Advanced Optical Resolution of Trans-2-Aminocyclohexanol for Commercial Pharmaceutical Manufacturing
Advanced Optical Resolution of Trans-2-Aminocyclohexanol for Commercial Pharmaceutical Manufacturing
The pharmaceutical and agrochemical industries continuously demand chiral building blocks with exceptional optical purity to ensure the efficacy and safety of final drug products. Patent CN101743218B introduces a transformative methodology for the preparation of optically active trans-2-aminocyclohexanol, a critical intermediate in the synthesis of various bioactive compounds. This technology addresses the longstanding challenges associated with traditional resolution techniques by employing optically active 2-methoxyphenylacetic acid as a superior resolving agent. By shifting away from complex enzymatic pathways or inefficient tartaric acid derivatives, this invention offers a robust, high-yield route that is perfectly aligned with the rigorous demands of modern GMP manufacturing environments. The strategic implementation of this process allows for the production of high-purity intermediates while significantly simplifying the downstream purification workflow.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the production of optically active trans-2-aminocyclohexanol has been hindered by significant technical and economic bottlenecks inherent in prior art methodologies. As illustrated in the reaction scheme below, traditional approaches often relied on enzymatic asymmetric hydrolysis of acetate derivatives, which necessitated the use of dilute reaction solutions and expensive biocatalysts. These enzymatic processes frequently suffered from low volumetric productivity and required complex downstream processing to remove protein residues and enzyme inhibitors. Furthermore, alternative chemical resolution methods using agents like dibenzoyl-L-tartaric acid often failed to achieve satisfactory optical purity, typically capping at approximately 80% ee even after multiple recrystallization cycles. This inefficiency not only increased material costs due to yield loss but also extended production timelines, making these conventional routes economically unviable for large-scale commercial applications.
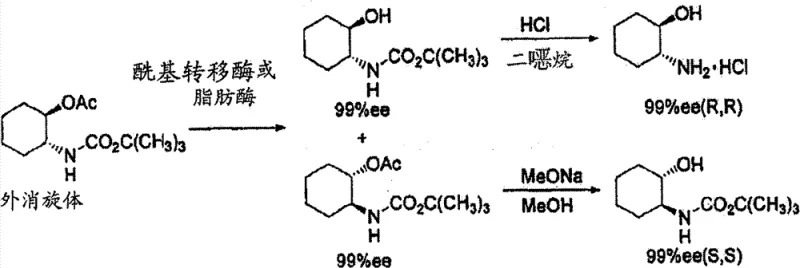
The Novel Approach
In stark contrast to these legacy methods, the novel approach disclosed in the patent utilizes a direct diastereomeric salt formation strategy that dramatically streamlines the production workflow. By reacting racemic trans-2-aminocyclohexanol directly with optically active 2-methoxyphenylacetic acid, the process leverages the distinct solubility differences between the resulting diastereomeric salts to achieve high-efficiency separation. This method eliminates the need for prior derivatization steps, such as acetylation, thereby reducing the number of unit operations and minimizing waste generation. The use of 2-methoxyphenylacetic acid as a resolving agent is particularly advantageous because it facilitates the attainment of optical purities exceeding 99% ee in a single crystallization step under optimized conditions. This breakthrough effectively resolves the purity issues associated with tartaric acid salts while avoiding the operational complexities of enzymatic systems, providing a clear path toward cost-effective industrial manufacturing.
Mechanistic Insights into Diastereomeric Salt Resolution
The core of this technological advancement lies in the precise molecular recognition between the chiral amine and the chiral acid, which drives the selective crystallization of the desired enantiomer. The process begins with the synthesis of the racemic starting material, which can be efficiently prepared from cyclohexene oxide and ammonia, as depicted in the reaction pathway below. This upstream synthesis is highly favorable for supply chain stability because it relies on inexpensive, commodity-grade raw materials that are readily available in the global chemical market. The ring-opening of cyclohexene oxide with excess ammonia proceeds with high regioselectivity to yield the trans-isomer predominantly, establishing a solid foundation for the subsequent resolution step. The ability to generate the racemate in high yield and purity ensures that the input quality for the resolution process remains consistent, which is critical for maintaining batch-to-batch reproducibility in a commercial setting.
