Advanced Gold-Catalyzed Cyclization for High-Purity Furan Intermediates Manufacturing
Advanced Gold-Catalyzed Cyclization for High-Purity Furan Intermediates Manufacturing
The synthesis of polysubstituted furan derivatives represents a critical challenge in modern organic chemistry, particularly for the production of high-value pharmaceutical intermediates and fine chemicals. As detailed in patent CN110437183B, a groundbreaking methodology has been developed that leverages gold catalysis to achieve efficient cyclization of alkynone compounds. This technical breakthrough addresses long-standing inefficiencies in heterocyclic synthesis, offering a pathway that combines operational simplicity with exceptional yield profiles. For R&D directors and procurement specialists seeking a reliable furan compound supplier, understanding the mechanistic superiority of this gold-mediated transformation is essential for securing a stable supply of complex building blocks.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of the furan ring from acyclic precursors has relied on harsh conditions that compromise both safety and efficiency. Early methodologies, such as the copper-catalyzed cyclization reported by Gevorgyan, necessitated heating substrates to 100°C for extended periods of up to 16 hours, as illustrated in prior art reaction schemes.
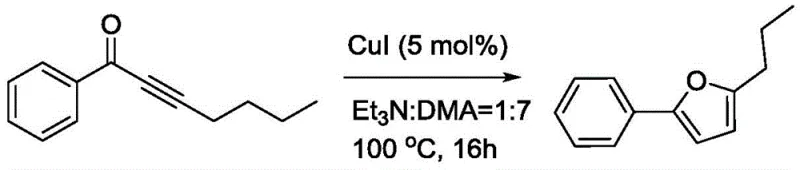
Furthermore, alternative routes involving phosphine-mediated deoxygenation of dioxanes utilize toxic reagents like carbon tetrabromide, posing significant environmental and handling hazards. Other approaches relying on strong bases like lithium diisopropylamide (LDA) require cryogenic conditions (-78°C) and exhibit moderate yields, making them unsuitable for large-scale commercial scale-up of complex pharmaceutical intermediates. These legacy processes create bottlenecks in manufacturing, driving up costs through energy consumption and waste disposal.
The Novel Approach
In stark contrast, the invention disclosed in CN110437183B introduces a streamlined gold-catalyzed protocol that operates under remarkably mild conditions. By utilizing a specialized gold catalyst in conjunction with sodium tetrakis(3,5-bis(trifluoromethyl)phenyl)borate, the reaction proceeds efficiently at temperatures ranging from 25°C to 80°C, with optimal results observed at 60°C. This shift from extreme thermal or cryogenic requirements to near-ambient conditions drastically simplifies reactor engineering and safety protocols.
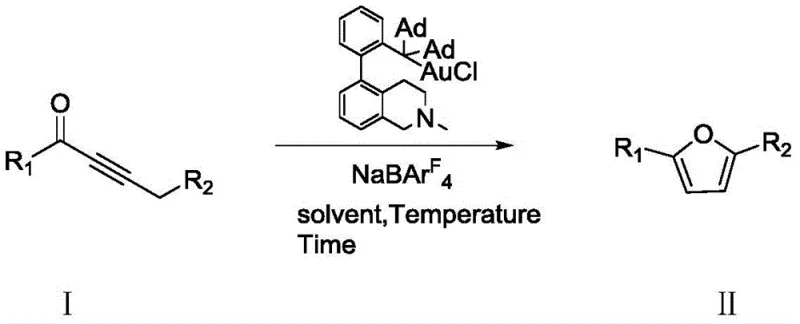
The reaction time is significantly compressed to merely 2-6 hours, representing a substantial improvement in throughput compared to the 16-hour cycles of conventional methods. This novel approach not only enhances the cost reduction in pharmaceutical intermediate manufacturing by lowering energy inputs but also ensures higher purity profiles by minimizing thermal decomposition pathways often seen in prolonged heating scenarios.
Mechanistic Insights into Gold-Catalyzed Cyclization
The core of this technological advancement lies in the unique electronic properties of the gold catalyst employed. The catalyst structure, featuring a bulky phosphine ligand system coordinated to gold chloride, facilitates the activation of the alkyne moiety within the alkynone substrate. This activation lowers the energy barrier for the intramolecular nucleophilic attack by the carbonyl oxygen, initiating the cyclization cascade.
Unlike Bronsted acid catalysis which can lead to polymerization or rearrangement side products, the soft Lewis acidity of the gold center provides precise control over the reaction trajectory. This specificity is crucial for maintaining the integrity of sensitive functional groups such as halogens or ethers present on the aromatic rings. The presence of the borate additive further stabilizes the cationic gold species in solution, ensuring consistent catalytic turnover throughout the reaction cycle without the need for excessive catalyst loading.
