Advanced Rhodium-Catalyzed C-H Activation for Scalable Gamma-Carboline Production
The pharmaceutical and fine chemical industries are constantly seeking robust methodologies to access bioactive heterocyclic scaffolds efficiently. Patent CN111961052B introduces a groundbreaking preparation method for γ-carboline derivatives, a class of alkaloids renowned for their significant biological activities including antiviral, antibacterial, and antitumor properties. This innovation leverages a sophisticated rhodium-catalyzed C-H activation strategy, utilizing ketoximes and alkynyl silane compounds as primary starting materials. The process operates under remarkably mild conditions, employing trifluoroethanol (TFE) as the solvent in the presence of a [Cp*RhCl2]2 and AgSbF6 catalytic system. For R&D directors and procurement specialists alike, this patent represents a pivotal shift towards more sustainable and economically viable manufacturing pathways for high-value nitrogen-containing heterocycles.
Traditional synthetic routes for γ-carboline frameworks have long been plagued by significant operational inefficiencies and chemical limitations. Conventional approaches often rely on transition metal-catalyzed couplings or acid-catalyzed condensations that necessitate cumbersome multi-step sequences, strictly anhydrous environments, or extreme temperatures. These legacy methods frequently suffer from poor regioselectivity, particularly when employing asymmetric internal alkynes or terminal alkynes, leading to complex mixtures that are difficult and costly to separate. Furthermore, the requirement for expensive oxidants or sensitive reagents increases the overall cost of goods and complicates waste management protocols. In contrast, the novel approach detailed in CN111961052B circumvents these historical bottlenecks by utilizing a direct C-H functionalization strategy that is both atom-economical and operationally simple.
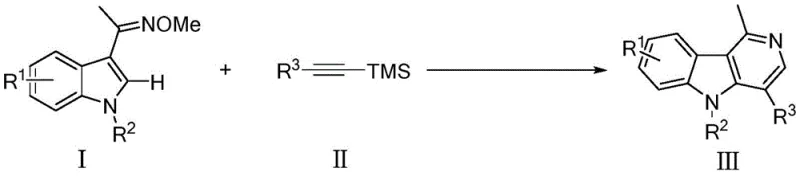
The new methodology distinguishes itself through its unique ability to control regioselectivity via steric hindrance, a feature rarely achieved in standard alkyne insertions. By employing alkynyl silanes with significant steric bulk, the reaction successfully directs the cyclization to produce C3-unsubstituted γ-carbolines with high precision, effectively reversing the typical regiochemical outcome observed in similar transformations. This level of control is critical for medicinal chemists aiming to explore structure-activity relationships (SAR) without being hindered by inseparable isomeric byproducts. The reaction proceeds smoothly in air at 120°C, eliminating the need for expensive inert gas setups and specialized low-temperature equipment, thereby drastically simplifying the engineering requirements for production facilities.
Mechanistic Insights into Rhodium-Catalyzed C-H Activation and Cyclization
The core of this transformative synthesis lies in the intricate mechanism of the rhodium(III) catalytic cycle, which facilitates the direct activation of inert C-H bonds. The process initiates with the coordination of the oxime nitrogen to the cationic rhodium species generated in situ from the dimer and silver salt. This coordination serves as a powerful directing group, guiding the metal center to the proximal C-H bond on the indole ring for metallation. Following C-H activation, the alkynyl silane undergoes migratory insertion into the Rh-C bond. Crucially, the bulky trimethylsilyl (TMS) group on the alkyne exerts a steric influence that dictates the orientation of insertion, ensuring the formation of the desired regioisomer. Subsequent reductive elimination and aromatization steps release the final γ-carboline product while regenerating the active catalyst species for the next turnover.
Impurity control in this system is inherently managed by the high specificity of the catalytic cycle and the stability of the intermediates. The use of trifluoroethanol (TFE) as a solvent plays a dual role, not only solubilizing the organic substrates but also potentially stabilizing charged intermediates through hydrogen bonding interactions. The redox-neutral nature of the transformation means that no external oxidants are required, which significantly reduces the formation of oxidative byproducts that often complicate downstream purification. For quality control teams, this translates to a cleaner crude reaction profile, allowing for simpler workup procedures such as direct concentration and flash chromatography. The robustness of the catalyst system against air and moisture further ensures consistent batch-to-batch reproducibility, a key metric for reliable pharmaceutical intermediate supplier operations.
How to Synthesize Gamma-Carboline Derivatives Efficiently
Implementing this synthesis protocol requires precise adherence to the molar ratios and reaction parameters outlined in the patent to maximize yield and purity. The standard procedure involves combining the ketoxime substrate and the alkynyl silane coupling partner in a reaction vessel, followed by the addition of the rhodium catalyst and silver additive. The mixture is then heated under aerobic conditions to drive the cyclization to completion. While the general concept is straightforward, optimizing parameters such as stirring rate and heating ramp can further enhance process performance. The detailed standardized synthesis steps for this specific transformation are provided in the guide below.
