Advanced Phase Transfer Catalysis for Commercial Scale-Up of High-Purity Sulbactam Pivoxil
The pharmaceutical industry continuously seeks robust synthetic routes for beta-lactamase inhibitors, and patent CN101747343A presents a significant technological leap in the preparation of sulbactam pivoxil. This specific intellectual property outlines a novel methodology that utilizes sulbactam sodium as the starting material, reacting it with chloromethyl pivalate under the influence of a phase transfer catalyst within an organic solvent system. Unlike traditional approaches that struggle with heterogeneous reaction dynamics, this invention leverages quaternary ammonium salt cationic surfactants to bridge the solubility gap between ionic reactants and organic electrophiles. The process operates under remarkably mild thermal conditions, specifically between 25°C and 30°C, which minimizes thermal degradation risks inherent to the sensitive beta-lactam ring structure. By optimizing the molar ratio of sulbactam sodium to chloromethyl pivalate to a range of 1:1 to 1:1.5, the inventors have achieved a synthesis pathway that is not only chemically efficient but also economically viable for modern supply chains. This report analyzes the technical depth of this patent to provide R&D directors and procurement leaders with actionable insights into adopting this superior manufacturing protocol.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historical synthesis routes for sulbactam pivoxil, such as the method originally described by Wayne E. Barth, suffer from significant inefficiencies that hinder modern commercial viability. The legacy process involves dissolving dioxopenicillanic acid in N,N-dimethylformamide and reacting it with diisopropylethylamine and chloromethyl pivalate at room temperature for an extended period of 24 hours. This approach typically results in a suboptimal yield of approximately 65%, necessitating complex workup procedures involving multiple washes with water and saturated sodium chloride solution. Furthermore, subsequent literature, including reports by Blagina K. Vassileva, describes suspending sulbactam sodium salt in dimethyl sulfoxide and reacting it for 18 hours, yet the total yield after refinement remains around 68%. These conventional methods are plagued by solid-liquid phase separation issues, making the reaction endpoint difficult to control precisely. The inability to recover solvents and catalysts efficiently leads to complicated three-waste treatment protocols, driving up both environmental compliance costs and overall production expenses, thereby rendering them less attractive for large-scale industrial applications.
The Novel Approach
The innovative method disclosed in CN101747343A fundamentally alters the reaction landscape by introducing a phase transfer catalyst to create a more homogeneous reaction environment. By employing quaternary ammonium salts such as cetyltrimethylammonium chloride or bromide, the process effectively transports the sulbactam anion into the organic phase where it can readily react with chloromethyl pivalate. This strategic modification allows the reaction to proceed at a controlled temperature range of 25°C to 30°C for 18 to 24 hours, achieving yields consistently above 92% as demonstrated in the patent examples. The operational simplicity is enhanced by the ability to monitor the reaction progress using thin-layer chromatography (TLC), providing precise endpoint control that was previously unattainable. Post-reaction processing is streamlined through a crystallization step in ice water followed by sequential washing with cold water and ethanol, eliminating the need for cumbersome extraction sequences. This novel approach not only maximizes material throughput but also aligns with green chemistry principles by reducing waste generation and energy consumption associated with prolonged heating or complex purification steps.
