Advancing Heterocyclic Drug Synthesis with Efficient Iron-Catalyzed (Z)-Azacycloalkenyl Chlorides
The landscape of organic synthesis for pharmaceutical intermediates is constantly evolving, driven by the need for greener, more cost-effective, and highly selective methodologies. A significant breakthrough in this domain is documented in patent CN116283931A, which discloses a novel preparation method for (Z)-azacycloalkenyl chloride compounds. These compounds serve as critical building blocks in the construction of diverse molecular skeletons found in natural products and bioactive molecules. The ability to efficiently construct alkenyl chloride structures with high stereoselectivity opens new avenues for the development of heterocyclic drug candidates. Unlike traditional approaches that struggle with selectivity or rely on hazardous reagents, this invention offers a robust pathway to access these valuable intermediates. The structural versatility of these compounds allows for further transformation into various C-C and C-hetero bonds, making them indispensable tools for medicinal chemists aiming to optimize lead compounds.
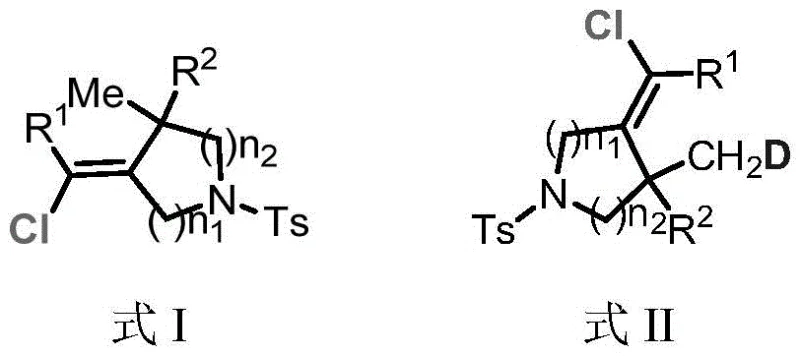
The significance of this technology extends beyond mere academic interest; it addresses practical challenges in the commercial manufacturing of fine chemicals. By enabling the selective introduction of functional groups, including deuterium isotopes, this method supports the growing demand for deuterated drugs which offer improved pharmacokinetic profiles. The patent outlines a comprehensive scope of substrates, accommodating various alkyl and aryl substituents, which ensures broad applicability across different therapeutic areas. For procurement and supply chain professionals, understanding the underlying chemistry of such innovations is key to securing reliable sources of high-purity intermediates. As the industry shifts towards more sustainable practices, technologies that minimize waste and maximize atom economy become increasingly valuable assets in a competitive market.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of alkenyl chloride compounds via hydrochlorination of triple bonds has been fraught with challenges regarding stereoselectivity and catalyst toxicity. Conventional methods often depend heavily on precious metal catalysts such as Palladium (Pd), Ruthenium (Ru), or Rhodium (Rh). While effective in certain contexts, these noble metals are not only expensive but also pose significant environmental and safety concerns due to their toxicity. Furthermore, many existing protocols require the use of directing groups to achieve asymmetry, which necessitates additional synthetic steps for installation and subsequent removal. This multi-step process increases the overall cost of goods sold (COGS) and extends the production timeline, creating bottlenecks in the supply chain. The residual metal contamination from these catalysts is another critical issue, as pharmaceutical regulations impose strict limits on heavy metal content in final drug substances, requiring costly purification and scavenging procedures that reduce overall yield.
The Novel Approach
In stark contrast, the methodology described in CN116283931A leverages earth-abundant, non-toxic iron salts as catalysts, representing a paradigm shift towards green chemistry. This approach eliminates the reliance on scarce precious metals, thereby drastically reducing raw material costs and mitigating supply chain risks associated with volatile metal markets. The reaction conditions are remarkably mild, typically operating between 20°C and 100°C, and utilize simple chlorine sources like trimethylsilyl chloride (TMSCl) or acetyl chloride. Perhaps most importantly, the process achieves high stereoselectivity, yielding the desired Z-isomer exclusively without the need for complex directing groups. This streamlining of the synthetic route not only accelerates the development timeline but also enhances the overall efficiency of the manufacturing process. The compatibility with water and even heavy water as reagents further underscores the environmental friendliness and versatility of this novel system.
