Advanced Synthesis of N-Methyl P-Nitroaniline: High Purity and Scalable Manufacturing Solutions
Advanced Synthesis of N-Methyl P-Nitroaniline: High Purity and Scalable Manufacturing Solutions
The global demand for high-performance energetic materials and specialized pharmaceutical intermediates continues to drive innovation in organic synthesis, particularly for nitro-aniline derivatives. Patent CN101580473B introduces a groundbreaking methodology for the preparation of N-methyl p-nitroaniline, a critical compound utilized extensively as a stabilizer in gunpowder propellants and as a benchmark reference substance in analytical chemistry. This proprietary technology addresses the longstanding inefficiencies of conventional manufacturing by establishing a温和 (mild), three-step synthetic pathway that eliminates the need for hazardous reagents and extreme reaction conditions. By shifting away from corrosive halogen exchange or high-pressure ammonolysis, this novel approach offers a robust framework for producing high-purity intermediates with exceptional consistency. For R&D directors and procurement specialists seeking reliable pharma intermediate supplier partnerships, understanding the mechanistic superiority of this route is essential for optimizing supply chain resilience and reducing overall production costs in the fine chemicals sector.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial synthesis of N-methyl p-nitroaniline has been plagued by severe safety hazards and engineering challenges that inflate capital expenditure and operational risks. Traditional Method 1, as illustrated below, relies on a halogen exchange reaction between p-nitrochlorobenzene and potassium fluoride at temperatures exceeding 170°C, which inevitably generates highly toxic hydrogen fluoride (HF) gas as a by-product.
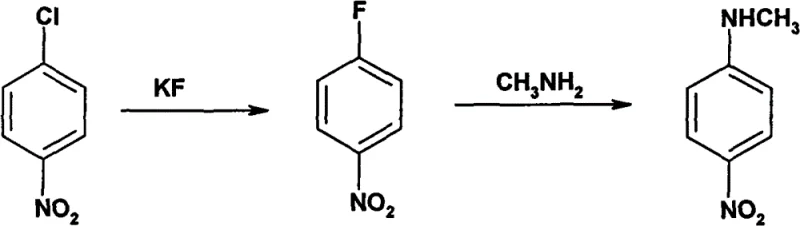
The presence of HF necessitates the use of specialized Hastelloy or lined reactors to prevent catastrophic equipment corrosion, while also requiring rigorous scrubbing systems to meet environmental compliance standards. Furthermore, alternative routes such as direct ammonolysis (Method 3) demand high-pressure autoclaves to force the nucleophilic substitution, creating significant bottlenecks in batch processing throughput. These legacy processes not only suffer from moderate yields around 70% but also introduce complex impurity profiles due to side reactions promoted by the harsh thermal and acidic environments, thereby complicating downstream purification and increasing waste treatment burdens for chemical manufacturers.
The Novel Approach
In stark contrast to these hazardous legacy protocols, the patented methodology outlined in CN101580473B employs a sophisticated protection-deprotection strategy that operates under significantly milder conditions, ensuring both operator safety and product integrity. As depicted in the reaction scheme below, the process initiates with the formylation of p-nitroaniline using formic acid, followed by a selective N-methylation using iodomethane and potassium tert-butoxide in DMF, and concludes with a gentle reduction using sodium borohydride.
