Breakthrough One-Step Synthesis of 4-Hydroxy-1,8-Naphthalimide Derivatives for Commercial Scale-Up
Breakthrough One-Step Synthesis of 4-Hydroxy-1,8-Naphthalimide Derivatives for Commercial Scale-Up
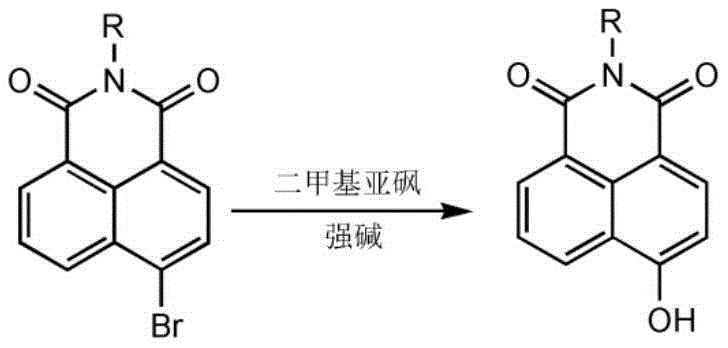
The landscape of fine chemical manufacturing for fluorescent probe intermediates is undergoing a significant transformation driven by the innovations disclosed in patent CN110183380B. This pivotal intellectual property introduces a novel, streamlined methodology for synthesizing 4-hydroxy-1,8-naphthalimide derivatives, which serve as critical building blocks in the development of advanced optical materials and biological sensors. Historically, the production of these specialized compounds has been hindered by multi-step processes involving hazardous reagents and严苛 reaction conditions that pose substantial risks to both operational safety and environmental compliance. The technical breakthrough detailed in this patent offers a robust alternative, utilizing a direct nucleophilic substitution strategy that bypasses traditional limitations. By leveraging dimethyl sulfoxide (DMSO) as a reaction medium and employing common strong bases like sodium hydroxide or potassium hydroxide, the process achieves high efficiency while drastically simplifying the workflow. For R&D directors and procurement specialists alike, this represents a paradigm shift towards more sustainable and economically viable manufacturing protocols for high-purity organic intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Prior to the advancements described in CN110183380B, the industry standard for preparing 4-hydroxy-1,8-naphthalimide derivatives relied heavily on a cumbersome two-step synthetic sequence that was fraught with inefficiencies and safety hazards. The conventional pathway typically commenced with the methoxylation of the 4-bromo precursor using sodium methoxide, followed by a harsh demethylation step requiring concentrated hydroiodic acid (HI) or hydrobromic acid (HBr) under reflux conditions. This legacy approach not only extended production timelines due to the necessity of isolating intermediate methoxy-compounds but also introduced severe safety liabilities associated with handling large volumes of corrosive mineral acids. Furthermore, the demethylation step invariably generated toxic volatile byproducts such as methyl iodide or methyl bromide, necessitating complex scrubbing systems and rigorous waste treatment protocols to meet environmental regulations. The cumulative effect of these factors was a process characterized by high operational costs, significant energy consumption for heating and refluxing, and challenging purification requirements to remove acidic residues and side products from the final API intermediate.
The Novel Approach
In stark contrast to the convoluted traditional pathways, the novel approach disclosed in the patent achieves the transformation in a single, elegant step that directly converts 4-bromo-1,8-naphthalimide derivatives into their corresponding 4-hydroxy analogues. This method capitalizes on the unique electronic properties of the naphthalimide scaffold, allowing for a direct nucleophilic aromatic substitution that was previously thought to require extreme conditions for simple aryl halides. By conducting the reaction in DMSO with sodium hydroxide or potassium hydroxide at mild temperatures ranging from 20°C to 55°C, the process eliminates the need for toxic methylating agents and corrosive acids entirely. The simplicity of this one-pot reaction significantly reduces the number of unit operations, thereby minimizing material handling and potential points of failure in the production line. Additionally, the use of DMSO facilitates an easy work-up procedure where the solid base can be simply filtered off due to its insolubility, streamlining the isolation of the crude product and setting the stage for efficient downstream purification.
