Advanced Catalytic Synthesis of Alpha-Azyl Aryl Alkyl Ketones for Industrial Scale Manufacturing
Advanced Catalytic Synthesis of Alpha-Azyl Aryl Alkyl Ketones for Industrial Scale Manufacturing
The production of high-purity alpha-azyl aryl alkyl ketone compounds represents a critical challenge in the synthesis of advanced pharmaceutical and agrochemical intermediates. Patent CN101633647B introduces a groundbreaking methodology that addresses long-standing inefficiencies in traditional synthetic routes, specifically targeting the optimization of atom economy and reaction selectivity. This technology leverages a sophisticated four-step sequence involving Friedel-Crafts acylation, auxiliary-assisted bromination, epoxidation, and catalytic amine ring-opening. By integrating specific auxiliary compositions during the halogenation phase and employing tailored solid acid catalysts for the final substitution, the process achieves a remarkable total molar yield ranging from 70% to 88%. For R&D directors and procurement specialists seeking a reliable pharmaceutical intermediate supplier, this patent outlines a pathway that drastically reduces waste discharge while maintaining stringent quality standards essential for downstream API manufacturing.
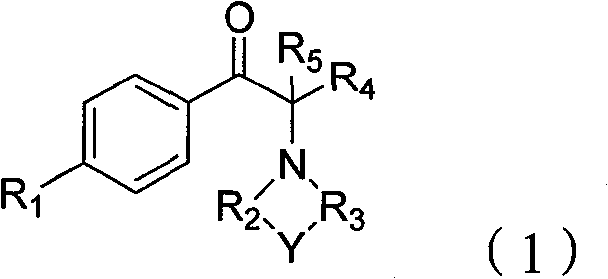
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of alpha-azyl aryl alkyl ketones has been plagued by significant inefficiencies, particularly regarding halogen utilization and reaction selectivity. Prior art, such as U.S. Pat 4582862A and U.S. Pat 4308400A, typically relies on direct bromination followed by epoxidation and ring-opening without specialized catalytic support. A major drawback of these conventional methods is the poor utilization ratio of bromine, which often remains below 50%, leading to excessive consumption of hazardous reagents and increased generation of acidic waste streams. Furthermore, the ring-opening reaction of epoxy intermediates with organic amines in traditional processes frequently suffers from low selectivity, resulting in complex impurity profiles that necessitate costly and time-consuming purification steps. These limitations not only inflate the cost reduction in API manufacturing but also pose substantial environmental compliance challenges for large-scale production facilities aiming to minimize their ecological footprint.
The Novel Approach
The innovative strategy detailed in CN101633647B fundamentally restructures the synthetic pathway to overcome these historical bottlenecks through precise chemical engineering. The core advancement lies in the introduction of a dual-component auxiliary composition during the bromination stage, consisting of an oxidant like hydrogen peroxide or peracetic acid paired with a mineral acid. This modification elevates the atom utilization of bromine to greater than 80%, effectively doubling the efficiency compared to standard protocols. Additionally, the process incorporates a specially prepared solid acid catalyst, derived from treated clays or kaolins, during the final amine substitution and ring-opening step. This catalytic intervention ensures that the reaction selectivity reaches upwards of 98%, thereby streamlining the isolation of the target molecule. The integration of these enhancements results in a robust, environmentally friendly preparation technology that is highly conducive to industrial scale-up.