Mechanistic Insights into FeCl3-Catalyzed Cyclization
The core of this innovation lies in the iron-catalyzed hydrogen functionalization cyclization of ene-yne compounds. The mechanism likely involves the activation of the alkyne moiety by the iron center, facilitating a nucleophilic attack by the chlorine source and water. This cascade reaction results in the simultaneous formation of the heterocyclic ring and the installation of the chloro-alkene functionality. The use of iron salts such as Fe(acac)3, FeCl3, or Fe(OAc)2 at loadings as low as 1 mol% demonstrates exceptional catalytic efficiency. The reaction proceeds rapidly, often completing within 10 minutes to 1 hour, which indicates a low activation energy barrier and high turnover frequency. This kinetic profile is highly advantageous for scale-up, as it allows for smaller reactor volumes and higher throughput. The precise control over the reaction parameters ensures that the thermodynamic Z-isomer is favored, providing consistent product quality batch after batch.
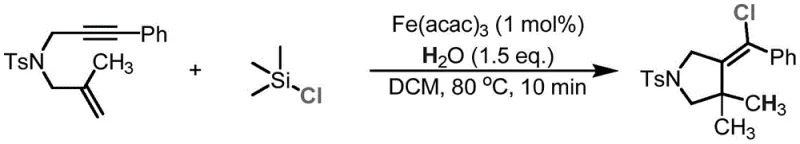
Furthermore, the mechanistic flexibility of this system allows for isotopic labeling, a feature of immense value in drug metabolism and pharmacokinetics (DMPK) studies. By simply substituting standard water with heavy water (D2O) in the reaction mixture, deuterium atoms can be site-selectively incorporated into the molecular framework. This capability is illustrated in the patent examples where deuterium-substituted heterocyclic compounds are synthesized with high yields. Such precision in isotopic labeling enables researchers to track the metabolic fate of drug candidates without altering their biological properties significantly. The ability to perform this transformation under mild conditions without protecting groups simplifies the synthesis of labeled standards, which are often required for regulatory submissions. This dual functionality—producing both chlorinated scaffolds and deuterated analogs—makes the technology a powerful tool for modern medicinal chemistry programs.
How to Synthesize (Z)-Azacycloalkenyl Chloride Efficiently
Implementing this synthesis in a laboratory or pilot plant setting requires adherence to specific operational parameters to ensure optimal yield and purity. The process begins with the preparation of the enyne substrate, which can be synthesized via standard alkylation and Sonogashira coupling methods. Once the substrate is ready, the iron-catalyzed cyclization is straightforward. The reaction mixture typically consists of the enyne compound, the iron catalyst, and a chlorine source dissolved in dichloromethane. The addition of water or D2O initiates the reaction, which is then heated to the specified temperature. Detailed standardized synthesis steps see the guide below.
- Mix the 1,6-enyne or 1,7-enyne substrate with a low loading (1 mol%) of an iron salt catalyst such as Fe(acac)3 or FeCl3 in a suitable reactor.
- Prepare a solution of the chlorine source (e.g., TMSCl, acetyl chloride) and water or heavy water (D2O) in dichloromethane and add it to the reaction mixture.
- Heat the reaction to 40°C-80°C for approximately 10 minutes to 1 hour, then purify the resulting (Z)-azacycloalkenyl chloride via column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this iron-catalyzed technology offers tangible strategic benefits that extend beyond the laboratory. The shift from precious metals to iron represents a fundamental change in the cost structure of intermediate manufacturing. Iron salts are ubiquitous and inexpensive compared to palladium or rhodium complexes, leading to a significant reduction in raw material expenditure. Moreover, the elimination of directing groups and the associated removal steps simplifies the process flow, reducing the consumption of solvents and reagents. This simplification translates directly into lower operational costs and a smaller environmental footprint, aligning with corporate sustainability goals. The robustness of the reaction conditions also implies greater reliability in supply, as the process is less sensitive to minor fluctuations in temperature or reagent quality.