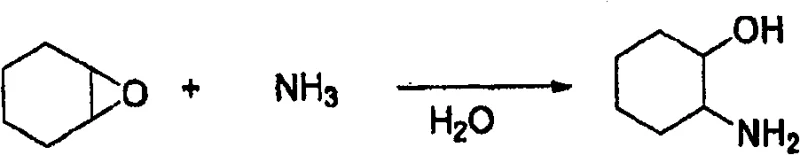
Furthermore, the resolving agent itself, optically active 2-methoxyphenylacetic acid, can be sourced reliably or synthesized via a straightforward methylation of mandelic acid, as shown in the following scheme. This synthetic accessibility ensures that the resolving agent does not become a bottleneck in the supply chain. The mechanism of resolution involves the formation of a crystalline lattice where the matching enantiomers pack more efficiently than the mismatched pair, leading to preferential precipitation. The patent data indicates that by controlling parameters such as solvent composition (preferably water or alcohols) and temperature gradients, the solubility gap between the diastereomers can be maximized. For instance, operating within a temperature range of 20°C to 80°C allows for complete dissolution followed by controlled crystallization, which is essential for excluding impurities and the unwanted enantiomer from the crystal lattice. This level of control is what enables the process to consistently deliver product with optical purity greater than 99% ee.
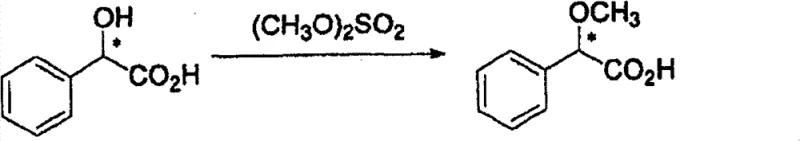
How to Synthesize Optically Active Trans-2-Aminocyclohexanol Efficiently
The execution of this synthesis requires careful attention to stoichiometry and thermal management to maximize the recovery of the target enantiomer. The patent outlines a procedure where the racemic amine and the chiral acid are mixed in a molar ratio of approximately 1:1, preferably in an aqueous or alcoholic solvent system. The mixture is heated to ensure complete dissolution, typically between 60°C and 80°C, and then slowly cooled to induce crystallization of the less soluble diastereomeric salt. This cooling phase is critical; a gradual reduction in temperature over several hours promotes the growth of large, pure crystals rather than rapid precipitation of impure solids. Once the crystals are formed, they are filtered and washed with cold solvent to remove any adhering mother liquor containing the unwanted enantiomer. The isolated salt is then subjected to decomposition, usually by treatment with a strong base or acid followed by extraction, to liberate the free optically active amine. Detailed standardized synthesis steps see the guide below.
- React racemic trans-2-aminocyclohexanol with optically active 2-methoxyphenylacetic acid in a solvent such as water or alcohol to form a diastereomeric salt.
- Crystallize the formed salt by cooling the solution, then filter and wash the crystals to isolate the desired enantiomer with high optical purity.
- Decompose the isolated salt using an acid or base treatment followed by extraction to recover the free optically active trans-2-aminocyclohexanol.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this resolution technology translates into tangible improvements in cost structure and operational reliability. The shift from enzymatic or multi-step chemical resolutions to a direct salt formation process significantly reduces the complexity of the manufacturing workflow. By eliminating the need for expensive enzymes and the associated buffer systems, the variable cost of goods sold is drastically lowered. Moreover, the ability to use water as a primary solvent removes the safety hazards and disposal costs associated with large volumes of volatile organic compounds. This green chemistry aspect not only aligns with increasingly stringent environmental regulations but also simplifies the permitting process for manufacturing facilities. The robustness of the crystallization process ensures high throughput, allowing manufacturers to meet tight delivery schedules without compromising on quality standards.
