Scalable Production of High-Purity (R)/(S)-6,6'-Dihydroxy-5,5'-Bisquinoline for Asymmetric Catalysis
The pharmaceutical and fine chemical industries are constantly seeking robust, scalable methods for producing high-value chiral ligands, which are the cornerstone of modern asymmetric catalysis. Patent CN102731388A introduces a groundbreaking methodology for the preparation of (R)/(S)-6,6'-dihydroxy-5,5'-bisquinoline, a sophisticated C2-symmetric nitrogen-containing ligand analogous to the widely used BINOL. This innovation addresses critical bottlenecks in the supply chain of chiral intermediates by replacing costly preparative HPLC separation with an efficient crystallization-based resolution strategy. For R&D directors and procurement managers alike, this represents a significant leap forward in process chemistry, offering a pathway to reduce the cost of goods sold (COGS) while maintaining stringent purity standards required for GMP manufacturing. The method leverages accessible starting materials and standard unit operations, making it an ideal candidate for commercial scale-up.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the isolation of optically pure 6,6'-dihydroxy-5,5'-bisquinoline has been a formidable challenge, primarily relying on the derivatization of the racemate followed by separation on expensive chiral stationary phases via High-Performance Liquid Chromatography (HPLC). This conventional approach suffers from severe limitations regarding throughput and economic viability; chiral preparative columns are not only capital-intensive but also have limited lifespans and loading capacities. Furthermore, the solvent consumption associated with large-scale chromatographic separation creates substantial waste disposal issues and environmental compliance burdens. For supply chain heads, the reliance on such specialized, low-throughput technology introduces significant risk regarding lead times and continuity of supply, particularly when demand surges for downstream API production. The complexity of optimizing mobile phases and the difficulty in recycling chiral selectors further exacerbate the operational inefficiencies inherent in the traditional HPLC-based workflow.
The Novel Approach
In stark contrast, the novel approach detailed in the patent utilizes a classical yet highly effective diastereomeric resolution strategy driven by fractional crystallization. By reacting the racemic bisquinoline with D-(+)-camphorsulfonyl chloride, the enantiomers are converted into diastereomeric biscamphorsulfonates, which exhibit distinct solubility profiles in specific ternary solvent systems. This physicochemical difference allows for the selective precipitation of the (S)-isomer using a mixture of ethyl acetate, methanol, and n-hexane, while the (R)-isomer can be subsequently recovered from the mother liquor through careful solvent adjustment and recrystallization. This shift from chromatographic separation to crystallization fundamentally transforms the economics of production, enabling the use of standard stainless steel reactors and filtration equipment. The result is a process that is not only simpler to operate but also inherently more scalable, providing a reliable source of high-purity chiral ligands without the prohibitive costs associated with chiral columns.
Mechanistic Insights into CuCl2-Catalyzed Oxidative Coupling
The synthesis begins with the construction of the biaryl backbone through an oxidative coupling reaction, a critical step that defines the overall yield and impurity profile of the process. As illustrated in the reaction scheme below, 6-hydroxyquinoline undergoes dimerization in the presence of copper(II) chloride and an organic amine base, such as benzylamine, within a methanol solvent system. The mechanism likely involves the coordination of the quinoline nitrogen and the phenolic oxygen to the copper center, facilitating a single-electron transfer (SET) process that generates radical intermediates. These radicals then couple at the C5 position to form the sterically hindered 5,5'-bond. The choice of organic amine is crucial, as it acts not only as a base to neutralize the generated acid but also potentially as a ligand that modulates the redox potential of the copper catalyst. This step proceeds at room temperature over an extended period, typically 48 to 96 hours, ensuring complete conversion while minimizing over-oxidation or polymerization side reactions that could complicate downstream purification.
