Advanced Palladium-Catalyzed Carbonylation for High-Purity Unsaturated Ketone Intermediates
Introduction to Advanced Carbonylation Technology
The landscape of fine chemical synthesis is constantly evolving, driven by the need for more efficient, atom-economical, and environmentally benign processes. A pivotal advancement in this domain is detailed in patent CN101891571A, which discloses a robust method for preparing unsaturated ketones through a transition metal-catalyzed three-component coupling reaction. This technology leverages the unique reactivity of carbon monoxide as a C1 building block, reacting benzyl halogenated compounds with tributylallyl tin in the presence of a zero-valent palladium catalyst and phosphine ligands. Unlike traditional routes that rely on harsh reducing agents or multi-step functional group manipulations, this carbonylation strategy offers a direct pathway to valuable α,β-unsaturated ketone scaffolds. These structures are ubiquitous in pharmaceutical intermediates, agrochemicals, and complex organic synthesis, serving as critical precursors for Michael additions and heterocycle formations. By integrating this methodology, manufacturers can achieve significant improvements in process mass intensity and operational safety, positioning it as a cornerstone technology for modern industrial organic chemistry.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of unsaturated ketones has relied heavily on the condensation of carbonyl carboxylic acids and their derivatives, often necessitating the use of stoichiometric metal hydrides or hydrosilanes as reducing agents. These classical approaches, while effective on a small laboratory scale, present substantial challenges when translated to commercial manufacturing. The reliance on expensive and hazardous reducing agents not only inflates raw material costs but also generates significant quantities of chemical waste, complicating downstream purification and environmental compliance. Furthermore, earlier attempts at palladium-catalyzed carbonylation, such as those reported by Heck in the 1970s, often required extreme reaction conditions, including temperatures of 80-100°C and exceedingly high carbon monoxide pressures ranging from 1200 to 1500 psi. Such demanding parameters impose severe constraints on equipment requirements, increase energy consumption, and elevate safety risks associated with high-pressure gas handling, thereby limiting the scalability and economic viability of these processes for widespread industrial adoption.
The Novel Approach
The methodology outlined in CN101891571A represents a paradigm shift by enabling the synthesis of unsaturated ketones under remarkably mild and controlled conditions. By utilizing a zero-valent palladium catalyst system, the reaction facilitates the insertion of carbon monoxide at atmospheric or near-atmospheric pressures (0.5 atm to 10 atm) and moderate temperatures (20°C to 100°C). This drastic reduction in operating severity translates directly into enhanced process safety and reduced capital expenditure for high-pressure reactors. The three-component coupling of benzyl halides, allyl tin reagents, and CO proceeds with high efficiency, as demonstrated by yields reaching up to 88% in specific embodiments. Moreover, the versatility of the solvent system, which accommodates common organic solvents like tetrahydrofuran, toluene, and 1,4-dioxane, allows for seamless integration into existing manufacturing infrastructure. This novel approach effectively bypasses the need for pre-functionalized carboxylic acid derivatives, streamlining the synthetic route and offering a cost reduction in pharmaceutical intermediate manufacturing by minimizing step count and reagent costs.
Mechanistic Insights into Palladium-Catalyzed Carbonylation
The efficacy of this transformation is rooted in a sophisticated catalytic cycle involving oxidative addition, migratory insertion, transmetallation, and reductive elimination. The cycle initiates with the oxidative addition of the benzyl halide to the zero-valent palladium center, generating a reactive benzyl-palladium(II) species. Subsequently, carbon monoxide coordinates to the metal center and undergoes migratory insertion into the palladium-carbon bond, forming an acyl-palladium intermediate. This key step introduces the carbonyl functionality essential for the ketone product. The cycle is completed through transmetallation with the tributylallyl tin reagent, followed by reductive elimination to release the desired unsaturated ketone and regenerate the active palladium(0) catalyst. Understanding this mechanism is crucial for optimizing reaction parameters, such as ligand selection and CO pressure, to maximize turnover frequency and minimize side reactions.
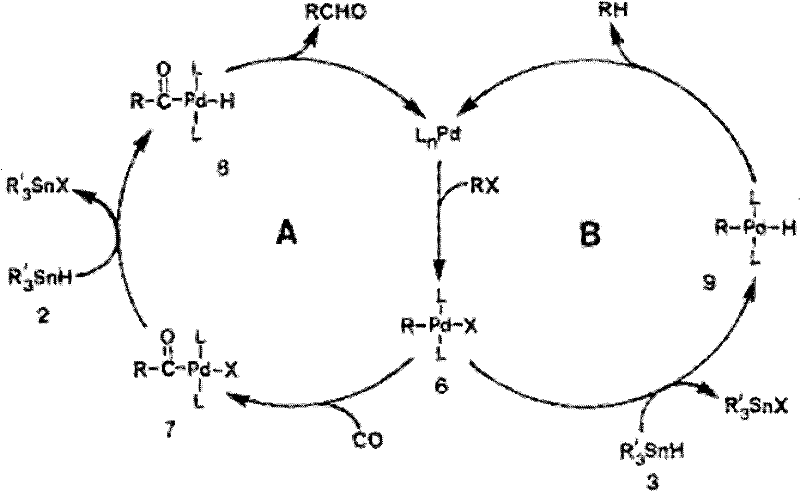
Impurity control is another critical aspect addressed by the mechanistic understanding of this process. A common challenge in organotin-mediated reactions is the removal of toxic tin byproducts, which can persist through standard aqueous workups. The patent specifies a targeted purification strategy wherein the crude reaction mixture is treated with aqueous solutions of metal fluorides, such as sodium fluoride (NaF) or potassium fluoride (KF). Fluoride ions exhibit a high affinity for tin, forming stable fluoro-tinate complexes that are highly soluble in the aqueous phase or precipitate out, thereby facilitating their separation from the organic product. This specific workup protocol ensures that the final unsaturated ketone meets stringent purity specifications required for pharmaceutical applications, effectively mitigating the risk of heavy metal contamination in the active pharmaceutical ingredient (API) supply chain.
