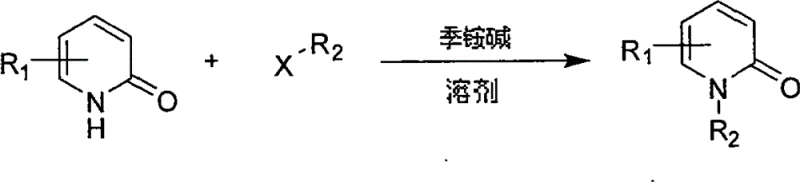
Advanced N-Alkylation of 2-Pyridone: A Scalable Route for High-Purity Pharmaceutical Intermediates
The landscape of pharmaceutical intermediate synthesis is constantly evolving, driven by the need for higher purity, better selectivity, and more sustainable processes. A pivotal advancement in this domain is detailed in patent CN101654433B, which introduces a robust method for the N-alkylation of 2-pyridone. This specific chemical transformation is critical because N-alkyl-2-pyridone scaffolds serve as fundamental building blocks for a vast array of bioactive molecules, including kinase inhibitors and antiviral agents. Historically, achieving high regioselectivity in these reactions has been a persistent challenge for process chemists. The traditional reliance on inorganic bases often leads to a mixture of N-alkylated and O-alkylated products, complicating downstream purification and reducing overall process efficiency. This new methodology addresses these pain points directly by employing quaternary ammonium hydroxides as the base of choice. By shifting the reaction paradigm, the patent demonstrates a pathway to obtain N-alkyl-2-pyridone derivatives with remarkable yield coefficients and selectivity profiles. For R&D directors and procurement managers alike, this represents a significant opportunity to optimize the supply chain for complex heterocyclic intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
In the realm of organic synthesis, the alkylation of ambident nucleophiles like 2-hydroxypyridine (which exists in tautomeric equilibrium with 2-pyridone) is notoriously difficult to control. Conventional protocols typically rely on strong inorganic bases such as potassium hydroxide (KOH) or potassium carbonate (K2CO3) to deprotonate the nitrogen or oxygen atom. However, these conditions frequently fail to discriminate effectively between the nitrogen and oxygen nucleophilic sites. As documented in prior art, including references like Bioorganic & Medicinal Chemistry Letters, these standard methods often result in the formation of 30-50% O-alkylated byproducts. This lack of selectivity is not merely a chemical curiosity; it is a major commercial bottleneck. The presence of such significant impurities necessitates rigorous and often expensive purification techniques, such as repeated recrystallization or preparative chromatography, to meet the stringent purity specifications required for Active Pharmaceutical Ingredients (APIs). Furthermore, alternative methods like the Mitsunobu reaction, while effective for inversion of stereochemistry, introduce stoichiometric amounts of phosphine oxide waste, creating substantial environmental and disposal costs that are increasingly untenable in modern green chemistry frameworks.
The Novel Approach
The innovative strategy outlined in the patent data fundamentally alters the reaction environment to favor N-alkylation exclusively. By utilizing quaternary ammonium hydroxides—specifically Tetra-n-butylammonium hydroxide (TBAH), Tetraethylammonium hydroxide (TEAH), or Tetramethylammonium hydroxide (TMAH)—the process achieves a dramatic improvement in regioselectivity. Unlike small inorganic cations, the bulky organic cations of quaternary ammonium salts appear to influence the transition state or the solvation shell of the anionic intermediate, sterically or electronically directing the alkyl halide to attack the nitrogen atom preferentially. This approach allows the reaction to proceed smoothly in common organic solvents such as tetrahydrofuran (THF), acetonitrile, or DMSO at moderate temperatures ranging from 20°C to 90°C. The result is a clean conversion where the desired N-alkylated product is formed with yields reaching approximately 90% or higher, while O-alkylation is suppressed to negligible levels. This shift from a mixed-product outcome to a highly selective transformation simplifies the entire manufacturing workflow, reducing both the time and resources required to produce high-quality intermediates.