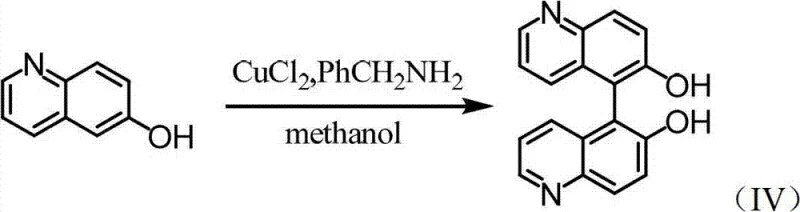
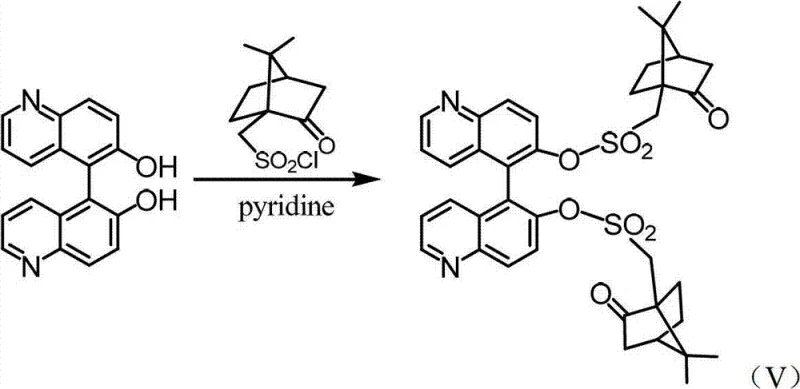
Following the formation of the racemic backbone, the chiral information is introduced via esterification with D-(+)-camphorsulfonyl chloride. This reaction, depicted in the subsequent scheme, converts the hydroxyl groups of the bisquinoline into bulky camphorsulfonate esters. The steric bulk of the camphor moiety, combined with its rigid chiral framework, creates significant diastereomeric differentiation between the (R,R) and (S,S) configurations of the bisquinoline core. This differentiation is the thermodynamic driving force for the subsequent resolution; the diastereomers pack differently in the crystal lattice, leading to the observed solubility differences. The use of pyridine as both solvent and base ensures mild conditions that preserve the integrity of the sensitive quinoline rings while driving the sulfonylation to completion. This derivatization step is pivotal, as it transforms an inseparable mixture of enantiomers into a separable mixture of diastereomers, unlocking the potential for high-efficiency purification through simple physical means.
How to Synthesize (R)/(S)-6,6'-Dihydroxy-5,5'-Bisquinoline Efficiently
The practical execution of this synthesis requires precise control over solvent composition and temperature gradients to maximize the recovery of both enantiomers. The process is designed to be telescoped where possible, minimizing the number of isolation steps and reducing solvent waste. Initial coupling yields the racemate, which is then directly subjected to sulfonylation without extensive purification, relying on the crystallization steps to remove impurities. The critical resolution phase involves dissolving the crude diastereomeric mixture in a heated ternary solvent system and allowing it to cool slowly, promoting the nucleation of the less soluble (S)-diastereomer. The mother liquor, enriched in the (R)-diastereomer, is then concentrated and treated with a slightly different solvent ratio to induce the precipitation of the complementary enantiomer. Finally, the pure diastereomeric esters are hydrolyzed under basic conditions to release the free chiral ligands. For detailed operational parameters and safety guidelines, please refer to the standardized protocol below.
- Perform oxidative coupling of 6-hydroxyquinoline using CuCl2 and organic amines in methanol to form racemic 6,6'-dihydroxy-5,5'-bisquinoline.
- React the racemic mixture with D-(+)-camphorsulfonyl chloride in pyridine to form diastereomeric biscamphorsulfonates.
