Advanced Manufacturing of 1,3-Benzodioxole Heterocycles for High-Purity PDE4 Inhibitor Intermediates
Advanced Manufacturing of 1,3-Benzodioxole Heterocycles for High-Purity PDE4 Inhibitor Intermediates
The pharmaceutical industry continuously seeks robust synthetic routes for complex heterocyclic scaffolds, particularly those serving as critical intermediates for Phosphodiesterase 4 (PDE4) inhibitors. Patent CN108368126B discloses a groundbreaking methodology for the preparation of 1,3-benzodioxole heterocyclic compounds, addressing significant bottlenecks found in earlier generations of synthesis. This technology represents a paradigm shift from laboratory-scale curiosity to industrial viability, offering a reliable pathway for producing high-purity API intermediates. The core innovation lies in the strategic manipulation of spiro-cyclic structures and the efficient introduction of difluoromethoxy groups without relying on cumbersome purification techniques. For R&D directors and procurement specialists, understanding this process is vital as it directly impacts the cost of goods sold (COGS) and supply chain stability for anti-inflammatory drug candidates.
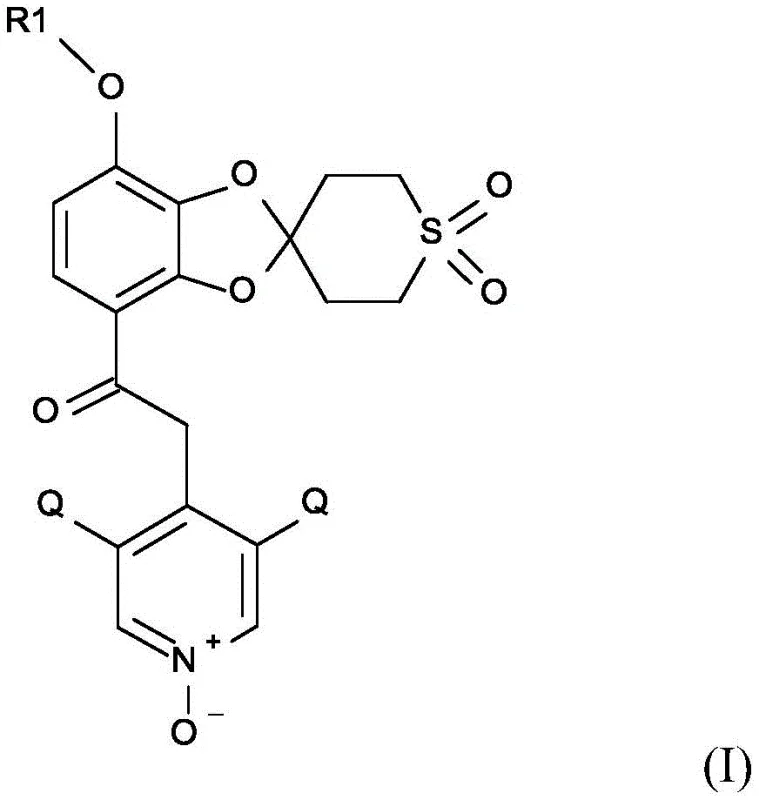
The structural complexity of the target molecule, characterized by a spiro-fused tetrahydrothiopyran ring and a substituted pyridine N-oxide moiety, presents inherent challenges in regioselectivity and functional group tolerance. Traditional approaches often struggled with low overall yields and the necessity for chromatographic separation, which is a prohibitive cost driver at the metric-ton scale. By leveraging specific acid catalysts and optimized reaction conditions, this patented process achieves superior conversion rates. The ability to produce these compounds with enhanced purity profiles ensures that downstream drug development phases proceed without the delays typically associated with impurity management. This technological advancement positions manufacturers to meet the rigorous quality standards demanded by global regulatory bodies while maintaining economic efficiency.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Prior art methods, such as those disclosed in WO 2011/160632, relied heavily on synthetic sequences that were inefficient and difficult to scale. A primary deficiency was the reliance on chromatography for product purification, a technique that is economically unfeasible for large-scale commercial production due to high solvent consumption and low throughput. Furthermore, the chemical yields in critical steps were suboptimal; for instance, the deprotection of phenolic groups in older processes yielded only around 32%, resulting in significant material loss and increased waste generation. The difluoromethylation steps in conventional routes also suffered from poor efficiency, with yields hovering near 52%, necessitating extensive recycling of starting materials or acceptance of high production costs. These limitations created a fragile supply chain vulnerable to raw material price fluctuations and processing delays.
