Advanced Synthesis of N-Methylpiperazine Substituted Aniline for Pharmaceutical Manufacturing
Advanced Synthesis of N-Methylpiperazine Substituted Aniline for Pharmaceutical Manufacturing
The pharmaceutical industry continuously seeks robust and cost-efficient pathways for synthesizing critical intermediates used in cardiovascular and antiarrhythmic therapies. Patent CN101168532B introduces a refined synthetic methodology for producing N-methylpiperazine substituted aniline derivatives, which serve as pivotal building blocks for novel phenylimidazole compounds. These target molecules are increasingly recognized for their ability to modulate ion channels, specifically by inhibiting sodium and calcium influx in myocardial cells, thereby stabilizing heart rhythm. The disclosed technology addresses long-standing challenges in process chemistry by optimizing reaction conditions to enhance yield while simultaneously lowering the barrier to entry for commercial manufacturing. By leveraging accessible starting materials and streamlined purification protocols, this innovation represents a significant leap forward for reliable pharmaceutical intermediate supplier networks aiming to secure stable supply chains for high-value active pharmaceutical ingredients.
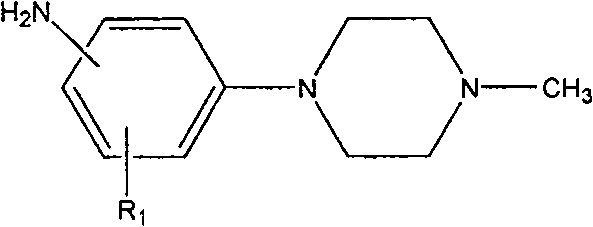
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Prior art methodologies for synthesizing these specialized aniline derivatives have been plagued by economic and operational inefficiencies that hinder large-scale adoption. For instance, Russian patent RU 2284325 describes a route utilizing tetrahydropyrrole as a solvent, which is prohibitively expensive and presents significant challenges regarding solvent recovery and recycling in an industrial setting. Furthermore, international patent WO2005007625 relies on substituted nitrofluorobenzene as a starting material; while fluorine is a good leaving group, the raw material cost is substantially higher compared to chlorinated analogs. Additionally, the use of dimethyl sulfoxide (DMSO) in these older processes creates downstream processing bottlenecks due to its high boiling point, making it energetically intensive and difficult to remove completely from the final product, often leading to lower overall yields and purity issues that complicate regulatory compliance.
The Novel Approach
The innovative strategy outlined in CN101168532B fundamentally reengineers the synthesis to prioritize cost reduction in pharmaceutical intermediate manufacturing without sacrificing quality. This novel approach substitutes the expensive nitrofluorobenzene with the much more economical substituted nitrochlorobenzene, utilizing a polar aprotic solvent system such as N,N-dimethylformamide (DMF) combined with inexpensive acid-binding agents like potassium carbonate. The process operates effectively at temperatures ranging from 80 to 140°C, facilitating a smooth nucleophilic substitution. Following the initial coupling, the subsequent hydrogenation step is optimized to proceed under moderate conditions, ensuring high conversion rates. This dual-step optimization not only drastically simplifies the workup procedure by enabling easy precipitation of the product through pH adjustment but also significantly shortens the total reaction time, making it an ideal candidate for the commercial scale-up of complex pharmaceutical intermediates.
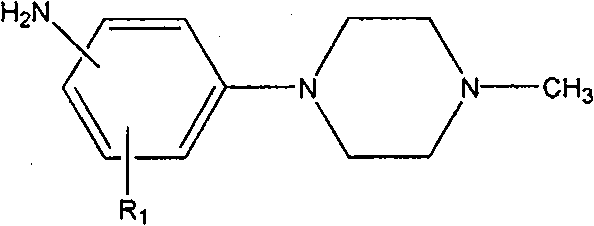
Mechanistic Insights into Nucleophilic Aromatic Substitution and Hydrogenation
The core of this synthetic route relies on a classic yet highly optimized nucleophilic aromatic substitution (SnAr) mechanism, where the electron-withdrawing nitro group activates the aromatic ring towards attack by the nucleophilic nitrogen of N-methylpiperazine. In the presence of a base such as potassium carbonate or sodium carbonate, the chloride leaving group is displaced, forming the nitro-substituted piperazine intermediate. The choice of polar aprotic solvents is critical here, as they stabilize the transition state and solvate the cationic species effectively, driving the equilibrium towards product formation. The reaction temperature is carefully controlled between 80°C and 140°C depending on the specific substituents, ensuring that the activation energy barrier is overcome while minimizing side reactions such as hydrolysis or over-alkylation, which are common pitfalls in similar heterocyclic syntheses.
Following the substitution, the nitro group is converted to the primary amine via catalytic hydrogenation, a transformation that requires precise control to maintain the integrity of the piperazine ring. Using 5% palladium on carbon (Pd/C) as the heterogeneous catalyst in a methanol solvent system allows for selective reduction of the nitro functionality under a hydrogen pressure of 5MPa. The reaction is typically conducted at temperatures between 30°C and 60°C, conditions that are mild enough to prevent catalyst poisoning or degradation of sensitive functional groups on the aromatic ring. This step is crucial for generating the high-purity aniline derivative required for downstream coupling reactions, and the use of a filterable solid catalyst simplifies the removal of metal residues, addressing a key concern for R&D directors focused on impurity profiles and heavy metal specifications in final drug substances.