Mechanistic Insights into Quaternary Ammonium Base Catalyzed Alkylation
To fully appreciate the value of this technology for a reliable pharmaceutical intermediate supplier, one must understand the mechanistic nuances that drive its success. The reaction begins with the deprotonation of the 2-pyridone substrate by the quaternary ammonium hydroxide. In traditional systems using metal hydroxides, the resulting anion is tightly paired with a small, hard metal cation (like K+), which can coordinate strongly with the oxygen atom, thereby enhancing its nucleophilicity and leading to O-alkylation. In contrast, the quaternary ammonium cation (R4N+) is large, soft, and diffuse. It forms a loose ion pair with the pyridone anion, effectively leaving the nitrogen atom more exposed and nucleophilic relative to the oxygen. This "naked anion" effect, often associated with phase transfer catalysis principles even in homogeneous solutions, lowers the activation energy for N-attack. Additionally, the choice of solvent plays a critical role; polar aprotic solvents like acetonitrile or THF further stabilize the transition state for N-alkylation without solvating the anion so strongly that it becomes unreactive. This delicate balance ensures that the kinetic product (N-alkyl) is formed rapidly and exclusively, preventing the thermodynamic equilibration that might otherwise lead to the O-alkyl isomer.
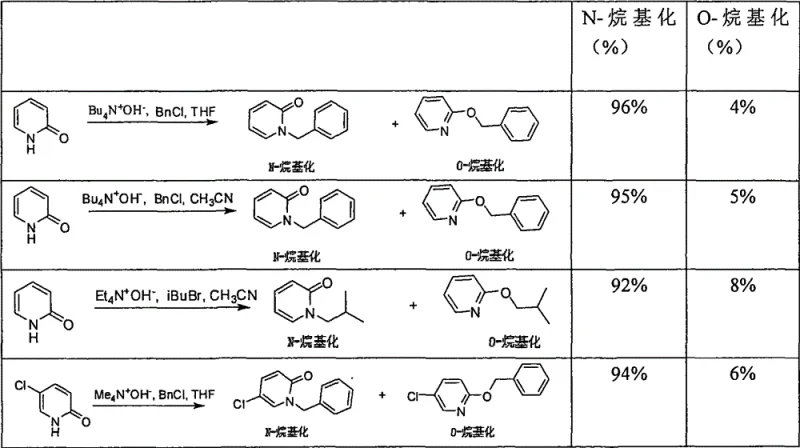
Impurity control is perhaps the most significant advantage of this mechanistic pathway. In the context of cost reduction in API manufacturing, the cost of goods is often driven less by the raw materials and more by the purification steps required to remove trace impurities. The data provided in the patent illustrates this point vividly. When comparing the new method against historical baselines, the suppression of O-alkylation is stark. Traditional methods might yield a nearly 1:1 mixture of isomers, requiring difficult separation. In this novel process, the selectivity for N-alkylation consistently exceeds 90%, and in many optimized examples, it reaches as high as 96%. This high fidelity means that the crude reaction mixture is already of high purity, often requiring only a simple aqueous workup and filtration to isolate the product. For quality control teams, this translates to a much cleaner impurity profile, reducing the risk of genotoxic impurities or difficult-to-remove structural analogs that could delay regulatory approval. The ability to predictably control the reaction outcome minimizes batch-to-batch variability, a key metric for supply chain reliability.
How to Synthesize N-Alkyl-2-Pyridone Efficiently
Implementing this synthesis route in a laboratory or pilot plant setting is straightforward and aligns well with standard operational procedures for fine chemical manufacturing. The process avoids the need for exotic reagents or extreme conditions, making it accessible for rapid scale-up. The general procedure involves dissolving the 2-pyridone substrate and the chosen alkyl halide in a solvent like THF or acetonitrile, followed by the addition of the quaternary ammonium base. The reaction is typically stirred at room temperature or slightly elevated temperatures until TLC analysis indicates the disappearance of the starting material. Post-reaction processing is equally simple, involving quenching into water and extraction with ethyl acetate. This operational simplicity is a major factor in its commercial viability. For detailed standard operating procedures and specific stoichiometric ratios tailored to your specific substrate, please refer to the technical guide below.
- Dissolve 2-pyridone and the corresponding alkyl halide in a suitable solvent such as THF or acetonitrile.
- Add a quaternary ammonium hydroxide base (e.g., TBAH, TEAH, or TMAH) to the reaction mixture at room temperature.
- Stir the reaction until completion, then perform an aqueous workup involving extraction with ethyl acetate and purification via column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this technology offers tangible strategic benefits beyond mere chemical elegance. The primary driver is the drastic simplification of the manufacturing process, which directly correlates to lower operational expenditures. By eliminating the need for complex purification trains designed to separate N- and O-isomers, manufacturers can significantly reduce solvent consumption, energy usage, and labor hours. This efficiency gain is compounded by the high yields reported in the patent examples, which often exceed 90%. Higher yields mean less raw material waste and a smaller environmental footprint, aligning with modern sustainability goals. Furthermore, the reagents involved—2-hydroxypyridines, alkyl halides, and quaternary ammonium hydroxides—are commodity chemicals with robust global supply chains. This ensures that production is not held hostage by the scarcity of specialized catalysts or ligands, thereby enhancing supply security and reducing lead times for high-purity pharmaceutical intermediates.