- Separate the (S) and (R) diastereomers via fractional crystallization in ethyl acetate/methanol/hexane mixtures, followed by alkaline hydrolysis.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain strategists, the adoption of this crystallization-based methodology offers profound advantages in terms of cost stability and supply security. By eliminating the dependency on consumable chiral HPLC columns, the variable costs associated with production are drastically reduced, leading to a more predictable pricing structure for long-term contracts. The raw materials required, including 6-hydroxyquinoline, copper salts, and camphorsulfonyl chloride, are commodity chemicals available from multiple global suppliers, mitigating the risk of single-source bottlenecks. Furthermore, the scalability of crystallization processes is well-understood in the chemical industry, allowing for seamless transition from kilogram-scale pilot runs to multi-ton annual production without the need for specialized chromatographic skids. This flexibility ensures that supply can be rapidly ramped up to meet fluctuating market demands, providing a reliable buffer against supply chain disruptions.
- Cost Reduction in Manufacturing: The elimination of expensive chiral stationary phases and the reduction in solvent volume compared to preparative HPLC result in substantial cost savings. The process utilizes standard reactor vessels and filtration equipment, avoiding the high capital expenditure associated with specialized separation technologies. Additionally, the ability to recover both the (R) and (S) enantiomers from the same batch maximizes atom economy and raw material utilization, further driving down the cost per kilogram of the final active ligand.
- Enhanced Supply Chain Reliability: Sourcing strategies are simplified as the process relies on widely available bulk chemicals rather than proprietary or niche reagents. The robustness of the crystallization steps ensures consistent batch-to-batch quality, reducing the likelihood of production delays caused by failed purification cycles. This reliability is critical for pharmaceutical customers who require uninterrupted supply of key intermediates to maintain their own API manufacturing schedules and regulatory filings.
- Scalability and Environmental Compliance: The process is inherently green, generating less hazardous waste compared to chromatographic methods which produce large volumes of contaminated solvent mixtures. The solvents used, such as ethyl acetate and hexane, are easily recoverable through distillation, supporting sustainability goals and reducing disposal costs. The simplicity of the unit operations facilitates easy scale-up, allowing manufacturers to increase capacity quickly to support commercial launches of new drugs utilizing these chiral ligands.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis route. Understanding these details is essential for process engineers and quality assurance teams evaluating the feasibility of integrating this intermediate into their supply chain. The answers are derived directly from the experimental data and claims presented in the patent documentation, ensuring accuracy and relevance for technical decision-making.
Q: Why is this crystallization method preferred over HPLC separation?
A: Traditional HPLC separation on chiral columns is time-consuming, expensive due to column costs, and difficult to scale. This patent utilizes diastereomeric crystallization, which significantly reduces operational costs and allows for large-scale industrial production without specialized chromatography equipment.
Q: What is the optical purity achieved with this method?
A: The method achieves high optical purity, with HPLC analysis showing ee values greater than 99% for both the (S)- and (R)-enantiomers after the crystallization steps, ensuring suitability for high-performance asymmetric catalysis.
Q: What solvents are used for the critical resolution step?
A: The resolution relies on ternary solvent systems. Solvent A (Ethyl Acetate:Methanol:n-Hexane = 1:0.8:1) is used to crystallize the (S)-isomer, while Solvent B (Ethyl Acetate:Methanol:n-Hexane = 1:1:1) is used to recover the (R)-isomer from the mother liquor.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 6,6'-Dihydroxy-5,5'-Bisquinoline Supplier
At NINGBO INNO PHARMCHEM, we recognize the strategic importance of high-quality chiral ligands in the development of next-generation pharmaceuticals and advanced materials. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory discovery to industrial reality is seamless and efficient. We are committed to delivering products that meet stringent purity specifications, supported by our rigorous QC labs equipped with state-of-the-art analytical instrumentation. Whether you require custom synthesis of novel derivatives or reliable supply of established intermediates, our infrastructure is designed to support your most demanding projects with speed and precision.
We invite you to engage with our technical procurement team to discuss how this innovative synthesis route can optimize your specific manufacturing requirements. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into the potential economic benefits of switching to this crystallization-based method. We encourage you to contact us today to obtain specific COA data and route feasibility assessments tailored to your project timelines, ensuring that your supply chain remains robust, compliant, and cost-effective in a competitive global market.
