Scalable Production of High-Purity Isoquinoline Intermediates for Rho Kinase Inhibitors
The pharmaceutical industry's demand for high-purity Rho kinase inhibitors, such as Fasudil and Ripasudil, continues to surge as these compounds prove critical in treating cerebral vasospasm and glaucoma. At the heart of synthesizing these life-saving medications lies the efficient production of isoquinoline sulfonyl chloride intermediates. Recent advancements documented in patent CN111909088B introduce a groundbreaking methodology that utilizes a BTC/Ph3PO chlorination system, marking a significant departure from traditional, hazardous synthetic routes. This innovation addresses long-standing challenges regarding impurity control and environmental impact, offering a robust pathway for the reliable pharmaceutical intermediate supplier seeking to optimize their manufacturing capabilities. By leveraging this novel approach, manufacturers can achieve exceptional product quality while adhering to increasingly stringent global regulatory standards for green chemistry.
Historically, the synthesis of isoquinoline-5-sulfonyl chloride has relied heavily on thionyl chloride (SOCl2) as both the solvent and chlorinating agent. While effective in small-scale laboratory settings, this conventional method presents severe limitations when translated to commercial scale-up of complex pharmaceutical intermediates. The process typically requires refluxing for extended periods, often between 4 to 8 hours, followed by energy-intensive distillation to remove excess thionyl chloride. Furthermore, the highly acidic environment generated during the reaction frequently leads to unwanted side reactions, such as the chlorination of the isoquinoline ring at unintended positions. These byproducts complicate downstream purification, increase waste treatment costs, and pose significant corrosion risks to stainless steel reactor equipment, thereby threatening supply chain continuity.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
The reliance on thionyl chloride creates a cascade of operational inefficiencies that procurement managers and supply chain heads must carefully evaluate. The necessity to distill off large volumes of unreacted thionyl chloride not only consumes substantial energy but also releases sulfur-containing byproducts that require expensive scrubbing systems to meet environmental compliance regulations. Additionally, the liquid nature of thionyl chloride at ambient temperatures introduces inherent safety hazards during storage and transportation, complicating logistics for a reliable agrochemical intermediate supplier or pharma partner. The aggressive acidity often degrades reactor linings over time, leading to unplanned maintenance downtime and potential contamination of the product with metal ions, which is unacceptable for high-purity API manufacturing.
The Novel Approach
In stark contrast, the method disclosed in CN111909088B employs bis(trichloromethyl) carbonate (BTC) activated by a catalytic amount of triphenylphosphine oxide (Ph3PO). This system operates under significantly milder conditions, typically between 25°C and 110°C, and eliminates the need for excessive chlorinating agents. The reaction proceeds smoothly in common organic solvents like ethyl acetate or dichloromethane, yielding a white solid product that can be isolated simply by filtration. This drastic simplification of the work-up procedure removes the need for vacuum distillation, thereby reducing energy consumption and minimizing thermal stress on the product. The result is a streamlined process that enhances cost reduction in pharmaceutical intermediate manufacturing while delivering superior chemical integrity.

Mechanistic Insights into BTC/Ph3PO-Catalyzed Chlorination
To fully appreciate the technical superiority of this route, one must understand the catalytic cycle that drives the transformation. Unlike stoichiometric methods where reagents are consumed in bulk, this system utilizes Ph3PO to activate the BTC, generating a highly reactive chlorophosphonium species in situ. This active species then efficiently transfers chlorine to the sulfonic acid group, regenerating the Ph3PO catalyst upon completion of the chlorination step. The elegance of this mechanism lies in its kinetic consistency; the rate of formation of the active chlorinating agent matches the dissolution rate of the substrate, preventing the accumulation of aggressive reagents that could attack the sensitive isoquinoline ring. This precise control is what allows the process to achieve yields stabilizing around 95% with HPLC purity exceeding 99.90%.
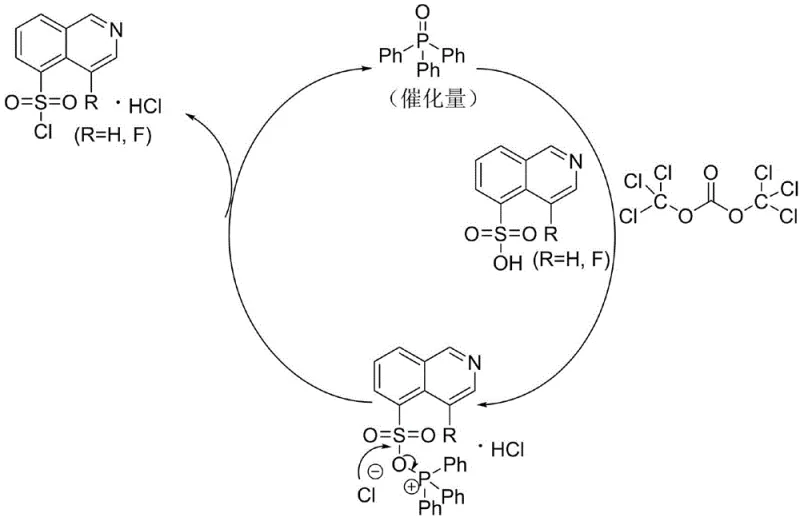
Furthermore, the impurity control mechanism is inherently built into the reaction thermodynamics. Traditional methods often struggle with polychlorinated byproducts due to the harsh, acidic conditions promoted by SOCl2. However, the BTC/Ph3PO system maintains a neutral to mildly acidic environment throughout the reaction course. This selectivity ensures that the chlorine atoms are directed exclusively towards the sulfonyl group, leaving the aromatic core intact. For R&D directors focused on impurity profiles, this means a drastically simplified purification workflow. The absence of ring-chlorinated impurities reduces the burden on chromatographic separation steps, allowing for a more direct path to the final active pharmaceutical ingredient with minimal material loss.