- Cost Reduction in Manufacturing: The economic impact of this method is profound due to the elimination of expensive and wasteful purification steps. In traditional routes, separating O-alkylated byproducts often requires preparative HPLC or multiple recrystallizations, which are cost-prohibitive at scale. By achieving >90% selectivity inherently, this process bypasses those costs entirely. Additionally, the avoidance of stoichiometric coupling reagents (like those in Mitsunobu reactions) removes a major cost center. The use of simple extraction workups instead of complex chromatographic separations further drives down the cost per kilogram, allowing for substantial margin improvements in the final API cost structure.
- Enhanced Supply Chain Reliability: Supply chain resilience is built on the availability of raw materials and the robustness of the process. This synthesis relies on widely available alkyl halides and standard quaternary ammonium bases, which are produced in massive quantities globally. There is no dependency on rare earth metals or proprietary ligands that might face geopolitical supply constraints. Moreover, the reaction conditions are mild (20-90°C) and operate at ambient pressure, reducing the engineering complexity of the reactor setup. This makes the process easier to transfer between different manufacturing sites or contract manufacturing organizations (CMOs), ensuring continuity of supply even if one facility faces disruptions.
- Scalability and Environmental Compliance: Scaling chemical processes often reveals hidden hazards or inefficiencies, but this method is inherently scalable. The exotherm is manageable, and the reagents are stable. From an environmental perspective, the reduction in waste is significant. By avoiding the generation of phosphine oxides (from Mitsunobu) or large volumes of salt waste (from inorganic bases), the E-factor (mass of waste per mass of product) is improved. This facilitates easier compliance with increasingly strict environmental regulations regarding solvent discharge and hazardous waste disposal, smoothing the path for regulatory approval and long-term commercial operation.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this N-alkylation technology. These insights are derived directly from the experimental data and technical specifications provided in the patent literature. Understanding these details is crucial for evaluating the feasibility of integrating this route into your existing production portfolio. Whether you are concerned about specific substrate compatibility or the handling of quaternary ammonium salts, the answers below provide a foundational understanding of the process capabilities.
Q: Why is traditional alkylation of 2-pyridone problematic for industrial scale-up?
A: Traditional methods using inorganic bases like KOH or K2CO3 often suffer from poor regioselectivity, producing significant amounts of O-alkylated byproducts (30-50%). This necessitates difficult and costly purification steps to isolate the desired N-alkylated product.
Q: What specific catalysts are used in this novel N-alkylation process?
A: The process utilizes quaternary ammonium hydroxides such as Tetra-n-butylammonium hydroxide (TBAH), Tetraethylammonium hydroxide (TEAH), or Tetramethylammonium hydroxide (TMAH). These bases provide superior selectivity for N-alkylation over O-alkylation.
Q: What are the primary advantages of this method for API manufacturing?
A: The method offers high yield (up to 95%) and exceptional selectivity (>90% N-alkylation). It operates under mild conditions (20-90°C) and simplifies post-reaction processing, making it highly suitable for cost-effective commercial production.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable N-Alkyl-2-Pyridone Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition from a patented laboratory method to a commercial reality requires deep technical expertise and robust infrastructure. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the high selectivity observed in the lab is maintained at the tonnage scale. Our facilities are equipped with rigorous QC labs capable of detecting trace impurities, guaranteeing that every batch of N-alkyl-2-pyridone meets stringent purity specifications required by global regulatory bodies. We understand that consistency is key for our partners, and our process engineering team is dedicated to optimizing every step of this quaternary ammonium base catalyzed route to maximize efficiency and minimize cost.
We invite you to collaborate with us to leverage this advanced synthesis technology for your next project. By partnering with us, you gain access to a Customized Cost-Saving Analysis that quantifies the potential economic benefits of switching to this high-selectivity route for your specific molecule. We encourage you to contact our technical procurement team to request specific COA data for similar intermediates and to discuss route feasibility assessments tailored to your timeline. Let us help you secure a competitive advantage through superior chemistry and reliable supply.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
