Advanced Cu-NHC Catalyzed Synthesis of Diverse Chiral Aminoboronic Acids for Commercial API Manufacturing
Introduction to Next-Generation Chiral Aminoboronic Acid Synthesis
The landscape of medicinal chemistry has been profoundly influenced by the discovery of alpha-amino boronic acids as potent pharmacophores, particularly as serine hydrolase inhibitors. Patent CN103483363A introduces a groundbreaking methodology for the preparation of diverse chiral aminoboronic acids, addressing critical bottlenecks in the supply chain of high-value pharmaceutical intermediates. This technology leverages a novel copper-catalyzed asymmetric borylation strategy that significantly enhances both yield and stereoselectivity compared to legacy methods. For R&D directors and procurement specialists, this represents a pivotal shift towards more robust and economically viable manufacturing routes for complex drug candidates such as Bortezomib and PT-100. The ability to access a rich library of these compounds with high optical purity opens new avenues for drug discovery while simultaneously optimizing production costs for established therapies.
The core innovation lies in the utilization of specific azepine carbene precursors which, under mild reaction conditions, facilitate the efficient construction of the carbon-boron bond with exceptional stereocontrol. This approach eliminates the need for harsh reagents and multi-step derivatizations that have historically plagued the synthesis of this compound class. By streamlining the synthetic pathway, the patent offers a direct route to high-purity intermediates that are essential for the development of oncology and diabetes treatments. As a reliable pharmaceutical intermediate supplier, understanding these mechanistic advancements is crucial for ensuring the continuity and quality of the global drug supply chain.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of alpha-amino boronic acids has relied heavily on the Matteson homologation or the Ellman asymmetric addition methods, both of which present significant operational challenges for large-scale manufacturing. The Matteson method, while effective for certain alkylboronic acids, involves multiple steps including the preparation of alpha-chloroboronic esters, which limits the diversity of side chains that can be introduced and complicates the overall process flow. Furthermore, the Ellman method, although a significant improvement in stereoselectivity, relies on pre-formed N-heterocyclic carbene (NHC) copper complexes that are highly sensitive to moisture and air.
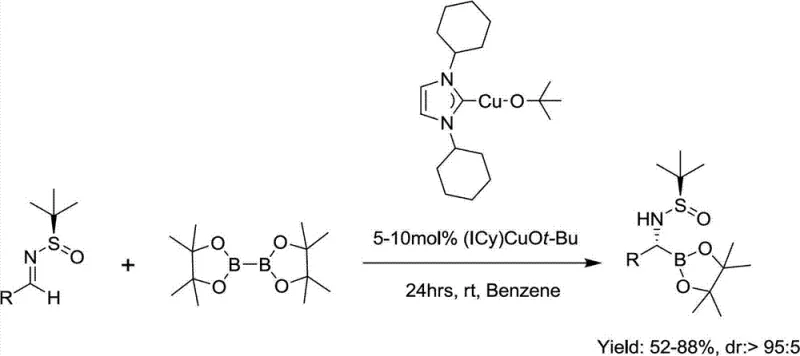
As illustrated in the prior art, the Ellman protocol typically requires the use of specialized catalysts like (ICy)CuOtBu, which must be prepared and stored under strict inert atmosphere conditions, often necessitating the use of gloveboxes. This requirement drastically increases the operational complexity and cost, making it less attractive for industrial scale-up. Additionally, the substrate scope of the Ellman method is somewhat restricted; it performs well with aliphatic imines but often yields poor results or fails entirely with aryl-substituted substrates. These limitations create a bottleneck for procurement managers seeking cost-effective and scalable sources of diverse chiral aminoboronic acid libraries.
The Novel Approach
The methodology disclosed in CN103483363A overcomes these hurdles by employing stable azepine carbene precursors that generate the active catalytic species in situ. This innovation allows the reaction to proceed under much milder and more operationally simple conditions, typically at temperatures between 15°C and 30°C, without the need for rigorous exclusion of moisture associated with pre-formed catalysts. The new approach utilizes readily available diboron reagents and tert-butyl sulfinimines, reacting them in the presence of a monovalent copper salt and the specific ligand system.
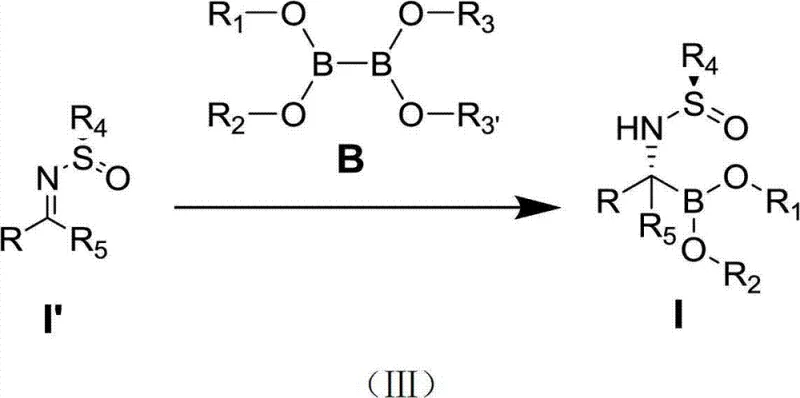
This novel reaction pathway, depicted in the scheme above, demonstrates a remarkable tolerance for a wide variety of substituents, including aromatic, heteroaromatic, and aliphatic groups, thereby vastly expanding the chemical space accessible to medicinal chemists. The process achieves yields ranging from 78% to 93.5% with an optical purity of 98%, metrics that are superior to many existing industrial standards. For supply chain heads, this translates to a more resilient manufacturing process that is less prone to failure due to environmental factors and capable of producing a broader range of intermediates from a single, unified platform.
