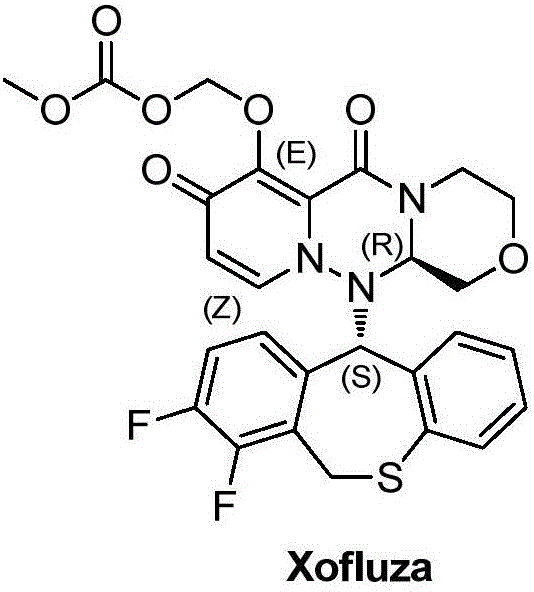
Advanced Manufacturing of Xofluza Sulfur-Heterocyclic Intermediates via Optimized Grignard and Radical Halogenation
Introduction to Next-Generation Xofluza Intermediate Manufacturing
The global pharmaceutical landscape has been significantly impacted by the approval of Xofluza (baloxavir marboxil), a groundbreaking cap-dependent endonuclease inhibitor for treating influenza. As demand for this potent antiviral agent grows, the efficiency and sustainability of its supply chain become paramount. Patent CN110872247A introduces a transformative preparation method for the critical sulfur-containing heterocyclic intermediates required for Xofluza synthesis. This technology addresses long-standing bottlenecks in the production of Compound 5, a pivotal precursor that eventually cyclizes to form the core heterocyclic structure of the drug. By shifting away from hazardous cryogenic conditions and toxic reagents, this innovation offers a robust pathway for reliable pharmaceutical intermediate supplier networks to enhance their production capabilities.
The structural complexity of Xofluza necessitates precise chemical transformations to ensure high purity and yield. The patented method focuses on optimizing the synthesis of the difluorophenyl-thioether scaffold, which is essential for the biological activity of the final API. Unlike traditional routes that struggle with scalability and safety, this new approach leverages mild reaction conditions and widely available raw materials. The strategic redesign of the synthetic pathway not only mitigates environmental risks but also aligns with modern green chemistry principles, making it an attractive option for cost reduction in API manufacturing. This report delves into the technical nuances and commercial implications of adopting this superior synthetic strategy.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
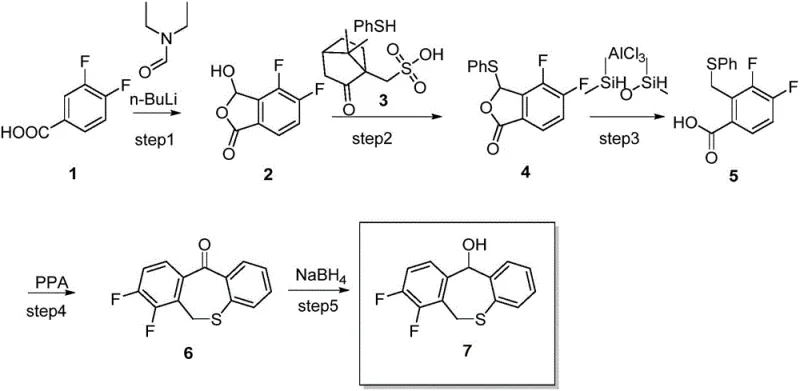
Prior art technologies, such as those disclosed in TW201802097A, have historically relied on aggressive and operationally challenging conditions to construct the sulfur-heterocyclic framework. A primary drawback is the dependence on n-butyllithium (n-BuLi) for lithiation steps, which mandates cryogenic temperatures as low as -40°C. Maintaining such low temperatures on an industrial scale requires specialized, energy-intensive refrigeration infrastructure, directly inflating operational expenditures. Furthermore, to prevent dangerous intermolecular side reactions, these legacy processes must operate at extremely low concentrations, often limiting feed batches to mere hundreds of grams in large 20-liter reactors. This gross underutilization of reactor volume creates a severe bottleneck for commercial scale-up of complex heterocycles, rendering the process economically unviable for high-volume production.
Safety and supply chain stability are further compromised by the reliance on thiophenol as a key nucleophile. Thiophenol is notorious for its intense, repulsive odor and high toxicity, posing significant occupational health risks and requiring stringent containment measures that many facilities cannot easily accommodate. Additionally, the market availability of high-quality thiophenol is often restricted due to regulatory controls on hazardous chemicals, leading to potential supply disruptions. The combination of harsh reaction conditions, poor reactor efficiency, and hazardous raw materials creates a fragile supply chain that struggles to meet the rigorous demands of the global antiviral market, highlighting the urgent need for a more robust manufacturing solution.
