Scalable Photocatalytic Synthesis of Beta-Caryophyllene Derived Difluoroalkyl Tricyclic Compounds for Advanced Drug Discovery
Scalable Photocatalytic Synthesis of Beta-Caryophyllene Derived Difluoroalkyl Tricyclic Compounds for Advanced Drug Discovery
The landscape of medicinal chemistry is continuously evolving towards the incorporation of fluorine atoms into bioactive scaffolds to enhance metabolic stability and binding affinity. A significant breakthrough in this domain is detailed in Chinese Patent CN112279754B, which discloses a novel preparation method for difluoroalkyl substituted tricyclic compounds derived from beta-caryophyllene. This technology leverages visible-light photocatalysis to achieve a complex molecular rearrangement and functionalization in a single operational step. For R&D directors and procurement specialists in the pharmaceutical sector, this represents a pivotal shift from multi-step, low-yield traditional syntheses to a streamlined, high-efficiency process. The ability to transform a readily available natural product like beta-caryophyllene into highly valued fluorinated intermediates opens new avenues for developing anti-inflammatory and anticancer agents with improved pharmacokinetic profiles.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the functionalization of the beta-caryophyllene skeleton has been fraught with challenges related to regioselectivity and harsh reaction conditions. Prior art, such as the hetero-Diels-Alder reactions reported by Lawrence et al., often requires specific aldehyde partners and rigorous control to achieve desired bicyclic or tricyclic architectures. Furthermore, alternative approaches involving hydroboration-oxidation followed by esterification, while effective for introducing oxygenated functionality, involve multiple discrete steps that cumulatively reduce overall yield and increase waste generation. These conventional pathways frequently rely on stoichiometric amounts of reactive reagents or transition metal catalysts that necessitate complex purification protocols to remove trace metal impurities, a critical concern for API manufacturing. The cumulative effect of these limitations is a high cost of goods and extended lead times, making the commercial supply of complex caryophyllene derivatives economically challenging for large-scale applications.
The Novel Approach
In stark contrast, the methodology described in patent CN112279754B introduces a direct, one-pot radical cyclization strategy that dramatically simplifies the synthetic landscape. By utilizing iododifluoro methyl aryl ketones or iododifluoro acetates as radical precursors, the process enables the simultaneous construction of the tricyclic core and the installation of the valuable difluoroalkyl moiety. This transformation is driven by blue light irradiation in the presence of a catalytic amount of aryl phenol and a mild inorganic base, eliminating the need for expensive photoredox catalysts or harsh thermal activation. As illustrated in the reaction scheme below, the process converts beta-caryophyllene directly into the target tricyclic structure with high atom economy.
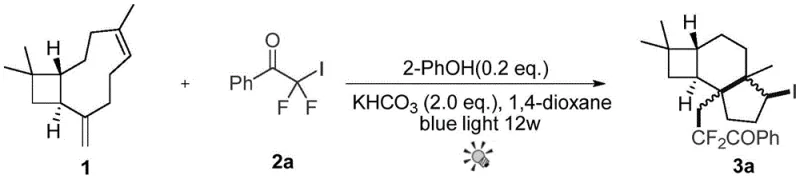
This novel approach not only reduces the number of unit operations but also operates under remarkably mild conditions, typically between 0°C and 120°C, ensuring the integrity of the sensitive terpene backbone is maintained throughout the synthesis. The use of common polar solvents like 1,4-dioxane further enhances the practicality of this method for industrial adoption, positioning it as a superior alternative for the cost reduction in pharmaceutical intermediate manufacturing.
Mechanistic Insights into Visible-Light Mediated Radical Cyclization
The core of this technological advancement lies in the efficient generation and stabilization of difluoroalkyl radicals under visible light irradiation. The mechanism likely involves the homolytic cleavage of the carbon-iodine bond in the iododifluoro reagent, facilitated by the interaction with the aryl phenol catalyst and blue light photons. This generates a reactive difluoroalkyl radical species that selectively attacks the electron-rich double bond of the beta-caryophyllene nine-membered ring. The resulting carbon-centered radical then undergoes a rapid intramolecular cyclization onto the adjacent cyclobutane-fused system, forging the new tricyclic architecture with high stereocontrol. The presence of the phenol additive is crucial, as it likely acts as a hydrogen atom transfer mediator or a stabilizer for the radical intermediates, preventing non-productive side reactions and ensuring high conversion rates. This mechanistic pathway avoids the formation of carbocation intermediates that are prone to skeletal rearrangements, thereby preserving the specific structural motifs required for biological activity.
From an impurity control perspective, the mildness of the photocatalytic conditions plays a vital role in maintaining a clean reaction profile. Traditional thermal radical initiators often require high temperatures that can trigger polymerization or decomposition of the starting materials, leading to complex impurity spectra that are difficult to separate. In this photochemical process, the energy input is precise and controlled, limiting the formation of thermal byproducts. Furthermore, the use of inorganic bases such as potassium bicarbonate ensures that the reaction medium remains neutral to slightly basic, preventing acid-catalyzed degradation of the product. This inherent cleanliness of the reaction translates directly to simplified downstream processing, where standard aqueous workups and silica gel chromatography are sufficient to isolate the product in high purity, meeting the stringent specifications required for reliable pharmaceutical intermediate suppliers.