- Mix ketoxime substrate, alkynyl silane coupling partner, [Cp*RhCl2]2 catalyst, and AgSbF6 additive in trifluoroethanol (TFE) solvent at room temperature.
- Seal the reaction vessel and heat the mixture to 120°C in air for approximately 24 hours to facilitate the C-H activation and cyclization.
- Cool the reaction to room temperature, concentrate under reduced pressure, and purify the crude product via flash column chromatography to obtain the target gamma-carboline.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this rhodium-catalyzed protocol offers substantial strategic advantages for supply chain optimization and cost management. The elimination of harsh reaction conditions and sensitive reagents directly correlates to reduced operational expenditures and lower capital investment in specialized reactor infrastructure. By removing the dependency on inert atmosphere techniques, facilities can increase throughput and reduce turnaround times between batches. This operational simplicity is a key driver for cost reduction in fine chemical manufacturing, allowing producers to allocate resources more efficiently towards quality assurance and scale-up activities rather than complex process controls.
- Cost Reduction in Manufacturing: The economic benefits of this process are driven primarily by the use of cheap and readily available raw materials, such as simple ketoximes and commercially sourced alkynyl silanes. The catalytic system, while utilizing precious metals, operates at low loading levels and does not require expensive ligands or stoichiometric oxidants. Furthermore, the simplified workup procedure minimizes solvent consumption and waste disposal costs. These factors combine to create a significantly leaner cost structure compared to traditional multi-step syntheses, providing a competitive edge in pricing for high-purity gamma-carboline derivatives.
- Enhanced Supply Chain Reliability: Supply chain resilience is bolstered by the robustness of the reaction conditions, which tolerate air and moisture. This tolerance reduces the risk of batch failures due to environmental fluctuations or minor procedural deviations, ensuring a more consistent supply of critical intermediates. The wide substrate adaptability demonstrated in the patent means that a single platform technology can be used to generate a diverse library of analogues, reducing the need for multiple distinct production lines. This flexibility allows for rapid response to changing market demands and accelerates the timeline for reducing lead time for high-purity intermediates.
- Scalability and Environmental Compliance: Scaling this chemistry from gram to kilogram quantities is facilitated by the absence of hazardous reagents and the use of relatively benign solvents like trifluoroethanol. The redox-neutral nature of the reaction minimizes the generation of toxic waste streams associated with heavy metal oxidants, aligning well with modern green chemistry principles and environmental regulations. The high atom economy of the C-H activation approach ensures that a greater proportion of starting material mass is incorporated into the final product, reducing the overall environmental footprint. This makes the process highly attractive for commercial scale-up of complex heterocycles in regulated markets.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented synthesis method. Understanding these details is crucial for technical teams evaluating the feasibility of integrating this route into their existing manufacturing portfolios. The answers are derived directly from the experimental data and beneficial effects described in the patent documentation, ensuring accuracy and relevance for decision-makers.
Q: What is the key advantage of this Rh-catalyzed method over traditional synthesis?
A: Unlike conventional methods that often require harsh conditions and suffer from poor regioselectivity, this patent describes a mild, redox-neutral process operating in air at 120°C with unique reverse regioselectivity induced by steric hindrance.
Q: Can this process be scaled for commercial manufacturing?
A: Yes, the process utilizes cheap and readily available raw materials, simple operation without inert gas protection, and demonstrates excellent substrate adaptability, making it highly suitable for commercial scale-up of complex heterocycles.
Q: What represents the typical yield range for this synthesis?
A: The patent data indicates excellent yields across a broad substrate scope, with many examples achieving yields between 60% and 92%, demonstrating high efficiency for pharmaceutical intermediate production.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Gamma-Carboline Derivatives Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of efficient synthetic routes in the development of next-generation therapeutics. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that promising laboratory discoveries like the rhodium-catalyzed γ-carboline synthesis can be successfully translated into industrial reality. We maintain stringent purity specifications and operate rigorous QC labs to guarantee that every batch of intermediate meets the exacting standards required by global pharmaceutical partners. Our commitment to technical excellence allows us to navigate the complexities of C-H activation chemistry with confidence and precision.
We invite you to collaborate with us to leverage this advanced technology for your drug discovery programs. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific project needs. We are prepared to provide specific COA data and comprehensive route feasibility assessments to demonstrate how our manufacturing capabilities can support your supply chain goals. Let us help you accelerate your path to market with reliable, high-quality chemical solutions.
