Advanced Manufacturing of Beta-Hydroxy Sulfone Derivatives: A Scalable Route for Pharmaceutical Intermediates
The landscape of fine chemical manufacturing is constantly evolving, driven by the need for safer, more efficient, and cost-effective synthetic routes. A pivotal advancement in this domain is documented in Chinese Patent CN1246308C, which discloses a novel preparation process for beta-hydroxy sulfone derivatives. This technology represents a significant departure from conventional methodologies, offering a robust pathway for generating valuable organic intermediates used extensively in pharmaceutical synthesis. As a leading entity in the fine chemicals sector, we recognize the immense potential of this acid-catalyzed approach to redefine supply chain reliability for complex intermediates. The patent outlines a method where sulfonyl chlorides react with olefins in the presence of water or alcohols and a catalytic amount of acid, typically within a tetrahydrofuran (THF) solvent system. This innovation addresses critical pain points in traditional synthesis, such as the handling of hazardous reagents and the complexity of purification, positioning it as a cornerstone technology for modern pharmaceutical intermediate production.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of beta-hydroxy sulfone derivatives has relied heavily on reactions involving carbonyl-containing sulfone derivatives and organometallic reagents. The most prevalent traditional routes utilize Grignard reagents or metal hydrides like sodium borohydride (NaBH4) and lithium aluminum hydride (LiAlH4). These reagents are notoriously sensitive to moisture and air, necessitating stringent anhydrous conditions and inert atmosphere protocols that drastically increase operational costs and safety risks. Furthermore, the requirement for cryogenic temperatures or strictly controlled environments often limits the scalability of these processes, making them less attractive for large-volume commercial scale-up of complex pharmaceutical intermediates. The sensitivity of these reagents also introduces significant variability in batch-to-batch consistency, posing challenges for quality control teams aiming for rigorous purity specifications.
The Novel Approach
In stark contrast, the methodology described in CN1246308C utilizes a direct addition reaction between olefins and sulfonyl chlorides mediated by a simple acid catalyst. This approach eliminates the need for pyrophoric or moisture-sensitive organometallic species, allowing reactions to proceed under much milder conditions, typically between 20°C and 70°C. By leveraging readily available starting materials such as styrenes and various sulfonyl chlorides, this process streamlines the synthetic workflow. The use of common protic sources like water or alcohols as nucleophiles further simplifies the reagent profile. This shift not only enhances safety but also opens the door for significant cost reduction in pharmaceutical intermediate manufacturing by removing the need for specialized containment systems and expensive drying agents associated with Grignard chemistry. The result is a more resilient supply chain capable of delivering high-purity beta-hydroxy sulfone derivatives with greater efficiency.
Mechanistic Insights into Acid-Catalyzed Electrophilic Addition
The core of this technological breakthrough lies in the mechanism of acid-catalyzed electrophilic addition. In this system, the sulfonyl chloride acts as the electrophilic source, activated by the presence of a catalytic acid such as p-toluenesulfonic acid, hydrochloric acid, or sulfuric acid. The acid protonates the reaction medium, facilitating the generation of a reactive sulfonium ion intermediate or enhancing the electrophilicity of the sulfur center. The olefin, acting as a nucleophile, attacks this activated species, leading to the formation of a carbocation intermediate. Subsequently, water or an alcohol molecule attacks the carbocation to finalize the addition, yielding the beta-hydroxy or beta-alkoxy sulfone structure. This mechanism is highly advantageous because it proceeds with high regioselectivity, ensuring that the functional groups are installed at the correct positions on the carbon skeleton without extensive isomerization.
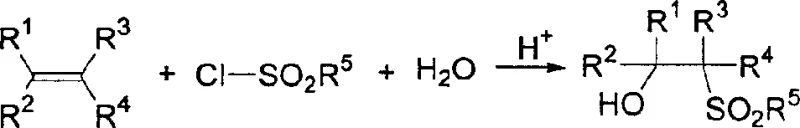
From an impurity control perspective, this mechanism offers distinct benefits over radical-based or metal-catalyzed alternatives. The absence of transition metals eliminates the risk of heavy metal contamination, a critical parameter for API intermediates destined for human consumption. Furthermore, the mild acidic conditions minimize side reactions such as polymerization of the olefin or decomposition of the sulfonyl group, which are common pitfalls in harsher synthetic environments. The patent data indicates that by carefully controlling the molar ratios—specifically maintaining an olefin to sulfonyl chloride ratio of roughly 100:150—and selecting appropriate acids, the formation of byproducts is significantly suppressed. This inherent selectivity reduces the burden on downstream purification units, allowing for simpler workup procedures involving standard extraction and washing steps rather than complex distillation or recrystallization sequences.
How to Synthesize Beta-Hydroxy Sulfone Derivatives Efficiently
Implementing this synthesis route requires precise adherence to the reaction parameters outlined in the patent to ensure optimal yield and purity. The process begins with the dissolution of the sulfonyl chloride in a tetrahydrofuran solvent, followed by the sequential addition of the olefin, water or alcohol, and the acid catalyst. The reaction mixture is then maintained at a controlled temperature ranging from 20°C to 70°C for a duration of 1 to 3 days, depending on the specific substrates involved. Following the reaction period, the product is isolated through a series of standard unit operations including extraction with ether or ethyl acetate, washing with water and brine, and drying over anhydrous sodium sulfate. The detailed standardized synthesis steps see the guide below.