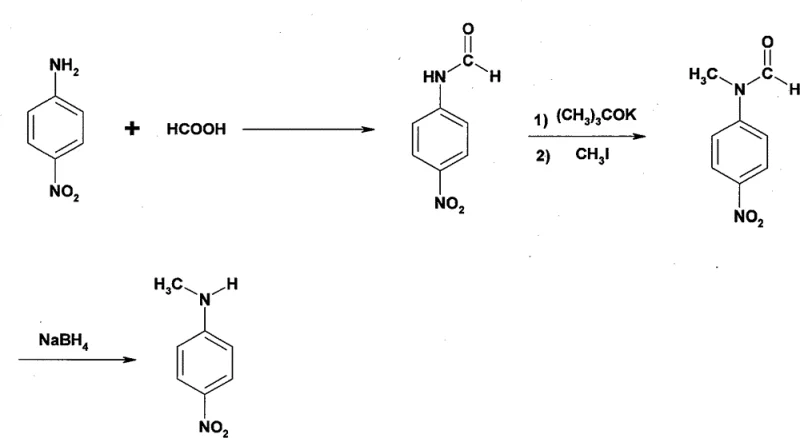
This innovative sequence completely bypasses the generation of corrosive acids and eliminates the requirement for high-pressure vessels, allowing the reaction to proceed efficiently at atmospheric pressure and temperatures ranging from ambient to 105°C. The strategic use of the formyl group acts as a protecting group that directs the methylation specifically to the nitrogen atom, preventing unwanted C-alkylation or polymerization often seen in direct alkylation attempts. Consequently, this route achieves a remarkable total yield exceeding 80% with individual step yields consistently above 90%, demonstrating a level of efficiency and cost reduction in pharmaceutical intermediates manufacturing that is unattainable with older technologies.
Mechanistic Insights into Formylation-Methylation-Reduction Cascade
The core chemical ingenuity of this process lies in the sequential manipulation of the aniline nitrogen's nucleophilicity and steric environment to achieve precise mono-methylation. In the first stage, p-nitroaniline reacts with formic acid to form N-formyl p-nitroaniline; this step reduces the electron density on the nitrogen lone pair through resonance with the carbonyl group, effectively moderating its reactivity for the subsequent alkylation. The second stage utilizes potassium tert-butoxide as a strong, non-nucleophilic base to deprotonate the amide nitrogen, generating a highly reactive amidate anion in situ within the DMF solvent matrix. This anion then undergoes a clean SN2 nucleophilic attack on methyl iodide, installing the methyl group with high regioselectivity while the electron-withdrawing nitro group on the aromatic ring further stabilizes the intermediate against oxidative degradation.
The final reduction step is equally critical for ensuring the high-purity specifications required by downstream applications. Sodium borohydride is employed as a chemoselective reducing agent that targets the formyl carbonyl group without affecting the sensitive nitro functionality on the benzene ring, a selectivity that is difficult to achieve with stronger reducing agents like lithium aluminum hydride. The mechanism involves the hydride transfer to the carbonyl carbon, followed by hydrolysis during the quench with saturated ammonium chloride solution, which cleaves the C-N bond to release the free secondary amine. This mild reduction environment prevents the formation of azo or hydrazo impurities that typically arise from the partial reduction of nitro groups, thereby guaranteeing a final product purity of ≥98% after a simple ethanol-water recrystallization, which is vital for meeting the stringent quality control standards of global API manufacturers.
How to Synthesize N-Methyl P-Nitroaniline Efficiently
The implementation of this three-step synthesis requires careful control of stoichiometry and temperature to maximize the economic and technical benefits described in the patent literature. The process begins with heating p-nitroaniline and formic acid to 100°C to ensure complete conversion to the formyl intermediate, followed by cooling before the addition of the base and alkylating agent to prevent exothermic runaway. Detailed standard operating procedures regarding exact molar ratios, solvent recovery protocols, and crystallization parameters are essential for reproducibility at scale.
- React p-nitroaniline with formic acid at 80-105°C to form N-formyl p-nitroaniline.
- Dissolve the intermediate in DMF, add potassium tert-butoxide and methyl iodide at room temperature to obtain N-methyl-N-formyl p-nitroaniline.
