Scalable Palladium-Catalyzed Synthesis of Almotriptan Intermediates for Commercial API Production
The pharmaceutical landscape for migraine treatment has long relied on the efficient production of serotonin receptor agonists, with Almotriptan standing out as a critical therapeutic agent. A pivotal advancement in the manufacturing of this active pharmaceutical ingredient is detailed in patent CN109928910B, which discloses a novel preparation method that fundamentally shifts the synthetic paradigm from hazardous, low-yielding processes to a robust, palladium-catalyzed tandem reaction. This technological breakthrough addresses the longstanding challenges of impurity profiles and operational complexity that have plagued previous generations of synthesis routes. By leveraging a sophisticated cascade reaction between N-protected o-haloaniline and 4-acetoxycrotonate, the patented methodology delivers the essential indole skeleton with remarkable efficiency and purity. For global procurement and R&D teams, understanding this shift is vital, as it represents a transition towards greener, more cost-effective, and scalable chemical manufacturing capable of meeting the rigorous demands of modern regulatory bodies.
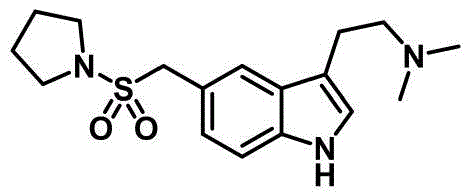
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial synthesis of almotriptan has been fraught with significant technical and economic hurdles that compromise supply chain stability and product quality. Early methods, such as those disclosed in US5565447, relied on the decarboxylation of intermediates using copper oxide in quinoline, a process characterized by excessive energy consumption and severe equipment corrosion issues that drive up capital expenditure. Furthermore, alternative routes utilizing Fisher indole synthesis, while conceptually short, introduce dangerous variables such as the use of tin chloride reducing agents which leave behind toxic tin residues that are notoriously difficult to remove to pharmaceutical standards. These legacy processes also suffer from the instability of phenylhydrazine intermediates, leading to batch-to-batch variability and the formation of polymeric byproducts that complicate purification. Additionally, other palladium-catalyzed approaches described in prior art often necessitate the use of cryogenic conditions and hazardous organolithium reagents like n-butyllithium, imposing strict safety protocols and limiting the feasible scale of production due to thermal runaway risks.
The Novel Approach
In stark contrast to these cumbersome legacy techniques, the method described in CN109928910B introduces a streamlined, one-pot tandem cyclization that constructs the indole core under remarkably mild and controllable conditions. This innovative route utilizes readily available N-protected o-haloanilines and 4-acetoxycrotonates, reacting them in the presence of a palladium catalyst and phosphine ligand to directly forge the carbon-carbon bonds necessary for the almotriptan skeleton. The process operates at moderate temperatures ranging from 80°C to 120°C, eliminating the need for cryogenic cooling or high-pressure reactors, thereby drastically reducing energy costs and safety liabilities. Crucially, the workup procedure is simplified to basic aqueous quenching and filtration, allowing for the isolation of high-purity intermediates without the need for column chromatography. This operational simplicity not only accelerates the production timeline but also ensures a consistent impurity profile that is far easier to manage during final API crystallization.
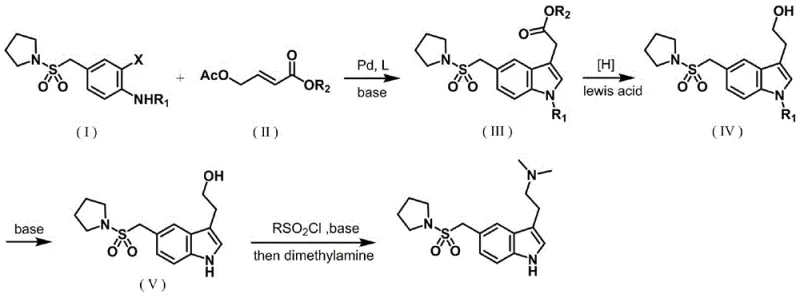
Mechanistic Insights into Pd-Catalyzed Tandem Cyclization
The core of this technological advancement lies in the elegant mechanistic pathway facilitated by the palladium catalyst, which orchestrates a sequence of oxidative addition, migratory insertion, and reductive elimination to construct the heterocyclic ring system. Initially, the palladium(0) species undergoes oxidative addition into the carbon-halogen bond of the protected o-haloaniline, generating a reactive aryl-palladium complex. This intermediate then coordinates with the electron-deficient double bond of the 4-acetoxycrotonate, followed by a migratory insertion that establishes the critical carbon-carbon linkage at the 3-position of the future indole ring. The presence of the acetoxy group serves as a latent leaving group, facilitating an intramolecular nucleophilic attack by the aniline nitrogen onto the activated alkene moiety, effectively closing the five-membered ring. Finally, beta-hydride elimination and subsequent reductive elimination regenerate the active palladium catalyst, completing the cycle and releasing the protected indole ester. This mechanism is highly selective, minimizing the formation of regioisomers that typically plague non-catalytic cyclization methods.
From an impurity control perspective, the choice of the N-protecting group, such as the p-toluenesulfonyl group, plays a pivotal role in stabilizing the aniline nitrogen against unwanted side reactions during the harsh conditions of the catalytic cycle. The robustness of this protecting group ensures that the nitrogen remains inert until the specific deprotection step, preventing the formation of oligomeric impurities that often arise from free amine participation in radical pathways. Furthermore, the use of specific phosphine ligands like tri(o-tolyl)phosphine modulates the electronic density on the palladium center, optimizing the rate of oxidative addition while suppressing competing homocoupling reactions of the aryl halide. This precise tuning of the catalytic environment results in a reaction mixture that is chemically clean, allowing for straightforward crystallization of the intermediate and significantly reducing the burden on downstream purification units.
