Advanced Manufacturing of L-2-Aminobutyrylamide Hydrochloride for Scalable Levetiracetam Production
Introduction to Novel Synthetic Pathways for Chiral Intermediates
The pharmaceutical industry continuously seeks robust and scalable methodologies for producing high-value chiral intermediates, particularly for blockbuster antiepileptic drugs like Levetiracetam. Patent CN102020584A introduces a transformative approach to synthesizing L-2-aminobutyrylamide hydrochloride, a critical precursor that dictates the quality and cost-efficiency of the final active pharmaceutical ingredient. This proprietary method diverges from traditional pathways by utilizing 2-bromobutyric acid as a foundational starting material, thereby circumventing the economic and safety bottlenecks associated with earlier technologies. By shifting the synthetic focus to a bromo-acid derivative, the process achieves a remarkable balance between operational simplicity and chemical efficiency, making it an ideal candidate for industrial adoption. The strategic implementation of mild reaction conditions and accessible reagents ensures that manufacturers can maintain consistent production schedules without compromising on safety standards or environmental compliance protocols.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of Levetiracetam intermediates has been plagued by significant technical and economic hurdles that hinder large-scale commercial viability. Traditional routes often rely on L-2-aminobutyric acid as a starting material, which is prohibitively expensive and limits the cost-effectiveness of the entire supply chain for generic manufacturers. Alternatively, other established methods employ the Strecker synthesis using sodium cyanide, a highly toxic reagent that imposes severe safety risks on operators and necessitates complex, costly waste treatment systems to manage cyanide residues. Furthermore, prior art methods involving ammonification of 2-bromobutyric acid directly often suffer from difficult separation processes where the generated ammonium bromide and the target amino acid are challenging to isolate, leading to suboptimal molar yields around 70%. These legacy processes frequently require high-pressure equipment and extended reaction times, such as 24 hours of ammonia gas introduction, which increases energy consumption and equipment maintenance costs significantly.
The Novel Approach
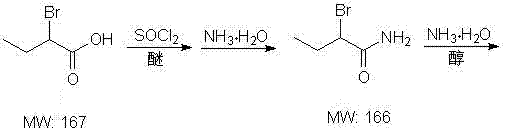
In stark contrast to these cumbersome legacy techniques, the methodology disclosed in CN102020584A offers a streamlined, three-step sequence that prioritizes safety and yield optimization through intelligent chemical design. The process initiates with the conversion of 2-bromobutyric acid into a 2-bromobutyramide intermediate using thionyl chloride and ammonia water in an ether-based solvent system, a reaction that proceeds smoothly at normal pressure and temperatures ranging from -5 to 20°C. This initial amidation step avoids the formation of difficult-to-separate salts by creating a stable amide intermediate that is easily isolated via layering and concentration. Subsequent ammonolysis of this bromo-amide generates the racemic DL-2-aminobutyrylamide with high efficiency, setting the stage for a precise chiral resolution using L-tartaric acid. This innovative route not only drastically reduces the toxicity profile of the manufacturing process by eliminating cyanide but also simplifies unit operations, allowing for easier scale-up in standard glass-lined or stainless steel reactors without specialized high-pressure infrastructure.
Mechanistic Insights into Amidation and Chiral Resolution
The core chemical transformation in this synthesis relies on the activation of the carboxylic acid group followed by a nucleophilic substitution, a mechanism that is meticulously controlled to prevent side reactions and racemization. In the first stage, thionyl chloride acts as a potent activating agent, converting the hydroxyl group of 2-bromobutyric acid into a highly reactive acyl chloride species in situ, which is immediately captured by ammonia to form the primary amide bond. The use of ether solvents such as methyl tert-butyl ether or diisopropyl ether is critical here, as they provide an optimal medium for the reaction while facilitating the subsequent phase separation of the organic product from the aqueous ammonium salts. The temperature control between -5°C and 20°C is vital to suppress potential elimination reactions that could lead to unsaturated byproducts, ensuring that the bromine atom remains intact for the subsequent nucleophilic displacement. This careful modulation of reaction kinetics ensures that the 2-bromobutyramide intermediate is obtained with high purity, often exceeding 98%, which is essential for the success of the downstream amination step.