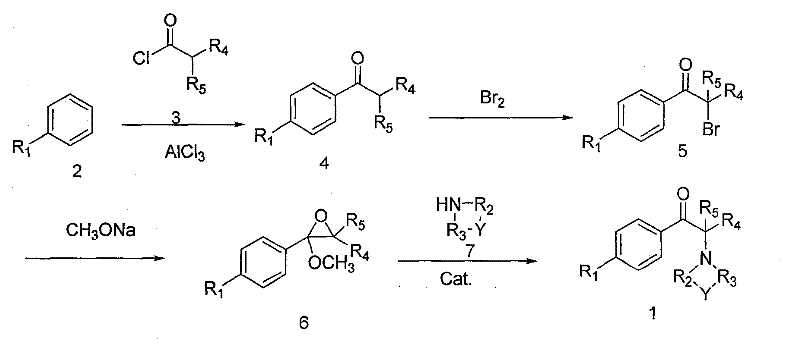
Mechanistic Insights into Auxiliary-Assisted Bromination and Catalytic Ring-Opening
The mechanistic superiority of this synthesis route is anchored in the strategic manipulation of reaction kinetics and thermodynamics at two critical junctures. During the bromination of the alpha-position of the aryl alkyl ketone, the auxiliary composition functions by generating reactive brominating species in situ while simultaneously scavenging hydrogen bromide by-products. The presence of hydrogen peroxide or peracetic acid in conjunction with acids like sulfuric or hydrochloric acid facilitates a more controlled electrophilic substitution, minimizing poly-bromination and ensuring that the stoichiometric ratio of bromine to substrate can be optimized to nearly 1:1. This precise control is vital for maintaining high purity standards, as it prevents the formation of difficult-to-remove poly-halogenated impurities that often compromise the quality of fine chemical intermediates used in sensitive biological applications.
Furthermore, the mechanism of the final ring-opening step is significantly enhanced by the surface acidity and pore structure of the activated clay catalyst. When the epoxy intermediate reacts with organic amines such as piperidine or morpholine, the solid acid catalyst activates the epoxide ring towards nucleophilic attack, lowering the activation energy required for the substitution. This catalytic effect not only accelerates the reaction rate under reflux conditions but also directs the regioselectivity of the amine attack, ensuring that the nitrogen atom attaches exclusively at the desired carbon center. The result is a dramatic improvement in product consistency, with HPLC analysis confirming content levels exceeding 99.5% and transmittance values above 95%. Such rigorous impurity control mechanisms are indispensable for manufacturers supplying high-purity OLED materials or pharmaceutical precursors where trace contaminants can invalidate entire batches.
How to Synthesize Alpha-Azyl Aryl Alkyl Ketone Efficiently
The operational protocol for this synthesis is designed for seamless translation from laboratory benchtop to multi-ton commercial reactors. The process begins with the Friedel-Crafts acylation of substituted aromatic hydrocarbons, followed by the critical auxiliary-assisted bromination which sets the stage for high-yield downstream processing. The subsequent epoxidation in methanol solvent generates the reactive oxirane intermediate, which is then subjected to the catalytic ring-opening with the chosen amine nucleophile. Each step is monitored via gas chromatography to ensure complete conversion before proceeding, guaranteeing that the cumulative yield remains within the optimal 70% to 88% range. The detailed standardized synthesis steps, including specific temperature gradients, reagent addition sequences, and workup procedures necessary for replicating this high-efficiency route, are outlined in the guide below.
- Perform Friedel-Crafts acylation of substituted aromatic hydrocarbons with fatty acid acyl chlorides using anhydrous aluminum chloride.
- Conduct bromination of the resulting ketone using an auxiliary composition of hydrogen peroxide/peracetic acid and mineral acids to improve bromine utilization.
- Execute epoxidation in methanol with sodium methoxide, followed by catalytic ring-opening with organic amines using a solid acid catalyst.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this patented synthesis method offers transformative benefits that extend far beyond simple yield improvements. The primary economic driver is the substantial reduction in raw material costs achieved through the enhanced atom economy of the bromination step. By increasing bromine utilization from roughly 50% to over 80%, the process eliminates the need for excessive reagent purchasing and significantly lowers the volume of hazardous waste requiring disposal. This efficiency gain directly translates to a lower cost of goods sold (COGS), providing a competitive pricing advantage in the global market for specialty chemicals. Moreover, the simplified purification workflow, enabled by the high selectivity of the catalytic ring-opening, reduces the consumption of solvents and energy associated with extensive recrystallization or chromatographic separation, further driving down operational expenditures.