- Cost Reduction in Manufacturing: The replacement of expensive noble metal catalysts with廉价 iron salts removes a major cost driver from the bill of materials. Additionally, the high catalytic efficiency means that very small amounts of catalyst are needed, further driving down costs. The simplified workup procedure, which avoids complex metal scavenging steps, reduces the consumption of auxiliary materials and labor hours. Consequently, the overall cost of production for these heterocyclic intermediates is substantially lowered, allowing for more competitive pricing in the marketplace without compromising margin.
- Enhanced Supply Chain Reliability: Relying on earth-abundant metals like iron mitigates the geopolitical and economic risks associated with the supply of precious metals. Iron is readily available globally, ensuring a stable supply chain that is less prone to disruption. The short reaction times and mild conditions also facilitate faster turnaround times from order to delivery. This agility is crucial for pharmaceutical companies that need to respond quickly to changing clinical trial demands. The ability to source key intermediates from a supplier utilizing this robust technology ensures continuity of supply for critical drug development programs.
- Scalability and Environmental Compliance: The green nature of this synthesis, characterized by the use of non-toxic catalysts and water as a reagent, simplifies regulatory compliance regarding waste disposal and emissions. Scaling up exothermic reactions can be challenging, but the mild thermal profile of this process makes it inherently safer and easier to manage on a large scale. The high selectivity reduces the formation of by-products, minimizing the burden on waste treatment facilities. This environmental compatibility not only reduces disposal costs but also enhances the corporate social responsibility profile of the manufacturing partner, a factor increasingly weighted in vendor selection processes.
Frequently Asked Questions (FAQ)
To assist technical teams in evaluating this technology for their specific applications, we have compiled answers to common inquiries regarding the process capabilities and limitations. These insights are derived directly from the experimental data and technical specifications outlined in the patent documentation. Understanding these nuances is essential for integrating this methodology into existing development pipelines.
Q: How does this iron-catalyzed method improve upon traditional palladium-catalyzed hydrochlorination?
A: Traditional methods often rely on expensive and toxic noble metals like Palladium (Pd) or Ruthenium (Ru) and require complex directing groups that must be removed later. This novel protocol utilizes inexpensive, non-toxic iron salts, eliminating the need for costly metal scavenging steps and simplifying the purification process significantly.
Q: Can this methodology be used for synthesizing deuterium-labeled drug candidates?
A: Yes, a key advantage of this technology is its compatibility with heavy water (D2O) as a reagent. By substituting standard water with D2O during the reaction, deuterium atoms can be selectively introduced into the molecular structure at specific active sites, which is crucial for metabolic stability studies and ADME profiling.
Q: What is the stereoselectivity of the resulting alkenyl chloride products?
A: The process is highly stereoselective, yielding products with a single Z-configuration. This high level of stereocontrol is vital for pharmaceutical applications where the biological activity of a drug molecule is often dependent on its specific geometric arrangement.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable (Z)-Azacycloalkenyl Chloride Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of advanced synthetic methodologies like the iron-catalyzed hydrochlorination described in CN116283931A. As a dedicated CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. Our facility is equipped to handle the specific requirements of this chemistry, ensuring that the transition from bench-scale discovery to commercial manufacturing is seamless. We maintain stringent purity specifications and operate rigorous QC labs to guarantee that every batch of (Z)-azacycloalkenyl chloride meets the highest industry standards. Our commitment to quality ensures that your drug development timeline remains on track without interruptions due to material shortages or quality issues.
We invite you to collaborate with us to leverage this cutting-edge technology for your next project. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and target specifications. By partnering with us, you gain access to not just a chemical supplier, but a strategic ally committed to optimizing your supply chain. Please contact us to request specific COA data and route feasibility assessments for your target molecules. Let us help you accelerate your path to market with efficient, cost-effective, and high-quality pharmaceutical intermediates.