How to Synthesize Unsaturated Ketones Efficiently
Implementing this synthesis requires careful attention to reaction setup and workup procedures to ensure reproducibility and safety. The process begins with the charging of a dried autoclave with the palladium catalyst, substrate, and solvent under an inert atmosphere to prevent catalyst deactivation by oxygen. Following the addition of the organotin reagent, the system is pressurized with carbon monoxide and heated to the target temperature with vigorous stirring. Upon completion, the safe release of excess gas and the subsequent fluoride treatment are critical steps to isolate the pure product. For a comprehensive guide on executing this protocol with precision, please refer to the standardized operating procedures below.
- Load the autoclave with zero-valent palladium catalyst (e.g., Pd(PPh3)4), benzyl halide substrate, organic solvent, and tributylallyltin under inert atmosphere.
- Pressurize the reactor with carbon monoxide (0.5-10 atm) and heat the mixture to 20°C-100°C with stirring for 12-24 hours.
- Treat the crude reaction mixture with aqueous metal fluoride (NaF or KF) to remove tin byproducts, followed by extraction and column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this palladium-catalyzed carbonylation technology offers compelling advantages for procurement and supply chain management. The shift away from stoichiometric reducing agents and high-pressure equipment significantly lowers the barrier to entry for manufacturing these complex intermediates. By utilizing readily available benzyl halides and allyl tin reagents, companies can secure a more resilient supply chain that is less susceptible to the volatility associated with specialized precursors. Furthermore, the mild reaction conditions reduce energy consumption and extend the lifespan of production equipment, contributing to long-term operational sustainability.
- Cost Reduction in Manufacturing: The elimination of expensive metal hydride reducing agents and the ability to operate at low carbon monoxide pressures drastically simplify the process economics. This streamlined approach reduces the overall cost of goods sold (COGS) by minimizing reagent expenses and lowering utility costs associated with heating and pressurization. Additionally, the high atom economy of the three-component coupling ensures that a greater proportion of raw materials are incorporated into the final product, reducing waste disposal costs.
- Enhanced Supply Chain Reliability: The reliance on commodity chemicals such as benzyl chlorides and common solvents enhances supply chain stability. Unlike proprietary reagents that may have single-source suppliers, the inputs for this reaction are widely available from multiple global vendors. This diversification mitigates the risk of supply disruptions and allows procurement teams to negotiate more favorable pricing terms, ensuring consistent availability of high-purity unsaturated ketones for downstream synthesis.
- Scalability and Environmental Compliance: The process is inherently scalable, having been demonstrated in autoclave systems that can be easily adapted from pilot plant to commercial scale. The use of a fluoride-based workup for tin removal provides a robust method for meeting strict environmental regulations regarding heavy metal discharge. This compliance capability is essential for maintaining uninterrupted production schedules and avoiding regulatory penalties, making it an ideal choice for the commercial scale-up of complex pharmaceutical intermediates.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation and optimization of this carbonylation technology. These insights are derived directly from the experimental data and claims presented in the patent literature, providing a reliable foundation for process development decisions. Understanding these nuances helps R&D and operations teams anticipate potential challenges and leverage the full potential of this synthetic route.
Q: What are the optimal reaction conditions for this carbonylation process?
A: According to patent CN101891571A, the reaction proceeds efficiently at mild temperatures between 20°C and 100°C (optimally 80°C) and low carbon monoxide pressures ranging from 0.5 atm to 10 atm, typically completing within 12 to 24 hours.
Q: How are tin byproducts removed to ensure high purity?
A: The process utilizes a specialized workup involving treatment with saturated or supersaturated aqueous solutions of metal fluorides, such as NaF or KF. This step effectively precipitates or complexes organotin residues, allowing for clean separation via organic extraction.
Q: Which catalysts and ligands are compatible with this synthesis?
A: The method employs zero-valent palladium catalysts, specifically Pd(PPh3)4 or Pd2(dba)3. It supports various phosphine ligands including PPh3, dppb, dppe, dppf, and dppp to optimize turnover and selectivity.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Unsaturated Ketone Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the success of your drug development programs. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that we can meet your volume requirements without compromising on quality. We adhere to stringent purity specifications and utilize rigorous QC labs to verify that every batch of unsaturated ketone delivered meets the exacting standards required for GMP manufacturing. Our commitment to technical excellence allows us to navigate the complexities of palladium-catalyzed reactions, delivering products with minimal impurity profiles.
We invite you to collaborate with us to optimize your supply chain for these vital building blocks. By engaging with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your specific project needs. We encourage you to reach out today to obtain specific COA data and route feasibility assessments, ensuring that your transition to this advanced manufacturing platform is seamless and economically advantageous.