- Cost Reduction in Manufacturing: The elimination of expensive transition metal catalysts in favor of readily available solid acid clays removes the necessity for costly heavy metal removal steps, which are often regulatory bottlenecks in pharmaceutical production. This switch to earth-abundant catalytic materials not only lowers the direct cost of catalysts but also simplifies the equipment requirements, as there is no need for specialized lined reactors to prevent corrosion from aggressive homogeneous acids. The overall process design minimizes the number of unit operations required, allowing for a more streamlined production line that maximizes throughput while minimizing labor and utility costs associated with batch processing.
- Enhanced Supply Chain Reliability: The reliance on commodity chemicals such as hydrogen peroxide, mineral acids, and common aromatic hydrocarbons ensures a stable and resilient supply chain that is less susceptible to geopolitical disruptions or raw material shortages. Unlike processes dependent on exotic ligands or scarce precious metals, this method utilizes reagents that are globally sourced and available in bulk quantities, guaranteeing consistent production schedules. The robustness of the reaction conditions, which tolerate a wide range of temperatures and pressures without compromising yield, further enhances operational reliability, reducing the risk of batch failures that could lead to delivery delays for critical downstream customers.
- Scalability and Environmental Compliance: The low discharge of three wastes (wastewater, waste gas, and solid waste) positions this technology as a leader in green chemistry, aligning perfectly with increasingly stringent environmental regulations worldwide. The ability to recover and recycle solvents like methanol and dichloroethane, along with the potential to repurpose by-product hydrochloric acid, creates a closed-loop system that minimizes environmental impact. This eco-friendly profile facilitates easier permitting for new production facilities and reduces the liability associated with waste management, making it an ideal solution for the commercial scale-up of complex pharmaceutical intermediates in regions with strict environmental oversight.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation and optimization of this synthesis route. These insights are derived directly from the experimental data and comparative examples provided in the patent documentation, offering clarity on how this method outperforms legacy technologies. Understanding these nuances is crucial for technical teams evaluating the feasibility of integrating this process into existing manufacturing portfolios to achieve superior product specifications.
Q: How does the new bromination auxiliary composition improve efficiency?
A: The auxiliary composition, comprising hydrogen peroxide or peracetic acid combined with mineral acids, increases bromine atom utilization from less than 50% in conventional methods to over 80%, significantly reducing raw material costs and waste.
Q: What is the overall yield and purity achievable with this method?
A: The patented process achieves a total molar yield of 70% to 88% calculated from the starting aromatic hydrocarbon, with an average single-step yield of 91.5% to 97%. Final product purity exceeds 99.5% by HPLC.
Q: Why is the solid acid catalyst critical for the ring-opening step?
A: The modified clay or kaolin catalyst enhances the selectivity of the amine substitution and ring-opening reaction to over 98%, preventing side reactions and simplifying downstream purification compared to uncatalyzed thermal methods.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Alpha-Azyl Aryl Alkyl Ketone Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of deploying advanced synthetic methodologies to meet the evolving demands of the global pharmaceutical and agrochemical industries. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of patents like CN101633647B are fully realized in practical, large-scale operations. We are committed to delivering products that meet stringent purity specifications through our rigorous QC labs, which utilize state-of-the-art analytical instrumentation to verify every batch against the highest industry standards. Our dedication to process excellence allows us to offer high-purity pharmaceutical intermediates that empower our clients to accelerate their own drug development timelines with confidence.
We invite you to collaborate with us to leverage this cutting-edge technology for your specific project needs. By engaging with our technical procurement team, you can request a Customized Cost-Saving Analysis that quantifies the potential economic benefits of switching to this optimized synthesis route for your supply chain. We encourage you to contact us today to obtain specific COA data and route feasibility assessments tailored to your target molecules, ensuring a partnership built on transparency, technical expertise, and mutual success in the competitive landscape of fine chemical manufacturing.
