Advanced Manufacturing of p-Toluenesulfonylmethyl Isonitrile for High-Value Heterocycle Synthesis
The pharmaceutical and agrochemical industries rely heavily on versatile building blocks capable of constructing complex nitrogen-containing heterocycles, and p-toluenesulfonylmethyl isonitrile (TosMIC) stands out as a critical reagent in this domain. As detailed in the recent patent CN115819298A, a significant technological breakthrough has been achieved in the preparation of this valuable intermediate, addressing long-standing challenges related to waste generation and process complexity. This new methodology offers a robust pathway for producing high-purity TosMIC, which serves as a pivotal precursor for synthesizing imidazoles, thiazoles, oxazoles, and triazoles found in numerous active pharmaceutical ingredients. By leveraging a novel dehydration strategy using triphosgene, the process circumvents the environmental hazards associated with traditional phosphorus-based reagents, marking a substantial advancement in green chemistry for fine chemical manufacturing.
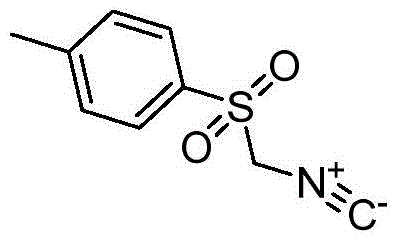
For R&D directors evaluating synthetic routes, the structural integrity and purity of the starting material are paramount, and the molecular architecture of p-toluenesulfonylmethyl isonitrile allows for unique reactivity patterns due to the presence of both electrophilic and nucleophilic centers alongside a good leaving group. The innovation described in the patent data provides a clear roadmap for scaling this chemistry without compromising on quality, ensuring that the resulting intermediates meet the stringent specifications required for downstream drug synthesis. This report analyzes the technical merits of this approach, highlighting how it aligns with modern demands for sustainable and cost-effective production of reliable pharmaceutical intermediate supplier materials.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
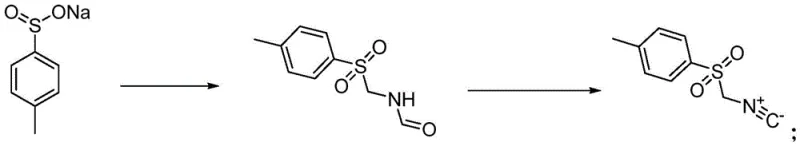
Historically, the synthesis of p-toluenesulfonylmethyl isonitrile has been plagued by significant environmental and operational inefficiencies, primarily stemming from the reliance on phosphorus oxychloride (POCl3) as the dehydrating agent. As illustrated in the prior art referenced by patent CN104387301, the conventional route involves reacting sodium p-toluenesulfinate with formamide and paraformaldehyde, followed by a dehydration step using POCl3 under alkaline conditions. This legacy method presents severe drawbacks, including the generation of large volumes of phosphorus-containing wastewater that is notoriously difficult and expensive to treat, thereby increasing the overall environmental footprint of the manufacturing process. Furthermore, the isolation of the intermediate formamide often requires low-temperature water crystallization, which leads to product loss due to solubility issues and necessitates high energy consumption for drying, ultimately eroding profit margins and process sustainability.
The Novel Approach
In stark contrast to these outdated techniques, the method disclosed in CN115819298A introduces a streamlined two-step protocol that replaces hazardous phosphorus reagents with solid triphosgene, fundamentally altering the waste profile of the reaction. By utilizing triphosgene in conjunction with an organic base such as triethylamine, the new process achieves efficient dehydration of the formamide intermediate while completely avoiding the formation of inorganic phosphorus waste streams. This shift not only simplifies the post-reaction workup by eliminating the need for complex phosphorus removal steps but also enhances the overall safety profile of the operation by using a stable solid reagent instead of corrosive liquids or toxic gases. The result is a cleaner, more atom-economical process that delivers the target isonitrile with exceptional purity, directly addressing the pain points of cost reduction in pharmaceutical intermediate manufacturing identified by procurement teams.
Mechanistic Insights into Triphosgene-Mediated Dehydration
The core of this technological advancement lies in the precise control of reaction conditions during the dehydration phase, where the formamide intermediate is converted into the isonitrile functionality through a mechanism facilitated by triphosgene and a tertiary amine base. In the first stage, sodium p-toluenesulfinate undergoes an acid-catalyzed condensation with paraformaldehyde and formamide, typically employing formic acid to maintain the necessary acidic environment at temperatures ranging from 60 to 100°C, with 80°C being optimal for maximizing conversion rates. This step generates the N-(p-toluenesulfonylmethyl)formamide intermediate with high fidelity, setting the stage for the subsequent transformation where the formyl group is removed to reveal the reactive isonitrile group. The use of formic acid as both a catalyst and a reactant component ensures a homogeneous reaction medium that promotes efficient mass transfer and minimizes side reactions.
Following the formation of the intermediate, the system is subjected to basic conditions using triethylamine, which activates the triphosgene to generate the reactive chloroformate species in situ. This species reacts with the formamide nitrogen, facilitating the elimination of water and carbon dioxide to yield the final isonitrile product. The strict temperature control between -5 and 5°C during this exothermic step is crucial for suppressing decomposition pathways and ensuring the stability of the sensitive isonitrile group. From an impurity control perspective, this mechanism is superior because it avoids the introduction of phosphorus atoms into the reaction matrix, thereby eliminating the risk of phosphorus-containing impurities that are difficult to purge and can interfere with downstream catalytic processes in API synthesis, ensuring a cleaner impurity profile for the final active ingredient.