The Novel Approach
In stark contrast, the process described in CN108368126B introduces a series of optimizations that drastically enhance process economics and scalability. The novel approach replaces chromatographic purification with simple unit operations such as crystallization, precipitation, and filtration, which are standard in modern chemical plants. Surprisingly, the yield in the phenolic deprotection step (Step 2a) exceeds 80%, more than doubling the efficiency of previous methods. Similarly, the introduction of difluoromethyl groups now achieves yields of ≥76%, ensuring a much higher throughput of valuable intermediates. This shift not only reduces the environmental footprint by minimizing solvent waste but also stabilizes the production timeline, making it a preferred choice for cost reduction in pharmaceutical manufacturing. The use of readily available silicate mineral catalysts further underscores the practical advantages of this new methodology.
Mechanistic Insights into Silicate-Catalyzed Spiro-Cyclization and Difluoromethylation
The heart of this synthetic strategy involves a sophisticated acid-catalyzed cyclization to form the spiro-[1,3-benzodioxole-2,4'-tetrahydrothiopyran] core. The reaction utilizes silicate minerals like Montmorillonite K10 or specific zeolites (e.g., HSZ-360 HUA) to facilitate the condensation between a dihydroxy-methoxyphenyl ketone and tetrahydrothiopyran-4-one. This heterogeneous catalysis offers distinct advantages over homogeneous acids, including easier separation and reusability. The mechanism likely involves the activation of the carbonyl group of the thiopyranone by the acidic sites on the clay surface, promoting nucleophilic attack by the phenolic hydroxyls. Following cyclization, the process employs a unique deprotection strategy using aromatic or aliphatic thiols in the presence of a base. This step cleaves the methyl ether protecting group to reveal the free phenol, which is then converted into a tetrabutylammonium salt to enhance its nucleophilicity for the subsequent alkylation.
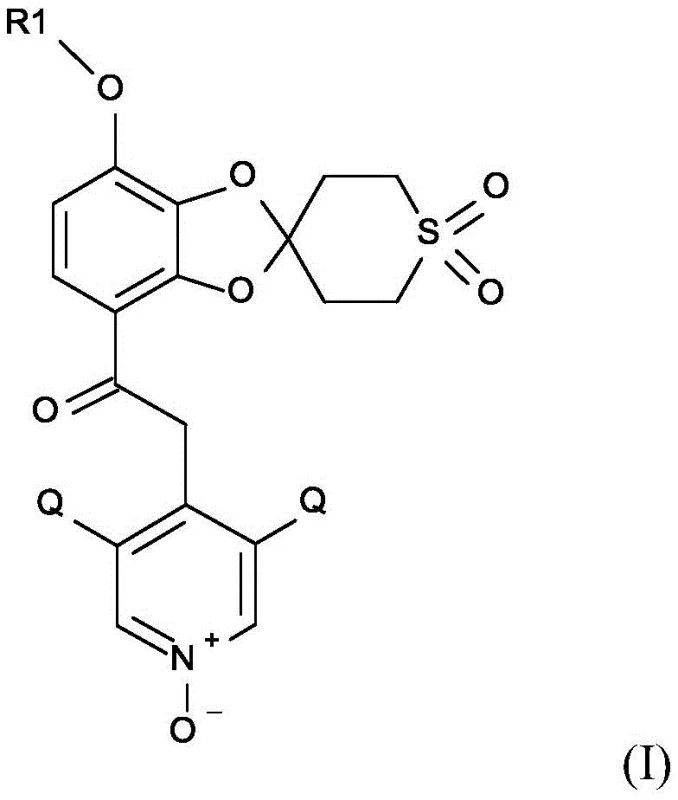
The introduction of the difluoromethoxy moiety is achieved through the alkylation of the phenoxide salt with chlorodifluoromethane gas in a polar aprotic solvent. This gas-phase alkylation is highly efficient and avoids the use of liquid alkylating agents that might introduce additional impurities. Alternatively, the process allows for the use of difluorocarbene sources like sodium chlorodifluoroacetate, providing flexibility depending on plant infrastructure. The final stages involve coupling with a chloropyridine derivative followed by oxidation. The oxidation step, converting the pyridine nitrogen to an N-oxide, is carefully controlled using peracetic acid to prevent over-oxidation of the sulfur atom in the spiro ring. This precise control of chemoselectivity is crucial for maintaining the integrity of the complex molecular architecture and ensuring the final product meets stringent purity specifications required for clinical applications.
How to Synthesize 1,3-Benzodioxole Heterocycles Efficiently
The synthesis of these high-value intermediates requires precise control over reaction parameters to maximize yield and minimize impurity formation. The process begins with the formation of the spiro-intermediate under reflux conditions in toluene, followed by a workup involving aqueous treatment and crystallization. Subsequent steps involve careful temperature management during the deprotection and alkylation phases to ensure safety and efficiency. The detailed standardized synthesis steps, including specific molar ratios, temperature ranges, and workup procedures, are outlined in the guide below to assist technical teams in replicating this robust methodology.