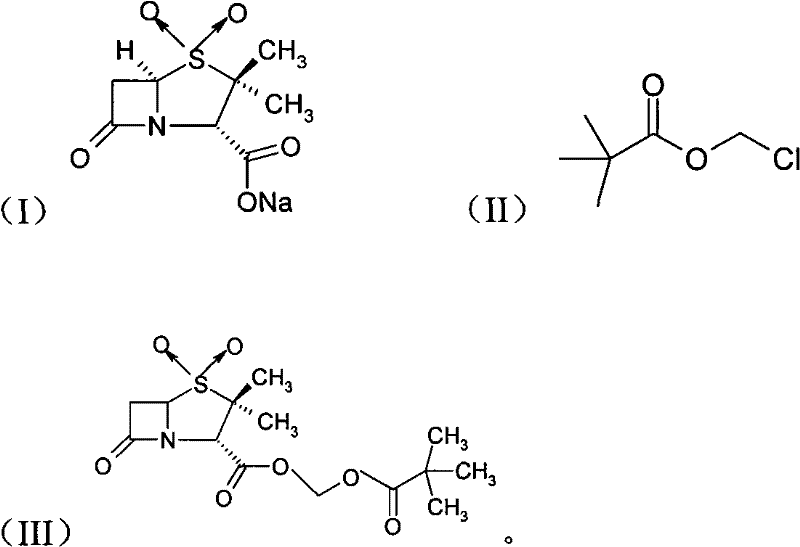
Mechanistic Insights into Phase Transfer Catalyzed Esterification
The core chemical transformation in this patent is a nucleophilic substitution reaction facilitated by phase transfer catalysis (PTC), a mechanism critical for understanding the high efficiency of this route. In the absence of a catalyst, the sulbactam sodium salt exists primarily as an ionic lattice or tightly solvated ion pair in polar aprotic solvents like DMF or DMSO, limiting its nucleophilicity towards the organic chloromethyl pivalate. The quaternary ammonium cation (Q+) acts as a phase transfer agent by exchanging with the sodium ion to form a lipophilic ion pair [Sulbactam-COO- Q+]. This lipophilic complex is highly soluble in the organic medium, effectively increasing the concentration of the reactive carboxylate anion in the vicinity of the alkylating agent. Once in the organic phase, the carboxylate anion attacks the methylene carbon of the chloromethyl pivalate, displacing the chloride ion and forming the ester linkage characteristic of sulbactam pivoxil. The chloride ion then pairs with the quaternary ammonium cation to return to the interface or aqueous micro-environment, completing the catalytic cycle. This continuous shuttling mechanism ensures that the reaction proceeds rapidly even at low temperatures, preserving the stereochemical integrity of the chiral centers within the penicillanic acid nucleus.
Impurity control in this synthesis is intrinsically linked to the mild reaction conditions and the specificity of the phase transfer mechanism. Traditional methods operating at ambient or elevated temperatures without catalytic enhancement often promote side reactions such as hydrolysis of the beta-lactam ring or elimination reactions of the chloromethyl species. By maintaining the temperature strictly between 25°C and 30°C, the thermal energy available for these degradation pathways is minimized. Additionally, the use of a specific molar excess of chloromethyl pivalate (1:1 to 1:1.5) ensures that the sulbactam sodium is fully consumed without leaving excessive unreacted starting material that could complicate downstream purification. The crystallization step in ice water further purifies the product by exploiting the differential solubility of the ester product versus the ionic starting materials and catalyst residues. The resulting product demonstrates high purity, with melting points ranging from 102.0°C to 106.0°C and specific rotation values consistent with high-quality standards, indicating minimal formation of diastereomers or degradation byproducts.
How to Synthesize Sulbactam Pivoxil Efficiently
To implement this high-yield synthesis in a laboratory or pilot plant setting, operators must adhere to strict procedural controls regarding reagent addition and temperature management. The process begins by dissolving the sulbactam sodium raw material in a measured volume of N,N-dimethylformamide or dimethyl sulfoxide, ensuring complete solvation before catalyst introduction. The phase transfer catalyst, typically cetyltrimethylammonium chloride, is added in a mass ratio of 1:0.05 to 0.10 relative to the sulbactam sodium to ensure sufficient catalytic turnover without excessive contamination. Following this, chloromethyl pivalate is added dropwise to manage the exotherm and maintain the reaction temperature within the narrow 25°C to 30°C window. Continuous monitoring via TLC is essential to determine the exact completion point, preventing over-reaction which could lead to impurity formation. Upon completion, the reaction mixture is quenched into ice water to induce crystallization, followed by a rigorous washing protocol involving cold water, 95% ethanol, and absolute ethanol to remove residual solvent and catalyst traces.
- Dissolve sulbactam sodium in an organic solvent such as DMF or DMSO and add a quaternary ammonium salt phase transfer catalyst.
- Dropwise add chloromethyl pivalate to the mixture while maintaining the temperature between 25°C and 30°C.
- Monitor the reaction progress via TLC, then pour the mixture into ice water to crystallize, followed by filtration and washing with ethanol.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this phase transfer catalyzed route offers substantial strategic advantages over legacy manufacturing methods. The most immediate impact is observed in the drastic improvement of reaction yield, which jumps from historical averages of roughly 65-68% to over 92% as validated by the patent examples. This increase in material efficiency directly translates to a significant reduction in the cost of goods sold (COGS), as less raw material is required to produce the same quantity of active pharmaceutical ingredient intermediate. Furthermore, the ability to control the reaction endpoint precisely reduces the risk of batch failures and off-spec material, enhancing overall supply chain reliability and predictability. The simplified workup procedure, which avoids complex extractions and utilizes straightforward crystallization, shortens the production cycle time and reduces the labor and utility costs associated with downstream processing. These factors combined make the supplier offering this technology a more competitive partner for long-term contracts requiring stable pricing and consistent quality.