- Reduce the N-methyl-N-formyl derivative using sodium borohydride in ethanol to yield high-purity N-methyl p-nitroaniline.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the transition to this patented synthesis route represents a strategic opportunity to mitigate risk and optimize the total cost of ownership for N-methyl p-nitroaniline sourcing. By eliminating the need for specialized high-pressure reactors and corrosion-resistant alloys, the capital expenditure (CAPEX) required for setting up production lines is drastically reduced, allowing for more flexible manufacturing footprints. Furthermore, the avoidance of toxic hydrogen fluoride removes the substantial operational costs associated with hazardous waste disposal, neutralization, and regulatory compliance monitoring, leading to substantial cost savings over the lifecycle of the product. The mild reaction conditions also translate to lower energy consumption, as the process does not require sustained high-temperature heating or energy-intensive compression, aligning with modern sustainability goals and green chemistry principles.
- Cost Reduction in Manufacturing: The elimination of expensive catalysts like tetramethylammonium chloride and the removal of corrosive reagents significantly lowers raw material costs and maintenance expenses. Since the process avoids the generation of HF, there is no need for costly scrubbing systems or specialized linings for storage tanks, which directly improves the margin profile for bulk chemical production. Additionally, the high yield per step minimizes the loss of valuable starting materials, ensuring that the input-to-output ratio is optimized for maximum economic efficiency without compromising on quality.
- Enhanced Supply Chain Reliability: The reliance on commodity chemicals such as formic acid, iodomethane, and sodium borohydride ensures a stable and diversified supply base, reducing the risk of shortages associated with specialized fluorinating agents. The simplicity of the work-up procedure, which primarily involves filtration and recrystallization rather than complex distillation or chromatography, shortens the batch cycle time significantly. This operational agility allows manufacturers to respond more rapidly to fluctuations in market demand, ensuring consistent delivery schedules and reducing lead time for high-purity pharmaceutical intermediates.
- Scalability and Environmental Compliance: The absence of high-pressure steps makes the commercial scale-up of complex nitro-aniline derivatives inherently safer and more straightforward, facilitating a smooth transition from pilot plant to multi-ton production. The use of ethanol and water in the final purification steps creates an aqueous waste stream that is far easier to treat biologically compared to the acidic, fluoride-rich effluents of traditional methods. This environmental compatibility simplifies permitting processes and reduces the liability associated with hazardous chemical handling, making it an ideal choice for facilities operating under strict environmental regulations.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation and benefits of this advanced synthesis technology. These insights are derived directly from the experimental data and comparative analysis provided in the patent documentation to assist decision-makers in evaluating the feasibility of adoption.
Q: What are the safety advantages of this new synthesis method compared to traditional routes?
A: Unlike traditional methods that generate highly toxic hydrogen fluoride (HF) or require high-pressure reactors, this patented route operates under mild atmospheric pressure and avoids corrosive by-products, significantly enhancing operational safety.
Q: What is the expected purity and yield of the final product?
A: The process consistently delivers a total yield exceeding 80% with a final product purity of ≥98%, achieved through simple recrystallization steps without complex chromatography.
Q: Is this process suitable for large-scale commercial production?
A: Yes, the reaction conditions are mild (mostly room temperature to 100°C) and utilize common solvents like ethanol and DMF, making it highly adaptable for multi-ton scale-up in standard chemical reactors.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable N-Methyl P-Nitroaniline Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of adopting advanced synthetic methodologies to maintain competitiveness in the global fine chemicals market. Our team of expert chemists possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of patent CN101580473B are fully realized in practical manufacturing settings. We are committed to delivering products with stringent purity specifications through our rigorous QC labs, utilizing state-of-the-art analytical instrumentation to verify that every batch meets the ≥98% purity threshold required for sensitive applications in energetics and pharmaceuticals.
We invite potential partners to engage with our technical procurement team to discuss how this optimized route can enhance your supply chain stability and reduce overall manufacturing costs. By requesting a Customized Cost-Saving Analysis, you can gain a deeper understanding of the economic impact of switching to this safer, more efficient process. We encourage you to contact us today to obtain specific COA data and route feasibility assessments tailored to your specific volume requirements, ensuring a seamless integration of high-quality N-methyl p-nitroaniline into your production pipeline.
