstrating that the carbonylation can be effectively conducted in a standard autoclave at 1.0 MPa pressure and 85°C, using methanol as the solvent and triethylamine as the acid scavenger. Following the carbonylation, the crude ester is isolated and subjected to hydrolysis using aqueous sodium hydroxide in a mixed solvent system. This second step proceeds smoothly at room temperature, highlighting the mildness of the overall process. For the complete standardized operating procedure and specific parameter optimization, please refer to the detailed guide below.
- Perform palladium-catalyzed carbonylation of 2-bromo-3,4-difluorotoluene with carbon monoxide in methanol using a base like triethylamine at 85°C and 1.0 MPa pressure.
- Isolate the methyl ester intermediate (Compound G) via extraction and distillation, achieving yields around 88%.
- Hydrolyze the ester using sodium hydroxide in a methanol/water mixture at room temperature, followed by acidification to precipitate the final acid product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this patented synthesis route offers tangible strategic benefits that extend beyond mere chemical curiosity. The primary advantage lies in the drastic simplification of the manufacturing infrastructure required. By eliminating the need for cryogenic reactors capable of maintaining -78°C, facilities can utilize standard glass-lined or stainless steel reactors equipped with heating capabilities, which are far more common and less capital-intensive. This reduction in specialized equipment requirements translates directly into lower capital expenditure (CAPEX) for new production lines and reduced maintenance costs for existing ones, facilitating a more agile response to market demand fluctuations.
- Cost Reduction in Manufacturing: The economic implications of switching from organolithium or Grignard reagents to a catalytic carbonylation process are profound. Organometallic reagents are not only expensive to purchase but also require costly disposal protocols due to their hazardous nature. In contrast, carbon monoxide is a commodity chemical, and the palladium catalyst, while precious, is used in minute catalytic quantities and can often be recovered. The elimination of stoichiometric metal waste reduces the burden on wastewater treatment plants, lowering operational expenditure (OPEX) related to environmental compliance. Additionally, the higher yields reported in the patent examples, often exceeding 85%, mean less raw material is wasted per unit of product, further driving down the cost of goods sold (COGS).
- Enhanced Supply Chain Reliability: Supply chain resilience is critically dependent on the availability and stability of raw materials. Traditional routes relying on n-butyllithium are vulnerable to supply disruptions and require cold-chain logistics for storage and transport. The new method utilizes stable, shelf-stable starting materials like 2-bromo-3,4-difluorotoluene and common solvents like methanol, which are readily available from multiple global suppliers. This diversification of the supply base reduces the risk of production stoppages due to single-source dependency. Furthermore, the improved safety profile reduces the likelihood of regulatory shutdowns or insurance premium hikes associated with handling pyrophoric materials, ensuring a more consistent and reliable supply of the intermediate to downstream API manufacturers.
- Scalability and Environmental Compliance: Scaling chemical processes often amplifies safety risks, particularly with exothermic organometallic reactions. The palladium-catalyzed carbonylation described here is inherently safer to scale because the reaction rate is controlled by gas-liquid mass transfer and catalyst activity rather than the rapid, uncontrollable kinetics of organolithium additions. The mild conditions allow for larger batch sizes without the fear of thermal runaway, enabling commercial scale-up of complex pharmaceutical intermediates to multi-ton quantities with confidence. From an environmental perspective, the process generates significantly less hazardous waste, aligning with increasingly strict global regulations on solvent use and heavy metal discharge. This 'greener' profile not only simplifies permitting but also enhances the corporate sustainability metrics of the manufacturing entity, a key factor for modern pharmaceutical partnerships.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These insights are derived directly from the experimental data and comparative analysis presented in the patent documentation, providing clarity on how this method outperforms legacy techniques in terms of safety, efficiency, and product quality. Understanding these nuances is essential for technical teams evaluating the feasibility of integrating this route into their current manufacturing portfolios.
Q: Why is the new carbonylation route safer than traditional Grignard methods?
A: Traditional methods rely on highly reactive organometallic reagents like n-butyllithium or Grignard reagents which require cryogenic conditions (-78°C) and pose significant fire and explosion risks. The new palladium-catalyzed route operates at mild temperatures (65-105°C) and avoids pyrophoric reagents, drastically improving operational safety.
Q: What are the scalability advantages of this synthesis method?
A: The process eliminates the need for complex low-temperature equipment and hazardous reagent handling. By using standard autoclaves for carbonylation and ambient temperature for hydrolysis, the method facilitates easier industrial scale-up from kilogram to multi-ton production without specialized cryogenic infrastructure.
Q: How does this method impact the purity profile of the intermediate?
A: The catalytic route minimizes side reactions associated with aggressive organometallic species. Patent data indicates high purity levels (e.g., 97.1% for the ester and 98.5% for the final acid) can be achieved through straightforward workup procedures, reducing the burden on downstream purification.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3,4-Difluoro-2-methylbenzoic Acid Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition to advanced synthetic routes requires a partner with deep technical expertise and robust manufacturing capabilities. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of patents like CN112851508A are fully realized in practical, large-scale operations. We maintain stringent purity specifications and operate rigorous QC labs equipped with state-of-the-art analytical instrumentation to guarantee that every batch of 3,4-difluoro-2-methylbenzoic acid meets the exacting standards required for antiviral drug synthesis. Our commitment to quality assurance ensures that our clients receive intermediates that facilitate smooth downstream processing and final API approval.
We invite global pharmaceutical partners to collaborate with us to leverage this superior synthesis technology for their Barosavir supply chains. By engaging with our technical procurement team, you can request a Customized Cost-Saving Analysis that quantifies the potential economic benefits of switching to this catalytic route for your specific volume requirements. We encourage you to contact us today to obtain specific COA data and route feasibility assessments, allowing you to make informed decisions that enhance both the profitability and resilience of your antiviral medication production.
