Advanced Nickel-Catalyzed Synthesis of High-Purity Agrochemical Intermediates for Commercial Scale-Up
The global demand for high-performance herbicides continues to drive innovation in the synthesis of complex aromatic intermediates, specifically within the realm of agrochemical manufacturing. Patent CN108349883B introduces a transformative methodology for the preparation of 2-alkyl-4-trifluoromethyl-3-alkylsulfonylbenzoic acids, which serve as critical building blocks for next-generation agricultural active ingredients. This technical breakthrough addresses long-standing inefficiencies in traditional synthetic routes by leveraging a robust nickel-catalyzed cyanation strategy followed by sequential functionalization. For R&D directors and procurement specialists, this patent represents a pivotal shift away from expensive palladium systems and hazardous cryogenic processes toward a more economically viable and scalable platform. The described process not only enhances the purity profile of the final intermediate but also streamlines the supply chain by utilizing readily available starting materials and minimizing waste generation. By integrating this novel approach, manufacturers can achieve substantial cost reductions while maintaining the rigorous quality standards required for regulatory approval in the agrochemical sector.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Prior art methods, such as those disclosed in WO 2008/125214 A1, rely heavily on the nucleophilic aromatic substitution of fluorine atoms, which necessitates the use of complex and difficult-to-prepare starting materials like 3-fluoro-2-methyl-4-trifluoromethylbenzoic acid. These conventional routes often require harsh conditions, including the use of at least two molar equivalents of butyllithium at cryogenic temperatures to effect metalation, followed by reaction with toxic methyl iodide to introduce the necessary alkyl groups. Such processes are inherently uneconomical due to the low yields observed, often achieving only around 50.7% of theoretical value for the methylation step, alongside significant safety hazards associated with handling pyrophoric reagents and volatile alkylating agents. Furthermore, the reliance on fluorine displacement limits the structural diversity and increases the cost of goods sold due to the premium pricing of fluorinated precursors. The accumulation of halogenated waste and the need for extensive purification to remove metal residues further complicate the manufacturing workflow, making these legacy methods unsuitable for modern, large-scale commercial production where efficiency and environmental compliance are paramount.
The Novel Approach
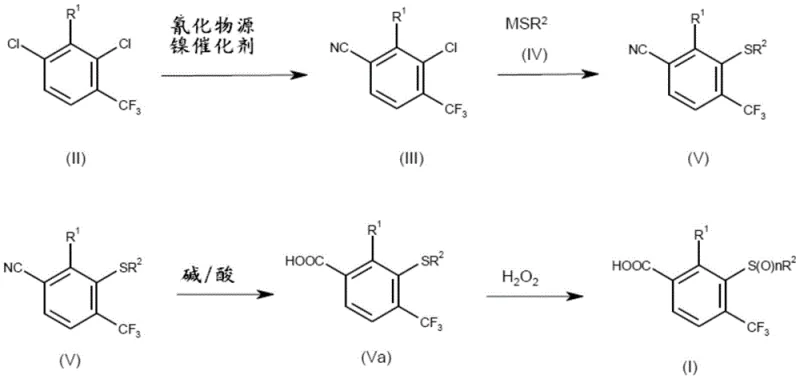
In stark contrast, the methodology outlined in CN108349883B utilizes a direct nickel-catalyzed cyanation of 1,3-dichloro-2-alkyl-4-trifluoromethylbenzene derivatives, bypassing the need for pre-functionalized fluorine substrates entirely. This innovative route begins with the conversion of readily accessible dichloro-aromatics into benzonitriles using inexpensive nickel catalysts activated by zinc powder, followed by a highly efficient thiolation step to install the sulfur moiety. The subsequent hydrolysis of the nitrile group to a carboxylic acid and final oxidation to the sulfone creates a seamless four-step sequence that operates under much milder and safer conditions compared to cryogenic lithiation. By replacing precious metal catalysts with nickel and eliminating toxic methyl iodide, this approach drastically simplifies the process flow and reduces the environmental footprint. The ability to use common solvents like acetonitrile and toluene, combined with phase transfer catalysis for the thiolation, ensures that the reaction kinetics are optimized for high throughput. This strategic redesign of the synthetic pathway offers a compelling value proposition for supply chain leaders seeking reliable sources of high-purity agrochemical intermediates without the baggage of legacy processing constraints.
Mechanistic Insights into Nickel-Catalyzed Cyanation and Functionalization
The core of this synthetic advancement lies in the sophisticated nickel-catalyzed cyanation mechanism, which facilitates the selective replacement of a chlorine atom on the aromatic ring with a cyano group. The catalytic cycle initiates with the reduction of a nickel(II) precursor, such as nickel(II) bromide, to an active nickel(0) species by metallic zinc in the presence of bulky phosphine ligands like dpephos or dppf. This active zero-valent nickel complex undergoes oxidative addition into the carbon-chlorine bond of the substrate, forming an aryl-nickel intermediate that is stabilized by the electron-rich ligand environment. Subsequent transmetallation with a cyanide source, typically sodium or potassium cyanide, introduces the cyano group to the metal center, followed by reductive elimination to release the desired benzonitrile product and regenerate the catalyst. The choice of ligand is critical, as bidentate phosphines with specific bite angles prevent the formation of inactive nickel clusters and enhance the turnover number, allowing for catalyst loadings as low as 1 to 2 mole percent. This mechanistic efficiency is further supported by the addition of small amounts of water, which has been found to accelerate the reaction rate, likely by facilitating the solubility of the inorganic cyanide salt in the organic phase.