How to Synthesize N-Methylpiperazine Substituted Aniline Efficiently
Implementing this synthesis requires strict adherence to the optimized parameters defined in the patent to ensure reproducibility and safety, particularly during the high-pressure hydrogenation phase. The process begins with the dissolution of the chlorinated starting material in DMF, followed by the addition of the amine and base, where temperature control is vital to manage the exotherm and drive the reaction to completion over a period of 10 to 35 hours. Once the intermediate is isolated via acid-base extraction and crystallization, it is subjected to hydrogenation in a pressure vessel. The detailed standardized synthesis steps below outline the precise operational sequence required to achieve the reported yields of up to 92% for the intermediate and 90% for the final product, providing a clear roadmap for process chemists looking to replicate this efficient route.
- Dissolve substituted nitrochlorobenzene in a polar aprotic solvent like DMF, add N-methylpiperazine and an acid-binding agent such as potassium carbonate, and react at 80-140°C.
- Perform workup by removing solvent, extracting with water and ethyl acetate, adjusting pH to precipitate the nitro-intermediate solid.
- Hydrogenate the nitro-intermediate using 5% Pd/C catalyst in methanol at 30-60°C under 5MPa hydrogen pressure to obtain the final amine product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the shift from fluorinated to chlorinated starting materials represents a strategic opportunity for substantial cost savings and supply security. The volatility of fluoro-chemical pricing often introduces uncertainty into long-term budgeting, whereas chlorinated aromatics are commodity chemicals with stable, global availability. By eliminating the need for expensive solvents like tetrahydropyrrole and difficult-to-remove high-boiling solvents like DMSO, the process inherently reduces utility costs associated with distillation and solvent recovery. This simplification of the solvent system translates directly into a leaner manufacturing process with a smaller environmental footprint, aligning with modern green chemistry initiatives while simultaneously enhancing the economic viability of the final product.
- Cost Reduction in Manufacturing: The replacement of high-cost nitrofluorobenzene with nitrochlorobenzene serves as the primary driver for raw material cost optimization, significantly lowering the bill of materials for each batch produced. Furthermore, the use of recoverable polar aprotic solvents and the avoidance of complex purification chromatography in favor of simple crystallization and filtration steps drastically reduces operational expenditures. The elimination of expensive transition metal catalysts beyond the standard palladium on carbon, and the ability to recycle the mother liquor in certain iterations, further compounds these savings, ensuring a highly competitive cost structure for bulk manufacturing.
- Enhanced Supply Chain Reliability: Sourcing substituted nitrochlorobenzenes is far less risky than sourcing specialized fluoro-derivatives, as the former are produced by numerous chemical manufacturers globally, mitigating the risk of single-source dependency. The robustness of the reaction conditions, which tolerate a range of temperatures and pressures without requiring exotic equipment, means that production can be easily transferred between different manufacturing sites or scaled up rapidly to meet surges in demand. This flexibility ensures reducing lead time for high-purity pharmaceutical intermediates, allowing partners to respond agilely to market fluctuations and clinical trial requirements without being bottlenecked by raw material shortages.
- Scalability and Environmental Compliance: The process is designed with scale-up in mind, utilizing unit operations such as filtration, phase separation, and distillation that are standard in multi-ton reactors. The workup procedure, which involves pH adjustment to precipitate the product, avoids the generation of large volumes of organic waste streams typical of column chromatography purifications. Additionally, the hydrogenation step uses a heterogeneous catalyst that can be filtered and potentially regenerated, minimizing heavy metal waste. These factors collectively simplify the environmental permitting process and reduce the cost of waste treatment, making the technology sustainable for long-term commercial production.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology, derived directly from the experimental data and comparative analysis provided in the patent documentation. Understanding these nuances is essential for technical teams evaluating the feasibility of adopting this route for their specific pipeline projects, as it clarifies the boundaries of the intellectual property and the practical expectations for yield and purity.
Q: Why is substituted nitrochlorobenzene preferred over nitrofluorobenzene in this synthesis?
A: Substituted nitrochlorobenzene is significantly more cost-effective than the corresponding nitrofluorobenzene derivatives used in prior art methods, directly reducing raw material costs without compromising reaction efficiency when optimized conditions are applied.
Q: What are the critical reaction conditions for the hydrogenation step?
A: The hydrogenation step requires precise control, utilizing 5% palladium on carbon as a catalyst in methanol solvent, maintained at temperatures between 30-60°C under a hydrogen pressure of 5MPa to ensure complete reduction of the nitro group.
Q: How does this method improve scalability compared to previous patents?
A: By avoiding high-boiling solvents like DMSO which are difficult to remove, and utilizing standard polar aprotic solvents like DMF with straightforward acid-base workup procedures, this method simplifies isolation and purification, making it highly suitable for large-scale industrial production.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable N-Methylpiperazine Substituted Aniline Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the successful development of cardiovascular therapeutics. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory bench to industrial reactor is seamless and efficient. We are committed to delivering products that meet stringent purity specifications through our rigorous QC labs, utilizing advanced analytical techniques to verify identity and assay, thereby providing our partners with the confidence needed to advance their drug candidates through clinical phases without supply interruptions.
We invite you to engage with our technical procurement team to discuss how this optimized synthesis route can be tailored to your specific project needs. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into the potential economic benefits of switching to this chlorinated pathway. We encourage you to contact us today to索取 specific COA data and route feasibility assessments, allowing us to demonstrate our capability as a strategic partner dedicated to accelerating your time-to-market through superior chemical manufacturing solutions.
