Advanced Palladium-Catalyzed Synthesis of 4-Allyl-3,5-Disubstituted Isoxazoles for Commercial Pharmaceutical Manufacturing
Introduction: A Breakthrough in Isoxazole Chemistry
The pharmaceutical and fine chemical industries continuously seek robust methodologies for constructing heterocyclic scaffolds, particularly the isoxazole ring, which serves as a privileged structure in numerous bioactive molecules. As illustrated in 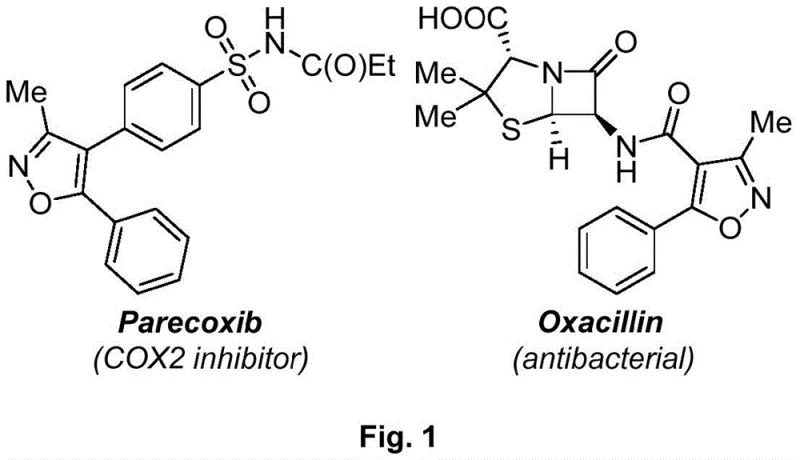 , compounds such as Parecoxib (a COX-2 inhibitor) and Oxacillin (an antibacterial agent) underscore the critical therapeutic value of this heterocycle. Addressing the persistent challenges in this domain, Chinese Patent CN108863969B, published on May 24, 2022, discloses a highly efficient synthesis method for 4-allyl-3,5-disubstituted isoxazoles. This innovation represents a significant leap forward by employing a palladium-catalyzed strategy that merges atom economy with operational simplicity. Unlike traditional routes that often struggle with regioselectivity or require hazardous reagents, this patented process utilizes readily accessible alkynone oxime ethers and 3-bromopropene to construct the target framework in a single catalytic step. For R&D directors and procurement specialists alike, this technology offers a compelling pathway to access high-value intermediates with reduced environmental impact and enhanced process safety.
, compounds such as Parecoxib (a COX-2 inhibitor) and Oxacillin (an antibacterial agent) underscore the critical therapeutic value of this heterocycle. Addressing the persistent challenges in this domain, Chinese Patent CN108863969B, published on May 24, 2022, discloses a highly efficient synthesis method for 4-allyl-3,5-disubstituted isoxazoles. This innovation represents a significant leap forward by employing a palladium-catalyzed strategy that merges atom economy with operational simplicity. Unlike traditional routes that often struggle with regioselectivity or require hazardous reagents, this patented process utilizes readily accessible alkynone oxime ethers and 3-bromopropene to construct the target framework in a single catalytic step. For R&D directors and procurement specialists alike, this technology offers a compelling pathway to access high-value intermediates with reduced environmental impact and enhanced process safety.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of isoxazole frameworks has relied heavily on transition metal-catalyzed [3+2] cycloaddition reactions or ring isomerization strategies involving pre-functionalized dipoles. While these methods have served the industry for decades, they are increasingly viewed as suboptimal for modern green chemistry standards. Conventional protocols frequently necessitate harsh reaction conditions, such as the use of strong bases or elevated temperatures that can degrade sensitive functional groups, thereby limiting substrate scope. Furthermore, many existing strategies suffer from low atom utilization rates, generating substantial waste streams that complicate downstream purification and increase disposal costs. The reliance on complex, multi-step substrate preparation further exacerbates these issues, leading to lower overall yields and extended production timelines. For supply chain managers, these inefficiencies translate into higher raw material costs and unpredictable delivery schedules, creating bottlenecks in the manufacturing of complex active pharmaceutical ingredients (APIs).