- Cost Reduction in Manufacturing: The implementation of phase transfer catalysis eliminates the need for expensive stoichiometric bases like diisopropylethylamine used in older methods, replacing them with catalytic amounts of inexpensive quaternary ammonium salts. This shift from stoichiometric reagents to catalytic systems inherently lowers the raw material cost per kilogram of product. Additionally, the high yield means that the effective cost of the starting sulbactam sodium is amortized over a much larger output of finished product, drastically improving the economic margin. The reduction in solvent usage and the potential for solvent recovery in a closed-loop system further contribute to lowering the operational expenditure, making the final product more price-competitive in the global market.
- Enhanced Supply Chain Reliability: The robustness of this synthesis route ensures a steady and reliable supply of sulbactam pivoxil, a critical intermediate for combination antibiotic therapies. Because the reaction conditions are mild and do not require specialized high-pressure or cryogenic equipment, the barrier to entry for manufacturing is lower, allowing for diversified sourcing options. The use of common industrial solvents like DMF and DMSO ensures that raw material availability is not a bottleneck, mitigating the risk of supply disruptions. Moreover, the reproducibility of the yield across different batches, as evidenced by the consistent results in the patent examples, guarantees that procurement teams can forecast inventory needs with greater accuracy and confidence.
- Scalability and Environmental Compliance: Scaling this process from laboratory to commercial production is straightforward due to the absence of hazardous reagents and the simplicity of the unit operations involved. The mild temperature profile reduces the energy load on reactor cooling systems, facilitating easier heat management in large-scale vessels. From an environmental perspective, the reduction in waste generation—specifically the avoidance of large volumes of aqueous washes required in extraction-heavy protocols—simplifies wastewater treatment and lowers disposal costs. This alignment with environmental, social, and governance (ESG) goals makes the manufacturing process more sustainable and resilient against tightening regulatory frameworks regarding chemical waste discharge.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production of sulbactam pivoxil using this advanced phase transfer methodology. These answers are derived directly from the experimental data and claims presented in patent CN101747343A, providing a factual basis for decision-making. Understanding these details helps stakeholders evaluate the feasibility of integrating this supply source into their existing value chains. The focus is on clarity regarding yield expectations, catalyst specifications, and process safety to ensure all parties have a unified understanding of the technology's capabilities.
Q: What is the primary advantage of using a phase transfer catalyst in sulbactam pivoxil synthesis?
A: The use of a phase transfer catalyst, specifically quaternary ammonium salts like cetyltrimethylammonium chloride, significantly improves the reaction yield to over 92% compared to conventional methods which often yield below 70%. It facilitates the reaction between the ionic sulbactam sodium and the organic chloromethyl pivalate in a homogeneous phase.
Q: Can this process be scaled for industrial production?
A: Yes, the patent explicitly states the method is suitable for large-scale industrial production due to mild reaction conditions (25°C-30°C), controllable endpoints via TLC, and simplified post-treatment processes involving crystallization and filtration.
Q: What solvents are compatible with this synthesis route?
A: The process utilizes polar aprotic solvents such as N,N-dimethylformamide (DMF) or dimethyl sulfoxide (DMSO), which effectively dissolve the sulbactam sodium salt and support the phase transfer mechanism.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Sulbactam Pivoxil Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the efficacy and safety of final antibiotic formulations. Our technical team has extensively analyzed the phase transfer catalysis route described in CN101747343A and possesses the expertise to execute this synthesis with precision and consistency. We boast extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met regardless of volume. Our facilities are equipped with stringent purity specifications and rigorous QC labs that utilize advanced analytical techniques to verify every batch against the physicochemical data outlined in the patent, including melting point and HPLC content analysis. We are committed to delivering sulbactam pivoxil that meets the highest international standards for pharmaceutical intermediates.
We invite you to collaborate with us to optimize your supply chain for beta-lactamase inhibitor production. By leveraging our manufacturing capabilities, you can access a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality targets. We encourage you to contact our technical procurement team today to request specific COA data and route feasibility assessments. Let us demonstrate how our commitment to technological excellence and operational efficiency can drive value for your organization and secure your position in the competitive pharmaceutical market.