How to Synthesize Beta-Caryophyllene Difluoroalkyl Tricyclic Compounds Efficiently
The operational simplicity of this synthesis makes it highly attractive for process chemists looking to implement new routes quickly. The procedure begins with the preparation of a reaction mixture under an inert atmosphere to prevent oxygen quenching of the radical species. Key to the success of the reaction is the precise ratio of reactants, where a slight excess of the iododifluoro reagent ensures complete consumption of the valuable beta-caryophyllene starting material. The reaction progress is conveniently monitored using thin-layer chromatography, allowing for real-time decision-making regarding reaction endpoints. Once the conversion is complete, the workup involves standard liquid-liquid extraction techniques that are easily scalable from gram to kilogram quantities. The detailed standardized synthesis steps for implementing this protocol in your laboratory are provided in the guide below.
- Prepare the reaction mixture by combining beta-caryophyllene, iododifluoro methyl aryl ketone or iododifluoro acetate, inorganic base, and aryl phenol in a polar solvent under inert gas protection.
- Irradiate the reaction mixture with blue light (400nm-500nm) while maintaining the temperature between 0°C and 120°C with continuous stirring until TLC indicates complete conversion.
- Quench the reaction with water, extract the organic phase with ethyl acetate, wash with brine, dry over anhydrous sodium sulfate, and purify via column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this photocatalytic technology offers substantial strategic benefits beyond mere technical feasibility. The primary advantage lies in the drastic simplification of the supply chain for raw materials. Beta-caryophyllene is a natural product extracted from essential oils like clove and cinnamon, making it abundant and price-stable compared to synthetic petrochemical feedstocks. The iododifluoro reagents, while specialized, are becoming increasingly commoditized due to the growing demand for fluorinated building blocks in the agrochemical and pharma sectors. By consolidating multiple synthetic steps into a single pot, manufacturers can significantly reduce labor costs, solvent consumption, and reactor occupancy time, leading to a leaner and more responsive production model.
- Cost Reduction in Manufacturing: The elimination of expensive transition metal catalysts and the use of inexpensive inorganic bases like potassium bicarbonate directly lower the bill of materials. Furthermore, the high reaction conversion rates minimize the loss of starting materials, ensuring that every gram of input contributes to the final output. The simplified purification process reduces the consumption of silica gel and eluents, which are often significant cost drivers in fine chemical manufacturing. These factors combine to create a robust economic model that supports competitive pricing for high-value intermediates without compromising on quality standards.
- Enhanced Supply Chain Reliability: Relying on a short, robust synthetic route reduces the risk of bottlenecks that often plague multi-step processes. With fewer intermediate isolation steps, there are fewer opportunities for yield losses or quality deviations that could disrupt delivery schedules. The use of stable, commercially available solvents and reagents ensures that production can continue uninterrupted even during periods of market volatility for specialized chemicals. This reliability is crucial for maintaining continuous supply to downstream API manufacturers who depend on just-in-time delivery models to manage their own inventory levels efficiently.
- Scalability and Environmental Compliance: The process is designed with scalability in mind, utilizing equipment and conditions that are compatible with existing industrial infrastructure. The absence of heavy metals simplifies waste treatment and disposal, aligning with increasingly stringent environmental regulations globally. The green chemistry attributes of this method, such as energy efficiency through LED lighting and reduced solvent waste, enhance the sustainability profile of the final product. This compliance not only mitigates regulatory risk but also appeals to end-users who are prioritizing environmentally responsible sourcing in their supply chains.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented technology. These insights are derived directly from the experimental data and beneficial effects described in the patent documentation, providing a clear understanding of the process capabilities. Understanding these details helps stakeholders evaluate the fit of this technology within their existing development pipelines and supply networks.
Q: What are the key advantages of this photocatalytic method over traditional thermal cyclization?
A: This method utilizes mild visible light irradiation instead of harsh thermal conditions, significantly reducing energy consumption and minimizing thermal degradation of the sensitive sesquiterpene framework, leading to higher purity profiles.
Q: Is this process suitable for large-scale industrial production?
A: Yes, the patent explicitly highlights the ease of scale-up due to the use of common polar solvents, inexpensive inorganic bases, and simple workup procedures involving standard extraction and chromatography.
Q: What types of difluoroalkyl groups can be introduced using this protocol?
A: The protocol demonstrates broad substrate scope, successfully incorporating various iododifluoro methyl aryl ketones with different electronic substituents as well as iododifluoro acetates, allowing for diverse functionalization.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Beta-Caryophyllene Derivative Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of this photocatalytic methodology for the next generation of fluorinated therapeutics. As a dedicated CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project transitions smoothly from benchtop discovery to full-scale manufacturing. Our state-of-the-art facilities are equipped with advanced photochemical reactors capable of handling the specific wavelength requirements of this process, guaranteeing consistent batch-to-batch reproducibility. We maintain stringent purity specifications and operate rigorous QC labs to ensure that every shipment of beta-caryophyllene derivatives meets the highest industry standards for potency and impurity profiles.
We invite you to collaborate with our technical team to explore how this innovative synthesis can optimize your drug development timeline and budget. By leveraging our expertise in process optimization, we can provide a Customized Cost-Saving Analysis tailored to your specific volume requirements. Please contact our technical procurement team today to request specific COA data and route feasibility assessments, and let us help you secure a sustainable supply of these critical high-purity pharmaceutical intermediates.