The Novel Approach
In stark contrast to these legacy methods, the technology described in CN108863969B introduces a streamlined palladium-catalyzed allylation and cyclization cascade. As depicted in the general reaction scheme 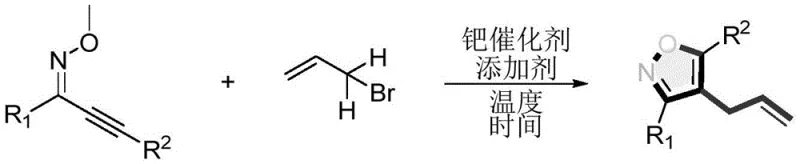 , the process directly couples an alkynone oxime ether with 3-bromopropene under mild thermal conditions. This approach eliminates the need for pre-functionalized dipoles or aggressive bases, operating effectively at temperatures between 70-80°C. The reaction demonstrates exceptional versatility, tolerating a wide array of substituents including electron-donating and electron-withdrawing phenyl groups, thienyl rings, and aliphatic chains. By simplifying the synthetic route to essentially two steps from basic starting materials (Sonogashira coupling followed by oxime formation and then this cyclization), the method drastically reduces the cumulative time and resource investment required. This shift towards milder, more direct chemistry not only improves the purity profile of the final product but also aligns perfectly with the industry's drive towards sustainable and cost-effective manufacturing practices.
, the process directly couples an alkynone oxime ether with 3-bromopropene under mild thermal conditions. This approach eliminates the need for pre-functionalized dipoles or aggressive bases, operating effectively at temperatures between 70-80°C. The reaction demonstrates exceptional versatility, tolerating a wide array of substituents including electron-donating and electron-withdrawing phenyl groups, thienyl rings, and aliphatic chains. By simplifying the synthetic route to essentially two steps from basic starting materials (Sonogashira coupling followed by oxime formation and then this cyclization), the method drastically reduces the cumulative time and resource investment required. This shift towards milder, more direct chemistry not only improves the purity profile of the final product but also aligns perfectly with the industry's drive towards sustainable and cost-effective manufacturing practices.
Mechanistic Insights into Pd-Catalyzed Cyclization and Allylation
The success of this synthesis lies in its elegant catalytic cycle, which orchestrates the formation of the isoxazole ring and the installation of the allyl group simultaneously. The reaction initiates with the oxidative addition of the palladium catalyst to the alkynone oxime ether substrate, generating a key alkenyl-palladium intermediate. This species then undergoes a crucial olefin migration and insertion step with 3-bromopropene, effectively weaving the allyl chain into the molecular architecture. The cycle concludes with a beta-bromine elimination that releases the final 4-allyl-3,5-disubstituted isoxazole product while regenerating the divalent palladium species to re-enter the catalytic loop. This mechanism is particularly advantageous because it avoids the formation of stable off-cycle intermediates that often plague similar transformations. From an impurity control perspective, the specificity of the beta-elimination step ensures that side reactions such as homocoupling or polymerization of the allyl bromide are minimized. The result is a clean reaction profile that facilitates easier purification, a critical factor for maintaining the stringent quality standards required in high-purity pharmaceutical intermediate production.
Furthermore, the substrate scope of this reaction is remarkably broad, accommodating diverse electronic and steric environments without significant loss in efficiency. For instance, substrates bearing electron-rich groups like methoxy substituents or electron-deficient groups like fluorine atoms proceed smoothly to afford the corresponding isoxazoles in excellent yields. The tolerance for heteroaromatic rings, such as thiophene, expands the utility of this method for synthesizing analogs relevant to agrochemical and medicinal chemistry programs. The ability to introduce a reactive allyl handle onto the isoxazole core is particularly valuable, as this group serves as a versatile pivot point for subsequent functionalization, allowing chemists to rapidly generate libraries of complex derivatives. This flexibility makes the process an ideal candidate for the commercial scale-up of complex fine chemical intermediates, where adaptability to different molecular targets is often a prerequisite for success.
How to Synthesize 4-Allyl-3,5-Disubstituted Isoxazole Efficiently
Implementing this synthesis in a laboratory or pilot plant setting requires precise adherence to the optimized conditions outlined in the patent to ensure maximum yield and reproducibility. The protocol is designed to be operationally simple, utilizing common reagents and standard equipment, which lowers the barrier to adoption for contract development and manufacturing organizations (CDMOs). The detailed standardized synthesis steps below provide a clear roadmap for executing this transformation, from reagent charging to final isolation. By following these guidelines, technical teams can reliably produce target compounds with the high purity necessary for downstream applications.
- Charge a reactor with alkynone oxime ether substrate, 3-bromopropene, palladium acetate catalyst, n-butylammonium bromide additive, and DMF solvent.
- Heat the reaction mixture to 70-80°C with stirring (600 rpm) for 10-40 minutes until TLC indicates completion.
