Advanced Tetramethyl Spiroindane Monophosphine Ligands for Industrial Asymmetric Catalysis
Advanced Tetramethyl Spiroindane Monophosphine Ligands for Industrial Asymmetric Catalysis
The landscape of asymmetric synthesis is continuously evolving, driven by the demand for more efficient and cost-effective chiral catalysts. Patent CN108659046B introduces a significant breakthrough in this domain by disclosing a novel class of monophosphine ligands based on a tetramethyl spiroindane skeleton. Unlike traditional ligands that often suffer from limited substrate applicability or complex synthesis, these new structures leverage the inherent rigidity and stability of the tetramethyl spiroindane core. This technological advancement addresses critical pain points for R&D directors and procurement managers alike, offering a pathway to high-enantioselectivity reactions with improved economic feasibility. The patent details a comprehensive library of derivatives, including phosphonites, phosphites, phosphoramidites, and phosphoric acids, all derived from a unified synthetic strategy that prioritizes scalability and raw material availability.
For organizations seeking a reliable chiral ligands supplier, the implications of this technology are profound. The core innovation lies in the utilization of 3,3,3',3'-tetramethyl-1,1'-spirodihydroindene-7,7'-dicarbaldehyde derivatives as starting materials. This approach bypasses the lengthy multi-step syntheses associated with older spiro-backbones, such as the six-step route required for racemic SPINOL. By streamlining the access to the chiral backbone, the patent enables the rapid generation of diverse ligand libraries tailored for specific metal-catalyzed transformations. Whether for asymmetric hydrogenation, coupling, or allylic alkylation, these ligands provide a versatile toolkit that enhances the efficiency of producing high-purity pharmaceutical intermediates and fine chemicals.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of chiral spiro-ligands has been hindered by inefficient routes that rely on scarce or expensive starting materials. For instance, the classical synthesis of SPINOL (1,1'-spirobiindane-7,7'-diol) reported by Birman et al. involves a cumbersome six-step sequence starting from m-methoxybenzaldehyde, followed by difficult chemical resolution to obtain optical enantiomers. This multi-step process not only accumulates impurities at each stage but also drastically reduces the overall atom economy and yield. Furthermore, the presence of active arylmethylene groups in some spiro-skeletons can lead to instability under harsh reaction conditions, limiting their utility in industrial settings. For procurement managers, these inefficiencies translate into higher costs of goods sold (COGS) and extended lead times, creating bottlenecks in the supply chain for critical catalytic components.
The Novel Approach
The methodology outlined in CN108659046B represents a paradigm shift by utilizing cheap and easily available tetramethyl spiroindane diphenols, which can be derived directly from commercial bisphenol-series products like bisphenol A in a single acid-catalyzed step. This drastic reduction in synthetic complexity allows for the preparation of the key diol intermediate (Formula II) with exceptional efficiency. The novel route eliminates the need for early-stage resolution of complex intermediates, as the chirality can be introduced or resolved at the more stable diol stage. Consequently, this approach offers substantial cost savings in ligand manufacturing by minimizing solvent usage, reducing waste generation, and shortening the production cycle. The resulting ligands exhibit enhanced rigidity and stronger steric hindrance, which are crucial for achieving high stereocontrol in asymmetric catalysis.