- React dihydroxy-methoxyphenyl ethanone with tetrahydrothiopyran-4-one using a silicate mineral catalyst like Montmorillonite K10 to form the spiro-intermediate.
- Deprotect the phenolic group using aromatic or aliphatic thiols in the presence of a base, followed by conversion to a tetrabutylammonium salt.
- Alkylate the phenoxide salt with chlorodifluoromethane to introduce the difluoromethoxy group, then couple with a chloropyridine derivative.
- Perform final oxidation using peracetic acid to generate the pyridine N-oxide functionality, followed by crystallization for purification.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this novel synthesis route offers transformative benefits that extend beyond simple chemical yield improvements. The elimination of chromatographic purification is perhaps the most significant economic driver, as it removes a major bottleneck in production capacity and drastically reduces solvent procurement costs. By shifting to crystallization-based purification, manufacturers can utilize standard stainless steel reactors and filtration equipment, thereby lowering capital expenditure requirements for new production lines. This transition also simplifies the regulatory filing process, as crystalline forms are generally more stable and easier to characterize than amorphous materials obtained via chromatography. Consequently, the time-to-market for drug candidates utilizing these intermediates can be accelerated, providing a competitive edge in the fast-paced pharmaceutical landscape.
- Cost Reduction in Manufacturing: The process achieves substantial cost savings by utilizing inexpensive silicate mineral catalysts instead of precious metal complexes or harsh liquid acids. The dramatic increase in chemical yield—from roughly 30-50% in old methods to over 80% in key steps—means that less raw material is required to produce the same amount of active intermediate. This efficiency gain directly lowers the variable cost per kilogram. Furthermore, the ability to recycle unconverted starting materials effectively minimizes waste disposal costs, contributing to a leaner and more profitable manufacturing operation without compromising on quality.
- Enhanced Supply Chain Reliability: The reliance on commodity chemicals such as montmorillonite clay, toluene, and common bases like potassium carbonate ensures a stable and resilient supply chain. Unlike processes dependent on specialized reagents with long lead times, this methodology leverages widely available industrial inputs. The robustness of the reaction conditions, which tolerate slight variations in temperature and stoichiometry, reduces the risk of batch failures. This predictability allows supply chain planners to maintain lower safety stock levels while ensuring continuous availability of critical intermediates, thereby mitigating the risk of production stoppages due to material shortages.
- Scalability and Environmental Compliance: The design of this process inherently supports commercial scale-up, moving seamlessly from kilogram to multi-ton production. The replacement of hazardous solvents and the reduction of total solvent volume through efficient crystallization align with green chemistry principles. This results in a significantly reduced environmental footprint, easing the burden on waste treatment facilities and ensuring compliance with increasingly strict environmental regulations. The simplified downstream processing also reduces energy consumption associated with solvent recovery, making the entire production lifecycle more sustainable and socially responsible.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These insights are derived directly from the experimental data and claims within the patent documentation, providing clarity on the feasibility and advantages of the method. Understanding these details is essential for technical teams evaluating the integration of this process into their existing manufacturing portfolios.
Q: How does this new process improve yield compared to prior art?
A: The novel process achieves yields of ≥80% in the deprotection step and ≥76% in difluoromethylation, significantly surpassing the 32% and 52% yields reported in previous methods like WO 2011/160632.
Q: Does this synthesis require chromatographic purification?
A: No, a major advantage of this invention is that it eliminates the need for chromatography, allowing for purification via simple unit operations like crystallization and filtration which are easily scalable.
Q: What catalysts are used for the spiro-cyclization step?
A: The process utilizes inexpensive silicate minerals such as Montmorillonite K10 or zeolites (e.g., HSZ-360 HUA) as acid catalysts, avoiding expensive transition metals.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 1,3-Benzodioxole Heterocycles Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the successful development of PDE4 inhibitors. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project needs are met with precision and reliability. We are committed to delivering products with stringent purity specifications, supported by our rigorous QC labs that employ advanced analytical techniques to verify every batch. Our facility is equipped to handle the specific requirements of spiro-cyclic chemistry and difluoromethylation, guaranteeing a consistent supply of materials that meet global regulatory standards.
We invite you to collaborate with us to leverage this advanced synthesis technology for your next drug development program. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are ready to provide specific COA data and comprehensive route feasibility assessments to demonstrate how our manufacturing capabilities can optimize your supply chain and accelerate your path to market.