From an impurity control perspective, the mildness of the gold-catalyzed system is paramount. Traditional strong base or high-temperature methods often generate complex impurity profiles due to competing elimination or substitution reactions. By operating at 60°C, the kinetic energy available for these side reactions is minimized, resulting in a cleaner crude reaction mixture. This inherent selectivity reduces the burden on downstream purification units, allowing for simpler chromatographic separation and higher overall recovery of the target high-purity furan compounds.
How to Synthesize Polysubstituted Furans Efficiently
The practical implementation of this synthesis route is designed for scalability and reproducibility in a GMP environment. The process begins with the precise weighing of the alkynone initiator, the gold catalyst, and the borate activator, followed by dissolution in a solvent such as dichloroethane or toluene. The reaction is conducted under an inert nitrogen atmosphere to prevent oxidation of the catalyst or substrate.
- Prepare the reaction mixture by combining the alkynone compound (Formula I), the specialized gold catalyst, and sodium tetrakis(3,5-bis(trifluoromethyl)phenyl)borate in a suitable solvent such as dichloroethane.
- Heat the reaction mixture to a temperature between 25°C and 80°C, preferably 60°C, and maintain stirring under a nitrogen atmosphere for a duration of 2 to 6 hours.
- Upon completion, cool the reaction liquid, add column chromatography silica gel, remove the solvent via reduced pressure distillation, and purify the residue using petroleum ether elution to isolate the target furan compound.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the transition to this gold-catalyzed methodology offers tangible strategic benefits beyond mere chemical elegance. The primary advantage lies in the drastic simplification of the manufacturing workflow, which directly translates to enhanced supply chain reliability and reduced operational expenditures. By eliminating the need for cryogenic cooling or prolonged high-temperature heating, facilities can utilize standard reactor configurations without specialized retrofitting.
- Cost Reduction in Manufacturing: The economic impact of this process is driven by the elimination of expensive and hazardous reagents found in legacy routes, such as carbon tetrabromide or LDA. Furthermore, the reduced reaction time from 16 hours to as little as 2 hours significantly increases reactor turnover rates, allowing for greater production volume within the same timeframe. The low catalyst loading (2-5 mol%) combined with the absence of heavy metal scavenging steps typically required for copper or palladium processes further optimizes the cost structure.
- Enhanced Supply Chain Reliability: The raw materials for this synthesis, specifically the alkynone precursors, are readily accessible and commercially available, mitigating the risk of raw material shortages. The robustness of the reaction conditions means that batch-to-batch variability is minimized, ensuring consistent delivery schedules for downstream API manufacturers. This reliability is critical for maintaining continuous production lines in the fast-paced pharmaceutical sector.
- Scalability and Environmental Compliance: The process is inherently greener, generating no waste gas or wastewater during the reaction phase, which simplifies environmental compliance and waste treatment costs. The straightforward workup procedure involving silica gel adsorption and solvent removal is easily scalable from laboratory benchtop to multi-ton production, facilitating the commercial scale-up of complex fine chemicals without the technical risks associated with exothermic runaway reactions common in older methodologies.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation and scope of this patented synthesis method. These insights are derived directly from the experimental data and comparative analysis provided in the patent documentation, aimed at clarifying feasibility for potential manufacturing partners.
Q: What are the primary advantages of this gold-catalyzed method over traditional copper-catalyzed routes?
A: The gold-catalyzed method operates at significantly milder temperatures (25-80°C vs 100°C) and shorter reaction times (2-6 hours vs 16 hours), reducing energy consumption and minimizing thermal degradation of sensitive substrates.
Q: Is the gold catalyst reusable or does it require complex removal steps?
A: The process utilizes a homogeneous gold catalyst system that allows for straightforward workup via standard column chromatography, eliminating the need for complex heterogeneous filtration or extensive heavy metal scavenging protocols often associated with transition metal catalysis.
Q: What is the substrate scope for this cyclization reaction?
A: The method demonstrates excellent substrate adaptability, successfully cyclizing alkynones with various substituents including phenyl, halophenyl (bromo, chloro, fluoro), alkylphenyl, and methoxyphenyl groups, as well as different side chains like benzyl and phenoxy groups.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Furan Compound Supplier
At NINGBO INNO PHARMCHEM, we recognize the pivotal role that efficient heterocyclic synthesis plays in accelerating drug discovery and development. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory innovation to industrial reality is seamless. We adhere to stringent purity specifications and utilize rigorous QC labs to guarantee that every batch of furan intermediate meets the highest global standards.
We invite you to collaborate with us to leverage this advanced gold-catalyzed technology for your specific project needs. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your volume requirements. We are prepared to provide specific COA data and comprehensive route feasibility assessments to demonstrate how our manufacturing capabilities can optimize your supply chain and reduce your overall cost of goods sold.