How to Synthesize p-Toluenesulfonylmethyl Isonitrile Efficiently
Implementing this optimized synthesis route requires careful attention to stoichiometry and thermal management to replicate the high yields reported in the patent examples. The process begins with the condensation of sodium p-toluenesulfinate, formamide, and paraformaldehyde in the presence of formic acid, followed by a controlled dehydration using triphosgene and triethylamine in a dichloromethane solvent system. Detailed operational parameters, including specific molar ratios and temperature ramps, are critical for achieving the reported total yields of nearly 95%, and operators must adhere to strict safety protocols when handling triphosgene despite its solid state. For a comprehensive guide on executing this procedure with precision, please refer to the standardized synthesis steps outlined below.
- React sodium p-toluenesulfinate with formamide and paraformaldehyde in the presence of formic acid at 80°C to form the formamide intermediate.
- Treat the intermediate with triphosgene and triethylamine in dichloromethane at -5 to 5°C to effect dehydration.
- Perform aqueous workup, concentration, and crystallization using n-heptane to isolate the final product with high purity.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this triphosgene-based methodology offers profound advantages for supply chain stability and cost management, primarily driven by the simplification of waste treatment and raw material utilization. By eliminating the need for phosphorus oxychloride, manufacturers can drastically reduce the costs associated with hazardous waste disposal and wastewater treatment facilities, which are often significant overheads in fine chemical production. This reduction in environmental compliance burden translates directly into lower operational expenditures, allowing for more competitive pricing structures without sacrificing margin, a key consideration for procurement managers seeking cost reduction in pharmaceutical intermediate manufacturing. Additionally, the use of solid triphosgene simplifies logistics and storage compared to liquid or gaseous alternatives, enhancing supply chain reliability and reducing the risk of shipment delays or regulatory hurdles associated with highly toxic substances.
- Cost Reduction in Manufacturing: The substitution of phosphorus oxychloride with triphosgene removes the necessity for expensive phosphorus waste neutralization processes, leading to substantial savings in utility and treatment costs. Furthermore, the high yield of the reaction minimizes raw material waste, ensuring that every kilogram of input contributes effectively to the final output, which optimizes the overall cost of goods sold. The simplified workup procedure also reduces labor hours and solvent consumption, compounding the financial benefits of this greener synthetic route.
- Enhanced Supply Chain Reliability: Triphosgene is a commercially available solid reagent with a stable supply chain, unlike certain gaseous reagents that may face transportation restrictions or availability fluctuations. The robustness of the reaction conditions, which tolerate standard industrial equipment without requiring specialized corrosion-resistant materials for phosphorus handling, ensures consistent production schedules. This reliability is essential for maintaining continuous supply to downstream API manufacturers, preventing bottlenecks that could disrupt the broader pharmaceutical value chain.
- Scalability and Environmental Compliance: The process is inherently scalable, as demonstrated by the successful execution of the reaction on multi-gram scales in the patent examples, indicating readiness for tonnage production. The absence of phosphorus effluents aligns with increasingly stringent global environmental regulations, future-proofing the manufacturing site against potential regulatory crackdowns. This environmental stewardship not only protects the company's license to operate but also appeals to eco-conscious partners seeking sustainable sourcing options for their chemical supply chains.
Frequently Asked Questions (FAQ)
Understanding the technical nuances of this new synthesis method is vital for stakeholders evaluating its integration into existing production lines. The following questions address common inquiries regarding safety, yield, and environmental impact, drawing directly from the experimental data and comparative analysis provided in the patent documentation. These insights are intended to clarify the operational benefits and assist decision-makers in assessing the feasibility of adopting this technology for commercial scale-up of complex pharmaceutical intermediates.
Q: How does this new method improve environmental compliance compared to traditional routes?
A: The novel process replaces phosphorus oxychloride (POCl3) with triphosgene, eliminating the generation of difficult-to-treat phosphorus-containing wastewater and significantly reducing environmental burden.
Q: What yields can be expected from this optimized synthesis route?
A: Experimental data indicates a total yield approaching 95%, with individual step yields exceeding 97%, demonstrating superior efficiency over prior art methods.
Q: Why is triphosgene preferred over phosgene or diphosgene in this reaction?
A: Triphosgene is a solid at room temperature, offering safer handling and dosing control compared to toxic gaseous phosgene or volatile liquid diphosgene, while maintaining high reaction efficiency.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable p-Toluenesulfonylmethyl Isonitrile Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates like p-toluenesulfonylmethyl isonitrile play in the development of next-generation therapeutics. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory bench to industrial reactor is seamless and efficient. We are committed to delivering products that meet stringent purity specifications through our rigorous QC labs, guaranteeing that every batch supports your R&D and manufacturing goals with consistency and reliability. Our expertise in implementing green chemistry principles allows us to offer this advanced intermediate with a superior environmental profile, aligning with the sustainability targets of modern pharmaceutical companies.
We invite you to collaborate with us to leverage this innovative synthesis technology for your specific project needs. Please contact our technical procurement team to request a Customized Cost-Saving Analysis tailored to your volume requirements. We are ready to provide specific COA data and route feasibility assessments to demonstrate how our optimized manufacturing process can enhance your supply chain efficiency and reduce your overall production costs while maintaining the highest standards of quality and safety.