The Novel Approach
The methodology outlined in CN110872247A represents a paradigm shift by replacing the problematic n-BuLi step with a Grignard reaction using isopropyl magnesium chloride. This modification allows the carboxylation step to proceed at mild temperatures ranging from 0°C to 40°C, completely eliminating the need for energy-draining cryogenic cooling. The reaction can be performed at significantly higher molar concentrations (0.5 to 5 mol/L), which dramatically improves the utilization rate of the reaction kettle and boosts overall throughput. This enhancement in spatial efficiency translates directly to lower capital expenditure per kilogram of product, facilitating a more agile response to market demand fluctuations without the need for massive infrastructure upgrades.
Crucially, the new route substitutes toxic thiophenol with sodium thiophenolate, a solid reagent that is far safer to handle, possesses minimal odor, and is readily available in bulk quantities from standard chemical suppliers. This switch not only enhances workplace safety and environmental compliance but also stabilizes the supply chain by removing dependency on restricted hazardous materials. The entire sequence, from the initial bromination to the final nucleophilic substitution, operates under温和 conditions (0-120°C), ensuring that the process is inherently safer and more controllable. This holistic optimization makes the technology ideally suited for industrial production, offering a clear path toward reducing lead time for high-purity intermediates while maintaining strict quality standards.
Mechanistic Insights into Grignard Carboxylation and Radical Halogenation
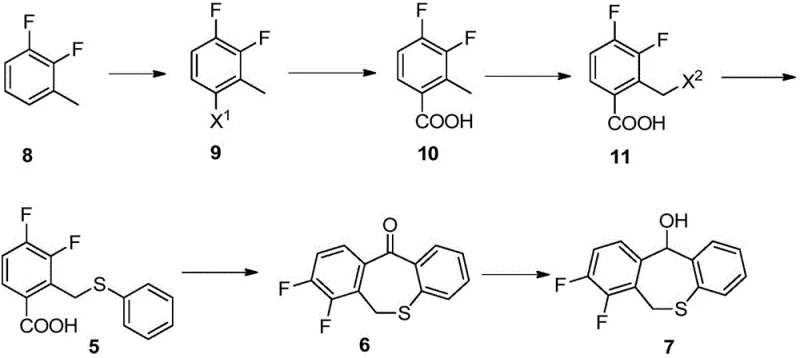
The core of this technological advancement lies in the precise orchestration of organometallic and radical chemistry to functionalize the aromatic core. The process initiates with the formation of a Grignard reagent from 4-bromo-2,3-difluorotoluene (Compound 9) using isopropyl magnesium chloride in tetrahydrofuran. Unlike lithium-halogen exchange which is highly exothermic and sensitive, the magnesium insertion is more controlled. Subsequent quenching with carbon dioxide gas efficiently installs the carboxylic acid moiety to yield Compound 10. This step is critical as it establishes the anchor point for the subsequent heterocyclic ring closure. The use of CO2 as a C1 source is not only atom-economical but also avoids the use of more expensive or hazardous carbonyl equivalents. The reaction kinetics are optimized by maintaining a specific molar ratio of Grignard reagent to substrate (preferably 1.5:1), ensuring complete conversion while minimizing the formation of homocoupling byproducts that could complicate downstream purification.
Following carboxylation, the methyl group on the aromatic ring undergoes selective radical bromination to generate the reactive benzyl bromide intermediate (Compound 11). This transformation utilizes N-bromosuccinimide (NBS) in the presence of a radical initiator like AIBN at elevated temperatures (80-85°C). The mechanism involves the generation of succinimidyl radicals which abstract a hydrogen atom from the benzylic position, creating a stable benzylic radical that subsequently reacts with bromine. The selectivity of this reaction is paramount; the conditions are tuned to brominate the methyl group exclusively without affecting the aromatic ring or the carboxylic acid functionality. Finally, the resulting bromomethyl group serves as an excellent leaving group for a nucleophilic substitution reaction with sodium thiophenolate. This SN2 displacement proceeds smoothly in polar aprotic solvents like DMF, installing the sulfur linkage with high fidelity. The mechanistic robustness of these steps ensures a clean impurity profile, which is essential for meeting the stringent purity specifications required for pharmaceutical intermediates.