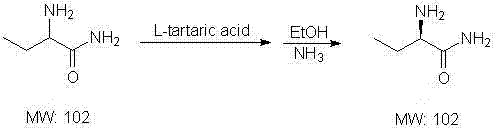
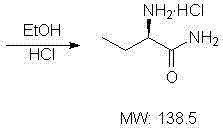
Following the formation of the bromo-amide, the process advances to a nucleophilic substitution where ammonia displaces the bromine atom to generate the alpha-amino functionality, creating the racemic DL-2-aminobutyrylamide. The resolution of this racemate is achieved through the formation of a diastereomeric salt with L-tartaric acid in an ethanol solvent, exploiting the differential solubility of the L-tartrate salt compared to its D-counterpart. This classical resolution technique is highly effective because the L-tartrate salt of the L-enantiomer crystallizes preferentially, allowing for physical separation via filtration while the mother liquor retains the unwanted D-isomer. The final step involves liberating the free base from the tartrate salt using ammonia, followed by salification with hydrochloric acid to yield the stable L-2-aminobutyrylamide hydrochloride salt. This multi-stage purification strategy ensures that the final product meets stringent optical purity requirements necessary for chiral drug synthesis, effectively removing trace impurities that could affect the efficacy of the final Levetiracetam API.
How to Synthesize L-2-Aminobutyrylamide Hydrochloride Efficiently
Executing this synthesis requires precise adherence to the stoichiometric ratios and thermal profiles outlined in the patent to maximize yield and minimize waste generation. The process begins with the careful addition of thionyl chloride to a cooled mixture of 2-bromobutyric acid and ammonia water, maintaining a molar ratio of approximately 1:1:2 to ensure complete conversion without excess reagent carryover. Operators must monitor the exothermic nature of the amidation reaction closely, utilizing efficient cooling systems to keep the temperature within the specified -5 to 20°C window during the dropwise addition and subsequent stirring period of 2 to 10 hours. For the detailed standardized operating procedures, including specific workup instructions and crystallization parameters, please refer to the structured guide below.
- React 2-bromobutyric acid with thionyl chloride and ammonia water in an ether solvent at -5 to 20°C to form 2-bromobutyramide.
- Subject the 2-bromobutyramide intermediate to ammonolysis with ammonia water at 10 to 60°C to generate DL-2-aminobutyrylamide.
- Resolve the DL-racemate using L-tartaric acid in ethanol, followed by alkalization and salification with HCl to obtain the pure L-enantiomer hydrochloride salt.
Commercial Advantages for Procurement and Supply Chain Teams
From a strategic procurement perspective, this synthetic route offers substantial advantages by decoupling production costs from the volatile pricing of chiral starting materials like L-2-aminobutyric acid. By utilizing 2-bromobutyric acid, a commodity chemical available in bulk quantities, manufacturers can achieve significant cost reduction in pharmaceutical intermediates manufacturing without sacrificing quality or yield. The elimination of sodium cyanide not only enhances workplace safety but also removes the need for expensive cyanide destruction units and specialized hazardous waste disposal contracts, leading to a leaner operational expenditure model. Furthermore, the ability to conduct reactions at normal pressure and moderate temperatures reduces the capital expenditure required for reactor specifications, allowing existing general-purpose facilities to be utilized for production without major retrofitting investments.