- Cost Reduction in Manufacturing: The economic benefits of this process are driven primarily by the elimination of costly reagents and the reduction of processing steps. Traditional enzymatic methods require specialized equipment and expensive biocatalysts that often have limited shelf lives, whereas the chemical resolution described here utilizes stable, small-molecule acids that are cost-effective to procure in bulk. Additionally, the high optical purity achieved in the initial crystallization minimizes the need for resource-intensive recrystallization loops, thereby saving both time and solvent. The overall yield improvement means that less raw material is required to produce the same amount of final product, directly impacting the bottom line through reduced material consumption and waste disposal fees.
- Enhanced Supply Chain Reliability: Supply chain resilience is bolstered by the use of commodity starting materials such as cyclohexene oxide and ammonia, which are produced on a massive global scale and are not subject to the supply volatility often seen with specialty biological reagents. The resolving agent, 2-methoxyphenylacetic acid, is also chemically stable and can be stockpiled without degradation, ensuring continuity of supply even during market fluctuations. The simplicity of the process technology means that it can be easily transferred between different manufacturing sites or scaled up without requiring highly specialized operator training. This flexibility allows companies to diversify their manufacturing base, reducing the risk of production stoppages due to site-specific issues.
- Scalability and Environmental Compliance: From an environmental and scalability perspective, the process is exceptionally well-suited for tonnage production. The reliance on aqueous solvent systems significantly reduces the facility's carbon footprint by lowering the energy demand for solvent recovery and incineration. The absence of heavy metal catalysts or toxic reagents simplifies the wastewater treatment process, ensuring compliance with strict discharge limits. The crystallization-based separation is inherently scalable; the physics of crystal growth remain consistent from the laboratory bench to the industrial reactor, de-risking the scale-up phase. This predictability allows for faster time-to-market for new drug candidates that rely on this chiral intermediate, providing a competitive advantage in the fast-paced pharmaceutical sector.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this optical resolution technology. These answers are derived directly from the experimental data and process descriptions found in the patent literature, providing a reliable basis for decision-making. Understanding the nuances of solvent selection, temperature control, and resolving agent stoichiometry is crucial for successfully translating this laboratory method into a commercial reality. We encourage technical teams to review these details closely when evaluating the feasibility of this route for their specific production needs.
Q: What are the advantages of using 2-methoxyphenylacetic acid over tartaric acid for resolution?
A: Unlike dibenzoyl-L-tartaric acid, which often yields optical purities around 80% ee even after multiple crystallizations, 2-methoxyphenylacetic acid enables the production of trans-2-aminocyclohexanol with optical purity exceeding 99% ee in fewer steps, significantly reducing processing time and material loss.
Q: Is the starting material for this process readily available industrially?
A: Yes, the racemic starting material can be synthesized efficiently from cyclohexene oxide and ammonia, which are low-cost, commodity chemicals. This ensures a stable and scalable supply chain for the final chiral intermediate.
Q: Can this process be scaled for commercial production?
A: Absolutely. The process utilizes water as a preferred solvent and involves standard unit operations like crystallization and filtration, avoiding complex enzymatic steps or hazardous reagents, making it highly suitable for large-scale industrial manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Trans-2-Aminocyclohexanol Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality chiral intermediates play in the development of next-generation therapeutics. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that we can meet your volume requirements with consistency and precision. We are committed to delivering products that meet stringent purity specifications, utilizing our rigorous QC labs to verify every batch against the highest industry standards. Our expertise in chiral resolution and process optimization allows us to offer tailored solutions that maximize yield and minimize cost, making us an ideal partner for your long-term supply needs.
We invite you to engage with our technical procurement team to discuss how this advanced resolution technology can be integrated into your supply chain. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into the potential economic benefits of switching to this more efficient manufacturing route. We are ready to provide specific COA data and route feasibility assessments to support your R&D and procurement decisions, ensuring a seamless transition to a more reliable and cost-effective source of optically active trans-2-aminocyclohexanol.