- Dissolve the selected sulfonyl chloride (aliphatic or aromatic) in a tetrahydrofuran (THF) solvent within a reaction vessel.
- Add the olefin substrate (e.g., styrene derivatives), water or alcohol, and a catalytic amount of acid (such as p-toluenesulfonic acid or sulfuric acid) to the mixture.
- Maintain the reaction temperature between 20°C and 70°C for 1 to 3 days, followed by extraction, washing, drying, and column chromatography purification.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this patented process translates into tangible strategic advantages beyond mere chemical efficacy. The primary benefit stems from the drastic simplification of the raw material portfolio. By replacing expensive, hazard-classified organometallic reagents with stable, commodity-grade sulfonyl chlorides and olefins, companies can achieve substantial cost savings in raw material procurement. Additionally, the elimination of strict anhydrous requirements reduces the energy consumption associated with solvent drying and inert gas purging, contributing to a lower overall carbon footprint and operational expenditure. This robustness ensures that production schedules are less susceptible to delays caused by reagent instability or environmental fluctuations.
- Cost Reduction in Manufacturing: The economic impact of switching to this acid-catalyzed route is profound. Traditional methods often incur high costs due to the need for specialized equipment capable of handling pyrophoric materials and the disposal of hazardous metal waste. This new process operates under ambient to moderate pressures and temperatures, utilizing standard glass-lined or stainless steel reactors found in most multipurpose facilities. The removal of transition metal catalysts also negates the need for expensive scavenging resins or additional purification steps to meet residual metal limits, directly lowering the cost of goods sold (COGS). Furthermore, the high selectivity of the reaction minimizes waste generation, aligning with green chemistry principles and reducing waste disposal fees.
- Enhanced Supply Chain Reliability: Supply chain continuity is often threatened by the scarcity or volatility of specialized reagents. Sulfonyl chlorides and styrene derivatives are widely produced commodity chemicals with stable global supply networks, reducing the risk of stockouts. The tolerance of the reaction to moisture (since water is a reagent) means that storage and handling requirements are less stringent, decreasing the likelihood of batch rejection due to minor environmental excursions. This resilience allows for more flexible inventory management and shorter lead times, ensuring that downstream API synthesis lines remain uninterrupted. The ability to source raw materials from multiple vendors further mitigates single-source dependency risks.
- Scalability and Environmental Compliance: Scaling chemical processes from the bench to the plant floor often reveals hidden bottlenecks, particularly regarding heat transfer and safety. The exothermic nature of Grignard reactions poses significant thermal runaway risks at scale, requiring complex cooling systems. In contrast, the mild exotherm of this acid-catalyzed addition is easily managed with standard jacketed reactors, facilitating a smoother transition from pilot plant to commercial production. Moreover, the aqueous workup and lack of heavy metals simplify wastewater treatment, ensuring compliance with increasingly stringent environmental regulations. This ease of scale-up supports the rapid deployment of capacity to meet market demand for high-purity pharmaceutical intermediates.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These insights are derived directly from the experimental data and claims presented in the patent literature, providing a factual basis for decision-making. Understanding these nuances is essential for R&D teams evaluating process feasibility and procurement officers assessing vendor capabilities. We encourage stakeholders to review these details to fully appreciate the operational improvements offered by this methodology.
Q: What are the key advantages of this acid-catalyzed method over traditional Grignard routes?
A: Unlike traditional methods requiring moisture-sensitive Grignard reagents or metal hydrides, this process utilizes stable sulfonyl chlorides and olefins under mild acidic conditions (20°C-70°C), significantly reducing operational hazards and equipment costs.
Q: What is the typical yield and purity profile for these beta-hydroxy sulfone derivatives?
A: According to the patent data, isolated yields range from 65% to 75% after purification via silica gel column chromatography. The method demonstrates high selectivity, minimizing byproduct formation and simplifying downstream processing.
Q: Can this process be adapted for large-scale commercial production?
A: Yes, the use of common solvents like THF and stable reagents, combined with moderate temperature requirements (20°C-70°C), makes this route highly amenable to scale-up from laboratory grams to multi-ton commercial batches without specialized cryogenic infrastructure.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Beta-Hydroxy Sulfone Supplier
At NINGBO INNO PHARMCHEM, we combine deep technical expertise with robust manufacturing capabilities to deliver superior chemical solutions. Our team has extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory discovery to industrial reality is seamless. We maintain stringent purity specifications through our rigorous QC labs, utilizing advanced analytical techniques to verify the identity and quality of every batch. Our commitment to excellence means that we do not just supply chemicals; we provide validated, scalable processes that enhance your own production efficiency and product reliability.
We invite you to collaborate with us to leverage this advanced synthesis technology for your specific applications. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your volume requirements and quality standards. Please contact us to request specific COA data and route feasibility assessments for your target molecules. By partnering with us, you gain access to a reliable supply chain partner dedicated to driving innovation and value in the global pharmaceutical and fine chemical markets.