- Cost Reduction in Manufacturing: The transition to a non-cyanide, bromo-acid based route fundamentally alters the cost structure by replacing expensive chiral pool starting materials with affordable racemic precursors that are resolved later in the process. This shift eliminates the premium pricing associated with enantiomerically pure raw materials, allowing for a more predictable and stable cost of goods sold over time. Additionally, the high yields reported in the patent examples, often exceeding 85% for the intermediate steps, mean that less raw material is wasted per kilogram of final product, further driving down the effective unit cost. The simplified workup procedures, which rely on standard layering and crystallization rather than complex distillations or chromatography, also reduce utility consumption and labor hours per batch.
- Enhanced Supply Chain Reliability: Sourcing 2-bromobutyric acid and thionyl chloride is significantly more reliable than securing specialized chiral acids or managing the strict regulatory controls associated with cyanide procurement. This abundance of raw materials ensures that production schedules are less susceptible to supply disruptions caused by vendor shortages or regulatory hold-ups on controlled substances. The robustness of the chemistry, which tolerates a range of temperatures and uses common solvents like ethanol and ether, means that production can be easily transferred between different manufacturing sites or scaled up rapidly to meet surging market demand for Levetiracetam. This flexibility is crucial for maintaining continuity of supply in the global pharmaceutical market, where interruptions can have severe downstream consequences.
- Scalability and Environmental Compliance: The process is inherently designed for industrial scale-up, utilizing unit operations that are well-understood and easily managed in large-scale reactors, from 100 kgs to 100 MT annual commercial production. The absence of toxic cyanide waste streams simplifies environmental compliance, reducing the burden on wastewater treatment facilities and minimizing the risk of regulatory fines or shutdowns due to effluent violations. Moreover, the ability to recycle the mother liquor containing the D-isomer back into the process for racemization or alternative use enhances the overall atom economy of the synthesis, aligning with modern green chemistry principles. This environmental stewardship not only protects the ecosystem but also strengthens the brand reputation of the manufacturer as a responsible and sustainable partner in the pharmaceutical supply chain.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented synthesis method for L-2-aminobutyrylamide hydrochloride. These insights are derived directly from the experimental data and claims presented in CN102020584A, providing a factual basis for evaluating the technology's feasibility. Understanding these details is essential for technical teams assessing the integration of this route into their existing manufacturing portfolios.
Q: Why is the 2-bromobutyric acid route preferred over Strecker synthesis for Levetiracetam intermediates?
A: The 2-bromobutyric acid route eliminates the use of highly toxic sodium cyanide required in Strecker synthesis, significantly improving process safety and reducing environmental compliance costs while utilizing cheaper, readily available starting materials.
Q: What are the critical reaction conditions for the initial amidation step?
A: The amidation of 2-bromobutyric acid with thionyl chloride and ammonia water is conducted under mild conditions, specifically at temperatures between -5°C and 20°C and at normal pressure, ensuring easy control and high yield.
Q: How is optical purity achieved in this synthesis method?
A: Optical purity is achieved through a classical resolution method where the racemic DL-2-aminobutyrylamide is reacted with L-tartaric acid to form a diastereomeric salt, allowing for the separation of the desired L-enantiomer via crystallization.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable L-2-Aminobutyrylamide Hydrochloride Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of high-purity intermediates in the successful development and commercialization of chiral drugs like Levetiracetam. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that we can meet your volume requirements with consistent quality and reliability. We operate stringent purity specifications and maintain rigorous QC labs equipped with advanced analytical instrumentation to verify the identity, potency, and chiral purity of every batch we produce. Our commitment to excellence means that we do not just supply chemicals; we deliver validated solutions that integrate seamlessly into your downstream synthesis processes, minimizing your risk and accelerating your time to market.
We invite you to engage with our technical procurement team to discuss how this optimized synthetic route can benefit your specific project needs. By requesting a Customized Cost-Saving Analysis, you can gain a clear understanding of the economic advantages of switching to this safer, more efficient methodology. We encourage potential partners to contact us directly to obtain specific COA data and route feasibility assessments tailored to your production targets, ensuring that you have all the necessary information to make informed sourcing decisions for your pharmaceutical supply chain.