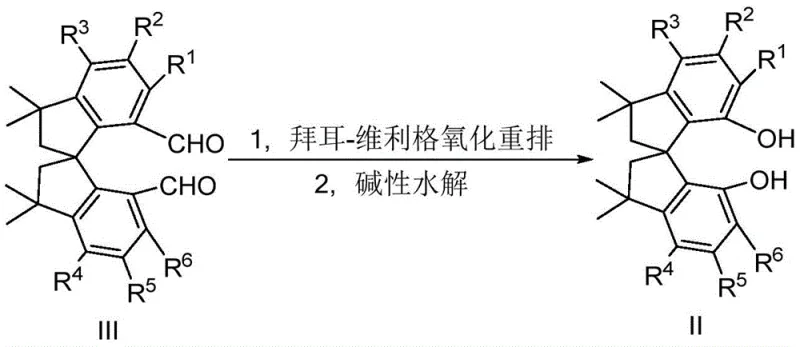
Mechanistic Insights into Baeyer-Villiger Oxidation and Phosphorylation
The heart of this synthetic strategy is the conversion of the tetramethyl spiroindane dialdehyde (Formula III) into the corresponding diol (Formula II) via a Baeyer-Villiger oxidation rearrangement followed by alkaline hydrolysis. This transformation is mechanistically fascinating as it involves the migration of an aryl group to an oxygen atom, effectively expanding the ring or modifying the oxidation state while preserving the stereochemical integrity of the spiro-center. The use of peroxy acids, such as m-chloroperoxybenzoic acid (m-CPBA), in the presence of trifluoroacetic acid ensures a controlled oxidation environment. The subsequent hydrolysis step cleaves the ester linkage to reveal the free hydroxyl groups necessary for phosphorylation. Understanding this mechanism is vital for R&D teams aiming to optimize reaction parameters, as factors like temperature (0-35°C) and acid concentration directly influence the ratio of desired product to over-oxidized byproducts.
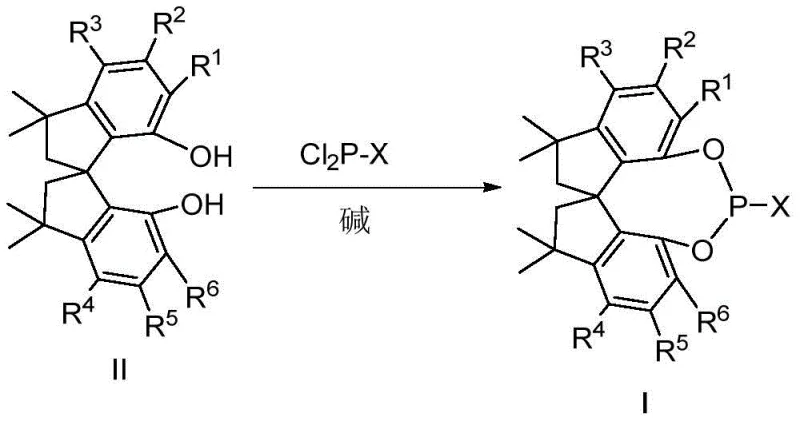
Following the formation of the diol, the phosphorylation step dictates the final ligand class. Reacting the diol with phosphorus trichloride (PCl3) or phosphorus oxychloride (POCl3) generates a reactive chlorophosphite or chlorophosphate intermediate. This species is highly electrophilic and must be handled under strict anhydrous conditions to prevent hydrolysis. The subsequent nucleophilic substitution with amines, phenols, or organometallic reagents allows for the fine-tuning of the ligand's electronic and steric properties. For example, introducing bulky aryl groups or electron-withdrawing substituents on the phosphorus atom can significantly alter the catalyst's performance in metal coordination. This modularity ensures that the ligand library can be expanded to meet the specific needs of various asymmetric reactions, from gold-catalyzed cyclizations to rhodium-catalyzed additions.
How to Synthesize Tetramethyl Spiroindane Ligands Efficiently
The synthesis of these advanced ligands follows a logical progression from commodity chemicals to high-value chiral catalysts. The process begins with the acid-catalyzed condensation of bisphenols to form the spiro-core, followed by formylation to introduce the aldehyde handles. The critical Baeyer-Villiger oxidation then converts these aldehydes into the pivotal diol intermediate. Finally, phosphorylation and functionalization yield the target ligand. This streamlined workflow minimizes unit operations and maximizes throughput, making it an ideal candidate for technology transfer. Detailed standardized synthesis steps see the guide below.
- Prepare the key tetramethyl spiroindane diol intermediate (Formula II) by subjecting the corresponding dialdehyde (Formula III) to Baeyer-Villiger oxidation using m-CPBA and trifluoroacetic acid, followed by alkaline hydrolysis.
- React the purified diol intermediate with phosphorus trichloride (PCl3) or phosphorus oxychloride (POCl3) in the presence of a base like triethylamine to generate the reactive chlorophosphite or chlorophosphate species.
- Complete the ligand synthesis by substituting the chlorine atom with the desired nucleophile (such as phenols, amines, or organolithium reagents) under inert atmosphere to yield the final monophosphine ligand (Formula I or I').