Mechanistic Insights into Direct Nucleophilic Aromatic Substitution
From a mechanistic perspective, the success of this synthesis is particularly noteworthy because aryl halides are generally resistant to nucleophilic attack by hydroxide ions under mild conditions due to the stability of the carbon-halogen bond and the electron-rich nature of the aromatic ring. Typically, hydrolyzing a bromobenzene derivative would demand extreme temperatures around 300°C and pressures exceeding 28 MPa to force the reaction, often resulting in poor selectivity and decomposition. However, the 1,8-naphthalimide core possesses two powerful electron-withdrawing carbonyl groups adjacent to the nitrogen atom, which significantly deactivate the aromatic system towards electrophilic attack but conversely activate it towards nucleophilic substitution. This electronic activation lowers the energy barrier for the addition-elimination mechanism, allowing the hydroxide ion to attack the C-4 position bearing the bromine atom effectively at temperatures as low as 20°C. The solvent DMSO plays a crucial dual role by stabilizing the transition state and solvating the cation of the base while leaving the hydroxide anion relatively "naked" and highly reactive, further enhancing the kinetics of this otherwise difficult transformation.
Controlling impurities in this reaction is inherently managed by the specificity of the nucleophilic attack and the physical properties of the reagents involved. Since the reaction proceeds via a defined addition-elimination pathway on the activated ring, side reactions such as polymerization or random ring substitution are minimized compared to radical-based or high-energy thermal processes. A key advantage highlighted in the patent data is the insolubility of the excess strong base (NaOH or KOH) in the DMSO solvent post-reaction, which allows for a straightforward physical separation via filtration. This prevents the carryover of inorganic salts into the organic phase, which is a common source of contamination in aqueous work-ups. Furthermore, the subsequent purification via silica gel column chromatography using a dichloromethane and acetic acid mixture ensures the removal of any trace unreacted starting material or minor hydrolysis byproducts, yielding a final product with the stringent purity specifications required for fluorescence applications. This robust impurity profile makes the process highly attractive for scaling up, as it reduces the burden on analytical quality control and ensures batch-to-batch consistency.
How to Synthesize 4-Hydroxy-1,8-Naphthalimide Efficiently
Implementing this synthesis route in a laboratory or pilot plant setting requires careful attention to stoichiometry and reaction monitoring to maximize the reported yields of 52% to 55%. The process begins by charging a reactor with the 4-bromo-1,8-naphthalimide substrate and a strong base, specifically maintaining a mass ratio between 1:2 and 1:3 to ensure complete conversion without excessive waste. The mixture is then heated gently, typically in a water bath maintained between 20°C and 55°C, and stirred for a duration of 10 to 50 hours depending on the specific R-group substituent. Following the reaction period, the mixture is allowed to stand for 5 to 10 hours to facilitate the settling of the solid base, which is then removed by filtration. The filtrate undergoes vacuum distillation at 55°C to 65°C to remove the DMSO solvent, yielding a crude solid that is subsequently dissolved in dichloromethane with added acetic acid for final purification. Detailed standardized operating procedures for each of these critical stages are outlined below to ensure reproducibility and safety.
- Dissolve 4-bromo-1,8-naphthalimide derivative in DMSO and add strong base (NaOH/KOH) at a mass ratio of 1: 2 to 1:3.
- Stir the mixture at mild temperatures between 20°C and 55°C for 10 to 50 hours to complete the nucleophilic substitution.
- Filter off the solid base, remove solvent via vacuum distillation, and purify the crude product using silica gel column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the adoption of this patented synthesis method offers profound strategic advantages that extend far beyond simple chemical curiosity. The elimination of the two-step methylation-demethylation sequence fundamentally alters the cost structure of producing these valuable intermediates by removing entire categories of expensive and regulated raw materials. By avoiding the use of concentrated hydroiodic acid and sodium methoxide, manufacturers can significantly reduce their expenditure on hazardous chemical inventory and the associated costs of storage, handling, and disposal. Moreover, the simplification of the process flow from two distinct reaction stages to a single pot operation drastically reduces the overall cycle time, allowing for faster throughput and improved responsiveness to market demand fluctuations. This agility is crucial in the fast-paced fine chemicals sector, where lead times can often be a deciding factor in supplier selection.