How to Synthesize Almotriptan Efficiently
The synthesis of almotriptan via this patented route involves a logical sequence of transformations that convert simple starting materials into the complex final drug substance through a series of high-yielding steps. The process begins with the construction of the indole core, followed by the reduction of the ester side chain to an alcohol, removal of the protecting group, and finally, the installation of the dimethylamino ethyl tail. Each step has been optimized to maximize atom economy and minimize waste generation, making it an ideal candidate for green chemistry initiatives within pharmaceutical manufacturing. The following guide outlines the standardized operational parameters derived from the patent examples, providing a clear roadmap for process chemists looking to implement this technology.
- React N-protected o-haloaniline with 4-acetoxycrotonate using a palladium catalyst and phosphine ligand at 80-120°C to form the protected indole ester.
- Reduce the ester intermediate to the corresponding alcohol using sodium borohydride and calcium chloride in DMAc solvent.
- Perform deprotection with base, followed by hydroxyl activation with sulfonyl chloride and substitution with dimethylamine to yield almotriptan.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this palladium-catalyzed synthesis route offers profound strategic advantages that extend far beyond simple chemical yield improvements. By transitioning away from processes that rely on scarce or hazardous reagents, manufacturers can secure a more resilient supply chain that is less susceptible to regulatory crackdowns or raw material shortages. The elimination of tin-based reducing agents and organolithium reagents removes significant environmental, health, and safety (EHS) liabilities, thereby lowering insurance premiums and waste disposal costs associated with hazardous material handling. Moreover, the ability to isolate intermediates via simple filtration rather than energy-intensive distillation or chromatography translates directly into reduced utility consumption and faster batch turnover times. These operational efficiencies compound to create a manufacturing profile that is both economically superior and environmentally sustainable.
- Cost Reduction in Manufacturing: The economic impact of this new method is driven primarily by the simplification of the unit operations and the avoidance of expensive, specialized reagents. By utilizing common palladium catalysts and inexpensive phosphine ligands instead of proprietary or exotic metal complexes, the direct material cost per kilogram of intermediate is significantly lowered. Furthermore, the high selectivity of the tandem reaction minimizes the formation of difficult-to-remove impurities, which reduces the volume of solvents required for recrystallization and washing steps. This reduction in solvent usage not only cuts purchasing costs but also decreases the load on solvent recovery systems, leading to substantial overall cost savings in the production of pharmaceutical intermediates.
- Enhanced Supply Chain Reliability: The reliance on commodity chemicals such as haloanilines and crotonates ensures a stable and diversified supply base, mitigating the risk of single-source bottlenecks that often disrupt API production schedules. Unlike methods requiring strictly anhydrous and anaerobic conditions which demand specialized glassware and inert gas infrastructure, this process can be executed in standard stainless steel reactors with conventional agitation and heating systems. This compatibility with existing general-purpose manufacturing assets allows for greater flexibility in production planning and enables rapid scale-up from pilot plant to commercial tonnage without the need for significant capital investment in new equipment.
- Scalability and Environmental Compliance: From a regulatory and environmental standpoint, this process aligns perfectly with the industry's push towards greener manufacturing practices. The absence of heavy metal contaminants like tin in the final product streamlines the regulatory filing process, as extensive data on metal clearance is not required. Additionally, the aqueous workup procedures generate waste streams that are easier to treat and neutralize compared to the organic-heavy wastes generated by traditional extraction methods. This ease of waste management facilitates compliance with increasingly stringent environmental regulations, ensuring long-term operational continuity and reducing the risk of production shutdowns due to environmental non-compliance.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis route, providing clarity on its practical application and benefits. These insights are derived directly from the experimental data and comparative analysis presented in the patent documentation, offering a realistic view of the technology's capabilities. Understanding these details is crucial for stakeholders evaluating the feasibility of integrating this method into their existing manufacturing portfolios.
Q: How does this palladium-catalyzed method improve upon traditional Fisher indole synthesis?
A: Unlike Fisher synthesis which generates unstable hydrazines and tin salt residues requiring complex purification, this method uses stable haloanilines and avoids heavy metal contaminants, significantly simplifying downstream processing.
Q: What are the critical reaction conditions for the tandem cyclization step?
A: The reaction requires a palladium catalyst (such as Pd(OAc)2) and a phosphine ligand in a polar aprotic solvent like DMAc or DMF, maintained at temperatures between 80°C and 120°C under an inert atmosphere.
Q: Is this process suitable for large-scale commercial manufacturing?
A: Yes, the process avoids hazardous reagents like n-butyllithium and strict Schlenk conditions, utilizing simple filtration and crystallization for purification, which makes it highly scalable for industrial production.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Almotriptan Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition to advanced synthetic routes requires a partner with deep technical expertise and proven execution capabilities. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of this palladium-catalyzed process are fully realized in a commercial setting. We maintain stringent purity specifications and operate rigorous QC labs equipped with state-of-the-art analytical instrumentation to guarantee that every batch of almotriptan intermediate meets the highest global pharmacopeial standards. Our commitment to quality assurance ensures that our clients receive materials that are ready for immediate downstream processing, minimizing delays in their own API manufacturing timelines.
We invite forward-thinking pharmaceutical companies to collaborate with us to leverage this cutting-edge technology for their migraine treatment portfolios. By engaging with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your specific volume requirements and logistical constraints. We encourage you to reach out today to obtain specific COA data and route feasibility assessments that demonstrate how our optimized manufacturing processes can enhance your supply chain efficiency and reduce your total cost of ownership for high-value pharmaceutical intermediates.