Mechanistic Insights into Cu-NHC Catalyzed Asymmetric Borylation
The success of this synthetic route is rooted in the unique electronic and steric properties of the azepine carbene ligands employed. Unlike traditional phosphine ligands, these N-heterocyclic carbenes act as strong sigma-donors, stabilizing the copper center and facilitating the transmetallation step with the diboron reagent. The catalytic cycle likely involves the formation of a copper-boryl species which then undergoes stereoselective addition to the C=N double bond of the tert-butyl sulfinimine. The chirality of the final product is induced by the chiral auxiliary on the nitrogen atom of the imine substrate, working in concert with the steric environment provided by the ligand.
Impurity control is inherently built into this mechanism due to the high regioselectivity and stereoselectivity of the copper catalyst. The reaction minimizes the formation of racemic byproducts or over-borylated species, which are common issues in non-catalyzed or poorly controlled Lewis acid-mediated reactions. The use of specific bases such as sodium tert-butoxide or cesium carbonate further tunes the reactivity of the copper complex, ensuring that the reaction proceeds cleanly to the desired beta-amino boronate ester. This level of control is critical for meeting the stringent purity specifications required for API intermediates, reducing the burden on downstream purification processes.
How to Synthesize Chiral Aminoboronic Acid Esters Efficiently
The synthesis protocol outlined in the patent provides a clear roadmap for laboratory and pilot-scale production. The process begins with the preparation of the specific azepine carbene precursor ligand, followed by the catalytic borylation reaction in a standard organic solvent such as benzene or THF. The reaction mixture is stirred at room temperature for 24 to 48 hours, after which the product is isolated via standard aqueous workup and silica gel chromatography.
- Preparation of the Azepine Carbene Precursor (Ligand L-n') via condensation of adjacent aryl diamines with trimethyl orthoformate.
- Catalytic asymmetric borylation of tert-butyl sulfinimines using Cu(I) salts, the prepared ligand, and diboron reagents at 15-30°C.
- Purification of the resulting chiral aminoboronic acid ester via silica gel column chromatography to achieve >98% optical purity.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this technology offers substantial strategic benefits beyond mere technical performance. The shift from sensitive, pre-formed catalysts to stable precursors significantly reduces the logistical complexity and cost associated with reagent storage and handling. Eliminating the need for glovebox operations lowers the barrier to entry for contract manufacturing organizations, thereby increasing the pool of potential suppliers and enhancing supply chain reliability.
- Cost Reduction in Manufacturing: The streamlined nature of this one-pot catalytic process eliminates multiple synthetic steps required in older methods like Matteson homologation. By reducing the number of unit operations and avoiding expensive, moisture-sensitive catalysts that require special handling, the overall cost of goods sold (COGS) is significantly lowered. The high yields reported (up to 93.5%) mean less raw material waste and higher throughput per batch, directly contributing to margin improvement for high-volume API production.
- Enhanced Supply Chain Reliability: The robustness of the reaction conditions—operating effectively at ambient temperatures with stable reagents—ensures consistent batch-to-batch quality. This reliability is paramount for maintaining uninterrupted supply of critical medicines like Bortezomib. The broad substrate scope means that a single manufacturing line can be adapted to produce various analogues, providing flexibility to respond to changing market demands without extensive retooling or process re-validation.
- Scalability and Environmental Compliance: The use of copper, an abundant and relatively low-toxicity metal, aligns well with green chemistry principles compared to processes relying on precious metals like platinum or palladium. The simplified workup procedure reduces solvent consumption and waste generation. Furthermore, the ability to scale this reaction from gram to kilogram quantities without loss of efficiency makes it an ideal candidate for commercial scale-up of complex pharmaceutical intermediates.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented synthesis method. These insights are derived directly from the experimental data and claims within the patent documentation, providing a factual basis for decision-making.
Q: How does this Cu-NHC method improve upon the traditional Ellman synthesis?
A: Unlike the Ellman method which requires sensitive NHC-Cu complexes stored in gloveboxes and shows limited substrate scope, this patented process utilizes stable azepine carbene precursors that generate the active catalyst in situ, allowing for broader substrate tolerance and easier industrial handling.
Q: What represent the typical yield and optical purity for this synthesis?
A: According to patent CN103483363A, the process consistently delivers yields between 78% and 93.5% with an exceptional optical purity of 98% ee, making it highly suitable for producing high-grade pharmaceutical intermediates.
Q: Can this method be applied to the synthesis of Bortezomib precursors?
A: Yes, the patent explicitly demonstrates the synthesis of Compound 4, which serves as a key intermediate for Bortezomib, and Compound C-3, a precursor for the diabetes drug PT-100, confirming its applicability to major commercial APIs.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Chiral Aminoboronic Acid Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of the technologies described in CN103483363A for the global pharmaceutical industry. Our team of expert chemists possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of this novel Cu-NHC catalyzed route are fully realized in a GMP-compliant environment. We maintain stringent purity specifications and operate rigorous QC labs to guarantee that every batch of chiral aminoboronic acid intermediate meets the exacting standards required for oncology and diabetes drug manufacturing.
We invite you to collaborate with us to leverage this advanced synthesis platform for your next project. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are ready to provide specific COA data and route feasibility assessments to demonstrate how we can become your strategic partner in delivering high-quality, cost-effective pharmaceutical intermediates.