- Cost Reduction in Manufacturing: The economic benefits of this route are driven primarily by the removal of costly reagents and the reduction of energy-intensive unit operations. Traditional methods require reflux conditions which consume substantial amounts of energy for heating and cooling, whereas this new process operates at near-ambient temperatures, leading to a drastic reduction in utility costs. Additionally, the avoidance of toxic methyl halide byproducts eliminates the need for specialized scrubbing equipment and expensive waste treatment protocols, further lowering the overhead associated with environmental compliance. The ability to filter off the base directly rather than performing complex aqueous extractions also reduces solvent consumption and wastewater generation, contributing to a leaner and more cost-effective manufacturing model.
- Enhanced Supply Chain Reliability: Supply chain resilience is significantly bolstered by the use of commodity chemicals such as sodium hydroxide and DMSO, which are readily available globally and less prone to supply disruptions compared to specialized reagents like concentrated hydroiodic acid. The robustness of the reaction conditions, which tolerate a wide temperature range and do not require high-pressure equipment, means that production can be easily transferred between different manufacturing sites without extensive requalification. This flexibility ensures continuity of supply even in the face of regional logistical challenges or equipment maintenance issues. Furthermore, the simplified purification process reduces the risk of batch failures due to purification bottlenecks, ensuring a more consistent and reliable delivery schedule for downstream customers.
- Scalability and Environmental Compliance: Scaling this process from gram-scale laboratory synthesis to multi-ton commercial production is facilitated by the absence of hazardous gas evolution and extreme pressure requirements. The reaction can be safely conducted in standard glass-lined or stainless steel reactors without the need for exotic metallurgy to resist corrosive acids, lowering the capital expenditure required for plant expansion. From an environmental standpoint, the process aligns perfectly with green chemistry principles by reducing the E-factor (mass of waste per mass of product) through the elimination of toxic byproducts and the recycling of the DMSO solvent. This strong environmental profile not only mitigates regulatory risk but also enhances the brand value of the final product for eco-conscious pharmaceutical and agrochemical clients.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology, derived directly from the experimental data and claims within patent CN110183380B. Understanding these nuances is essential for technical teams evaluating the feasibility of integrating this route into their existing manufacturing portfolios. The answers provided reflect the specific conditions and outcomes observed in the patent examples, offering a realistic baseline for project planning.
Q: Why is the direct hydrolysis of 4-bromo-1,8-naphthalimide considered a technical breakthrough?
A: Typically, aryl halides require extreme conditions (300°C, 28MPa) for hydrolysis. This patent demonstrates that the electron-withdrawing imide ring activates the position for substitution under mild conditions (20-55°C) in DMSO.
Q: How does this method improve safety compared to traditional demethylation routes?
A: Conventional methods use concentrated hydroiodic or hydrobromic acid, generating toxic methyl iodide or bromide gases. This new route eliminates these hazardous reagents and byproducts entirely.
Q: What is the expected yield for this synthesis method?
A: Experimental data in the patent indicates yields ranging from 52% to 55% for various alkyl-substituted derivatives, achieved through a simplified one-step process.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 4-Hydroxy-1,8-Naphthalimide Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of the synthesis method described in CN110183380B for the production of high-value fluorescent intermediates. As a premier CDMO partner, we possess the extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of this one-step process are fully realized in a GMP-compliant environment. Our facilities are equipped with state-of-the-art reactors capable of handling the specific solvent and temperature profiles required for this nucleophilic substitution, guaranteeing consistent quality and yield. We maintain stringent purity specifications and operate rigorous QC labs to verify that every batch of 4-hydroxy-1,8-naphthalimide meets the exacting standards demanded by the global pharmaceutical and fine chemical industries.
We invite forward-thinking organizations to collaborate with us to leverage this innovative technology for their supply chains. By partnering with our technical procurement team, you can access a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality targets. We encourage you to contact us today to request specific COA data and route feasibility assessments, allowing us to demonstrate how our expertise can drive efficiency and reliability in your production of complex organic intermediates.