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of tetramethyl spiroindane ligands offers compelling advantages for supply chain optimization and cost management. The reliance on bisphenol A and other commodity phenols as starting materials ensures a stable and abundant supply of raw materials, mitigating the risks associated with sourcing exotic chiral pool compounds. This foundational stability allows for predictable pricing models and reduces vulnerability to market fluctuations. Furthermore, the shortened synthetic route directly correlates to reduced manufacturing lead times, enabling faster response to customer demands for custom chiral catalysts. For supply chain heads, this means enhanced reliability in securing high-purity ligands necessary for continuous pharmaceutical production lines.
- Cost Reduction in Manufacturing: The elimination of multi-step resolution processes and the use of inexpensive reagents like PCl3 and m-CPBA significantly lower the direct material costs. By avoiding the need for precious metal catalysts in the ligand synthesis itself and focusing on organic transformations with high atom economy, the overall production expense is drastically simplified. This efficiency translates into substantial cost savings for the end-user, making high-performance asymmetric catalysis more accessible for large-scale API manufacturing without compromising on quality or enantioselectivity.
- Enhanced Supply Chain Reliability: The robustness of the tetramethyl spiroindane skeleton ensures that the ligands are stable during storage and transport, reducing the risk of degradation that often plagues sensitive phosphine ligands. The synthetic route is scalable from gram to ton quantities without requiring specialized equipment beyond standard glass-lined reactors. This scalability ensures that suppliers can maintain consistent inventory levels, reducing lead time for high-purity chiral ligands and preventing production stoppages due to catalyst shortages.
- Scalability and Environmental Compliance: The process utilizes standard solvents like dichloromethane and tetrahydrofuran, which are well-understood in terms of waste management and recycling. The high yields reported in the patent examples (often exceeding 80-90% for key steps) minimize the volume of chemical waste generated per kilogram of product. This aligns with modern green chemistry principles and facilitates easier regulatory compliance for environmental discharge, a critical factor for sustainable chemical manufacturing operations.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this technology. These insights are derived directly from the experimental data and structural analysis provided in the patent documentation, ensuring accuracy and relevance for industrial application.
Q: What are the structural advantages of tetramethyl spiroindane ligands over traditional SPINOL ligands?
A: The tetramethyl spiroindane skeleton offers superior rigidity and stability compared to the traditional spirobiindane (SPINOL) backbone because it lacks active arylmethylene groups. This structural integrity leads to better dihedral angle control, enhancing enantioselectivity in catalytic reactions while utilizing cheaper, commercially available bisphenol precursors.
Q: How does the Baeyer-Villiger oxidation step impact the overall yield and purity?
A: The patent demonstrates that converting the dialdehyde precursor to the diol intermediate via Baeyer-Villiger oxidation followed by hydrolysis achieves high yields (up to 91% in lab scale). This method avoids complex resolution steps early in the synthesis, significantly simplifying impurity profiles and reducing downstream purification costs.
Q: Can these ligands be scaled for industrial pharmaceutical manufacturing?
A: Yes, the synthesis route relies on commodity chemicals like bisphenol A derivatives and standard reagents (PCl3, POCl3), making it highly amenable to scale-up. The robustness of the tetramethyl spiroindane core ensures thermal and chemical stability required for large-batch production of high-purity pharmaceutical intermediates.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Tetramethyl Spiroindane Ligands Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of the tetramethyl spiroindane ligand platform disclosed in CN108659046B. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from lab-scale discovery to industrial manufacturing is seamless. Our facility is equipped with stringent purity specifications and rigorous QC labs capable of characterizing complex chiral phosphines to the highest standards, guaranteeing the batch-to-batch consistency required for GMP environments.
We invite you to collaborate with our technical team to explore how these ligands can optimize your specific catalytic processes. By leveraging our expertise in process chemistry, we can provide a Customized Cost-Saving Analysis tailored to your project's unique requirements. We encourage you to contact our technical procurement team to request specific COA data and route feasibility assessments, allowing us to demonstrate how our supply capabilities can support your long-term strategic goals in asymmetric synthesis.