How to Synthesize Isoquinoline-5-sulfonyl Chloride Efficiently
Implementing this synthesis route requires careful attention to reagent ratios and temperature control to maximize the benefits of the catalytic system. The process begins by charging isoquinoline-5-sulfonic acid, BTC, and a catalytic quantity of Ph3PO into a reactor containing a suitable organic solvent. The mixture is then heated to a specific range, preferably between 40°C and 90°C, and maintained for a duration of 1.0 to 5.0 hours to ensure complete conversion. Following the reaction, the product precipitates as a high-purity solid upon cooling, which is collected via filtration. The detailed standardized synthesis steps see the guide below for exact parameters.
- Charge isoquinoline-5-sulfonic acid, bis(trichloromethyl) carbonate (BTC), and catalytic triphenylphosphine oxide (Ph3PO) into a reactor with organic solvent A.
- Heat the mixture to 25-110°C and maintain reaction for 1.0 to 14.0 hours to ensure complete chlorination.
- Cool to room temperature, filter the white solid product, and recover Ph3PO from the mother liquor for reuse.
Commercial Advantages for Procurement and Supply Chain Teams
For decision-makers responsible for sourcing and logistics, the transition to this BTC-based methodology offers profound strategic advantages beyond mere chemical efficiency. The elimination of thionyl chloride removes a major bottleneck related to hazardous material handling and storage, thereby reducing insurance premiums and safety compliance overheads. Moreover, the ability to recover and reuse the Ph3PO catalyst from the mother liquor significantly lowers the raw material cost per kilogram of produced intermediate. This process intensification translates directly into a more resilient supply chain capable of withstanding market fluctuations in reagent pricing.
- Cost Reduction in Manufacturing: The primary driver for cost optimization in this protocol is the catalytic nature of the Ph3PO additive. Unlike traditional methods that consume stoichiometric amounts of activators like DMF or excess thionyl chloride, this system recycles the phosphine oxide, drastically cutting down on consumable expenses. Additionally, the simplified isolation procedure, which relies on filtration rather than energy-intensive distillation, reduces utility costs associated with steam and vacuum generation. The overall atom economy is substantially improved, meaning less raw material is wasted as byproduct, leading to significant cost savings over the lifecycle of the product.
- Enhanced Supply Chain Reliability: Sourcing thionyl chloride can be subject to regulatory restrictions and logistical challenges due to its classification as a hazardous corrosive liquid. By switching to BTC, a solid reagent that is easier to store and transport, manufacturers mitigate the risk of supply disruptions. The robustness of the reaction conditions also means that the process is less sensitive to minor variations in raw material quality, ensuring consistent batch-to-batch output. This reliability is crucial for maintaining the continuous production schedules required by global pharmaceutical clients who depend on just-in-time delivery models.
- Scalability and Environmental Compliance: Scaling chemical processes often amplifies waste management issues, but this green chemistry approach minimizes the generation of 'three wastes' (waste water, gas, and solid). The absence of sulfur dioxide emissions and acidic wastewater simplifies the effluent treatment process, making it easier to comply with strict environmental regulations in major manufacturing hubs. The mild reaction temperatures also reduce the thermal load on cooling systems, allowing existing infrastructure to handle larger batch sizes without costly capital upgrades, thus facilitating seamless commercial scale-up of complex pharmaceutical intermediates.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this advanced chlorination technology. These insights are derived directly from the experimental data and beneficial effects reported in the patent literature, providing a clear picture of what partners can expect when adopting this route. Understanding these nuances is essential for evaluating the feasibility of integrating this method into existing production lines.
Q: How does the BTC/Ph3PO system improve impurity profiles compared to thionyl chloride?
A: The BTC/Ph3PO system operates under milder conditions and avoids the excessive acidity associated with thionyl chloride, significantly reducing ring chlorination byproducts and improving the HPLC purity of the final Rho kinase inhibitor to over 99.90%.
Q: Is the triphenylphosphine oxide catalyst recoverable in this process?
A: Yes, the process is designed for high atom economy. The Ph3PO catalyst precipitates from the concentrated mother liquor at low temperatures and can be washed and reused repeatedly without significant loss of activity.
Q: What are the safety advantages of using BTC over traditional chlorinating agents?
A: BTC is a solid at room temperature, which eliminates the storage and transportation hazards associated with liquid thionyl chloride. It also generates fewer corrosive acidic byproducts, enhancing workshop safety and equipment longevity.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Isoquinoline-5-sulfonyl Chloride Supplier
At NINGBO INNO PHARMCHEM, we recognize that the theoretical advantages of a patent must be translated into tangible production capacity to create real value. As a dedicated CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the high purity specifications demonstrated in the lab are maintained at an industrial level. Our rigorous QC labs are equipped to monitor every critical parameter of the BTC/Ph3PO reaction, guaranteeing that every batch of isoquinoline intermediate meets the stringent purity requirements necessary for downstream Rho kinase inhibitor synthesis.
We invite forward-thinking procurement leaders to collaborate with us on optimizing their supply chains for these critical intermediates. By engaging with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We encourage you to reach out today to obtain specific COA data and route feasibility assessments that will demonstrate how our manufacturing expertise can enhance your project's timeline and budget efficiency.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