- Cool to room temperature, quench with saturated ammonium chloride, extract with ethyl acetate, dry over MgSO4, and purify via column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this patented synthesis method offers tangible strategic advantages that extend beyond mere chemical novelty. The primary driver of value here is the significant simplification of the supply chain for raw materials. The key starting materials—acid chlorides, terminal alkynes, and 3-bromopropene—are commodity chemicals available in bulk quantities from multiple global suppliers. This abundance mitigates the risk of supply disruptions that often accompany specialized or proprietary reagents. Moreover, the reaction conditions are mild enough to be performed in standard glass-lined or stainless steel reactors without the need for exotic high-pressure or cryogenic equipment. This compatibility with existing infrastructure means that manufacturers can integrate this process into their current production lines with minimal capital expenditure, accelerating the time-to-market for new products derived from these intermediates.
- Cost Reduction in Manufacturing: The economic benefits of this process are driven by its high atom economy and exceptional yield performance. With isolated yields reaching up to 99% in optimized examples, the amount of wasted raw material is negligible compared to traditional low-yielding routes. The elimination of expensive transition metal catalysts (beyond the catalytic amount of palladium) and the avoidance of stoichiometric strong bases further reduce the cost of goods sold (COGS). Additionally, the short reaction time of merely 10 to 40 minutes dramatically increases reactor throughput, allowing facilities to produce larger volumes within the same operational window. These factors combine to deliver substantial cost savings, making the final pharmaceutical intermediate more price-competitive in the global market.
- Enhanced Supply Chain Reliability: Reliability in chemical supply is often compromised by complex, multi-step syntheses that have multiple points of failure. By condensing the synthesis of the isoxazole core into a highly efficient one-step cyclization from stable precursors, this method reduces the overall process complexity. The robustness of the reaction against varying substrate electronics ensures consistent output quality even when raw material batches vary slightly. This consistency is vital for maintaining long-term contracts with downstream API manufacturers who demand rigorous quality assurance. Consequently, adopting this method enhances the resilience of the supply chain, ensuring reducing lead time for high-purity intermediates and fostering stronger partnerships with key clients.
- Scalability and Environmental Compliance: As regulatory pressures regarding waste disposal and solvent usage intensify, the green chemistry attributes of this method become a major commercial asset. The process generates minimal waste due to its high selectivity and uses DMF, a solvent that, while requiring careful handling, is well-understood in industrial recovery systems. The patent explicitly notes successful scale-up to a 5-gram level without yield erosion, providing strong proof-of-concept for larger tonnage production. The absence of hazardous byproducts simplifies wastewater treatment and reduces the environmental footprint of the manufacturing site. This alignment with environmental, social, and governance (ESG) goals makes the process attractive to multinational corporations seeking sustainable sourcing options for their specialty chemical needs.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These answers are derived directly from the experimental data and beneficial effects reported in patent CN108863969B, providing a factual basis for decision-making. Understanding these details helps stakeholders evaluate the feasibility of integrating this route into their existing portfolios.
Q: What are the limitations of conventional isoxazole synthesis methods compared to this new patent?
A: Conventional methods often rely on harsh reaction conditions requiring strong bases or high temperatures, utilize complex pre-functionalized dipoles, or suffer from low atom utilization. This novel Pd-catalyzed approach operates under mild conditions (80°C) with simple, readily available substrates.
Q: How does this synthesis method improve supply chain reliability for pharmaceutical intermediates?
A: The method utilizes cheap and commercially available starting materials like acid chlorides, terminal alkynes, and 3-bromopropene. The short reaction time (10-40 min) and high yields (up to 99%) significantly reduce production lead times and inventory holding costs.
Q: Is this process scalable for industrial production?
A: Yes, the patent explicitly demonstrates successful scale-up to a 5-gram level without compromising yield. The use of standard solvents like DMF and common purification techniques like column chromatography suggests high feasibility for further commercial scale-up.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 4-Allyl-3,5-Disubstituted Isoxazole Supplier
The synthesis method disclosed in CN108863969B represents a powerful tool for accessing valuable isoxazole scaffolds, yet translating patent literature into commercial reality requires deep process engineering expertise. At NINGBO INNO PHARMCHEM, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of this chemistry are fully realized in practice. Our rigorous QC labs and stringent purity specifications guarantee that every batch of 4-allyl-3,5-disubstituted isoxazole meets the exacting standards required for pharmaceutical applications. We understand that consistency is key, and our team is dedicated to optimizing every parameter of this Pd-catalyzed process to deliver a product that is both cost-effective and chemically superior.
We invite potential partners to engage with our technical procurement team to discuss how this innovative synthesis can benefit your specific projects. Whether you require a Customized Cost-Saving Analysis for your current supply chain or need specific COA data and route feasibility assessments for new analogs, we are ready to provide the support you need. By leveraging our manufacturing capabilities and technical acumen, we can help you secure a reliable supply of these critical intermediates, driving your drug development programs forward with confidence and efficiency.