How to Synthesize Xofluza Intermediate Efficiently
The synthesis of the key sulfur-containing intermediate involves a streamlined sequence that prioritizes operational simplicity and safety. The process begins with the preparation of the benzoic acid scaffold via Grignard chemistry, followed by selective functionalization of the side chain. Each step has been rigorously optimized to balance reaction rate with selectivity, ensuring that the final product meets the necessary quality thresholds for downstream cyclization. The detailed standardized synthesis steps below outline the specific reagents, stoichiometry, and workup procedures required to replicate this high-efficiency route in a GMP-compliant environment.
- Perform Grignard carboxylation on 4-bromo-2,3-difluorotoluene using isopropyl magnesium chloride and carbon dioxide to form the benzoic acid derivative.
- Execute selective radical bromination of the methyl group using N-bromosuccinimide (NBS) and AIBN initiator to generate the bromomethyl intermediate.
- Conduct nucleophilic substitution with sodium thiophenolate in an amide solvent to install the phenylthio group, yielding the final intermediate.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this patented synthesis route offers tangible strategic benefits that extend beyond simple chemical yield. The elimination of cryogenic requirements removes a significant barrier to entry for many contract manufacturing organizations, broadening the pool of qualified vendors capable of producing this intermediate. This increased competition and capacity availability naturally drive down costs and mitigate the risk of supply shortages. Furthermore, the substitution of hazardous thiophenol with benign sodium thiophenolate simplifies logistics, as the latter does not require the same level of specialized hazardous material handling and storage, thereby reducing insurance and compliance overheads associated with the transportation and warehousing of raw materials.
- Cost Reduction in Manufacturing: The shift from n-BuLi to Grignard reagents eliminates the need for expensive cryogenic cooling systems, resulting in substantial energy savings. Additionally, the ability to run reactions at higher concentrations means that the same reactor volume can produce significantly more product per batch, effectively lowering the fixed cost allocation per kilogram. The use of commercially abundant sodium thiophenolate instead of scarce, regulated thiophenol further reduces raw material procurement costs and minimizes waste disposal expenses related to toxic byproducts.
- Enhanced Supply Chain Reliability: By utilizing raw materials that are widely sourced and less regulated, the supply chain becomes more resilient to market shocks. Sodium thiophenolate is a commodity chemical with multiple global suppliers, ensuring continuity of supply even during regional disruptions. The robustness of the reaction conditions also means that production is less susceptible to delays caused by equipment maintenance or utility failures, providing a more predictable delivery schedule for downstream API manufacturers who rely on just-in-time inventory models.
- Scalability and Environmental Compliance: The process is inherently designed for scale, with reaction parameters that translate linearly from pilot plant to multi-ton commercial production. The avoidance of highly toxic reagents and the generation of less hazardous waste streams simplify environmental permitting and wastewater treatment. This alignment with green chemistry principles not only future-proofs the manufacturing site against tightening environmental regulations but also enhances the corporate sustainability profile of the final pharmaceutical product, a factor increasingly valued by end-market stakeholders.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this novel synthesis pathway. These answers are derived directly from the experimental data and comparative analysis provided in the patent documentation, offering clarity on how this method outperforms legacy technologies in terms of safety, efficiency, and product quality. Understanding these distinctions is crucial for technical teams evaluating process transfer feasibility.
Q: How does the new synthesis route improve safety compared to prior art?
A: The new method replaces highly toxic and malodorous thiophenol with sodium thiophenolate, significantly reducing occupational health hazards and odor complaints in the facility.
Q: What are the energy consumption benefits of this process?
A: By eliminating the need for cryogenic cooling down to -40°C required for n-BuLi reactions, the process operates at mild temperatures (0-40°C), drastically cutting energy costs.
Q: Is this process suitable for large-scale industrial production?
A: Yes, the method allows for much higher reaction concentrations compared to the dilute conditions of previous methods, maximizing reactor utilization and throughput for commercial scale-up.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Xofluza Intermediate Supplier
As the pharmaceutical industry continues to evolve, the demand for efficient and sustainable manufacturing processes for critical antiviral intermediates like those for Xofluza is at an all-time high. NINGBO INNO PHARMCHEM stands at the forefront of this evolution, leveraging advanced synthetic methodologies to deliver high-value chemical solutions. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that we can meet the volumetric needs of global partners without compromising on quality. We operate with stringent purity specifications and utilize rigorous QC labs to guarantee that every batch of intermediate meets the exacting standards required for API synthesis, providing our clients with peace of mind and regulatory confidence.
We invite you to explore how our technical expertise can optimize your supply chain for Xofluza production. By partnering with us, you gain access to a Customized Cost-Saving Analysis tailored to your specific volume requirements and logistical constraints. We encourage you to contact our technical procurement team today to request specific COA data and route feasibility assessments. Let us demonstrate how our commitment to innovation and operational excellence can drive value for your organization, ensuring a secure and cost-effective supply of high-purity pharmaceutical intermediates for the global market.