Following the cyanation, the introduction of the sulfur functionality is achieved through a nucleophilic aromatic substitution where the remaining chlorine atom is displaced by a thiolate anion. This step is markedly improved by the inclusion of phase transfer catalysts, such as tributyltetradecylphosphonium chloride, which shuttle the thiolate species into the organic layer, thereby increasing the reaction rate and conversion efficiency at moderate temperatures between 50°C and 60°C. The final oxidation of the sulfide to the sulfone is carefully controlled using hydrogen peroxide, optionally with a tungsten-based catalyst, to ensure selective oxidation without degrading the sensitive trifluoromethyl or carboxylic acid groups. This precise control over the oxidation state is vital for maintaining the integrity of the molecule, as over-oxidation could lead to impurities that are difficult to remove. The entire sequence demonstrates a high level of chemoselectivity, ensuring that the final product meets the stringent purity specifications required for downstream coupling reactions in herbicide synthesis, thus minimizing the risk of batch failures in commercial manufacturing.
How to Synthesize 2-Alkyl-4-Trifluoromethyl-3-Alkylsulfonylbenzoic Acid Efficiently
The practical implementation of this synthesis involves a carefully orchestrated sequence of reactions that balance reactivity with safety and cost-effectiveness. The process begins with the preparation of the nickel catalyst in situ, followed by the addition of the dichloro-substrate and cyanide source in a polar aprotic solvent like acetonitrile. Once the benzonitrile intermediate is isolated, it is subjected to thiolation conditions using aqueous sodium thiomethoxide and a phase transfer catalyst, creating the thioether linkage. The nitrile group is then hydrolyzed under basic conditions using sodium hydroxide in an alcohol solvent, converting it to the corresponding carboxylic acid salt before acidification. Finally, the sulfide moiety is oxidized to the sulfone using hydrogen peroxide in a biphasic system, yielding the target benzoic acid derivative. Detailed standardized operating procedures for each of these stages, including specific temperature ramps, addition rates, and workup protocols, are essential for reproducibility and scale-up success.
- Perform nickel-catalyzed cyanation of 1,3-dichloro-2-alkyl-4-trifluoromethylbenzene using a phosphine ligand and zinc activator.
- React the resulting benzonitrile with a thiolate salt in the presence of a phase transfer catalyst to form the thioether intermediate.
- Hydrolyze the nitrile group to a carboxylic acid under basic conditions, followed by oxidation of the sulfide to a sulfone using hydrogen peroxide.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this patented technology offers profound strategic benefits that extend beyond simple chemical transformation. The shift from palladium to nickel catalysts represents a significant reduction in raw material costs, as nickel salts are orders of magnitude cheaper than their precious metal counterparts, directly impacting the cost of goods sold. Furthermore, the elimination of cryogenic steps and toxic methylating agents simplifies the infrastructure requirements for manufacturing, allowing production to occur in standard glass-lined or stainless steel reactors without the need for specialized low-temperature equipment. This operational simplicity translates to enhanced supply chain reliability, as the process is less susceptible to disruptions caused by the scarcity of specialized reagents or the logistical challenges of handling hazardous materials. The high yields reported in the patent examples suggest a robust process capable of consistent output, reducing the variability that often plagues multi-step syntheses of complex intermediates. Additionally, the reduced waste profile aligns with increasingly strict environmental regulations, lowering the costs associated with waste disposal and treatment.
- Cost Reduction in Manufacturing: The substitution of expensive palladium catalysts with economical nickel systems, combined with the avoidance of low-yielding lithiation steps, drives down the overall production cost significantly. By utilizing commodity chemicals like zinc powder and sodium cyanide instead of bespoke fluorinated starting materials, the bill of materials is optimized for maximum financial efficiency. The high atom economy of the cyanation and thiolation steps ensures that a greater proportion of input mass is converted into valuable product, minimizing waste and maximizing return on investment for every batch produced.
- Enhanced Supply Chain Reliability: The reliance on widely available industrial chemicals rather than niche fluorinated intermediates mitigates the risk of supply shortages and price volatility. The robustness of the nickel catalyst system allows for flexible sourcing of raw materials, ensuring continuity of supply even in fluctuating market conditions. Moreover, the simplified process flow reduces the number of unit operations required, shortening the manufacturing cycle time and enabling faster response to market demand spikes without compromising product quality or safety standards.
- Scalability and Environmental Compliance: The mild reaction conditions and the use of common solvents facilitate straightforward scale-up from pilot plant to commercial tonnage production. The process generates less hazardous waste compared to traditional methods, easing the burden on effluent treatment plants and ensuring compliance with global environmental standards. This sustainability advantage not only reduces operational costs but also enhances the corporate social responsibility profile of the manufacturing site, making it a preferred partner for environmentally conscious agrochemical companies.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis route. These insights are derived directly from the experimental data and claims presented in the patent documentation, providing clarity on the feasibility and advantages of the technology. Understanding these nuances is critical for technical teams evaluating the integration of this process into existing manufacturing portfolios. The answers highlight the specific operational parameters and chemical principles that underpin the success of this method.
Q: What are the primary advantages of this nickel-catalyzed method over prior art?
A: This method eliminates the need for cryogenic conditions and toxic methyl iodide required in previous fluorine-substitution routes, significantly improving yield and safety while reducing raw material costs through the use of abundant nickel catalysts instead of palladium.
Q: Which ligands are most effective for the cyanation step?
A: The patent identifies bis(2-diphenylphosphinophenyl) ether (dpephos) as very particularly preferred, along with dppf and BINAP, ensuring high catalytic activity and stability during the transformation of dichloro-aromatics to benzonitriles.
Q: How is the sulfone functionality introduced in the final step?
A: The sulfone group is generated via oxidation of the corresponding sulfide intermediate using hydrogen peroxide, optionally catalyzed by sodium tungstate, which allows for controlled oxidation without over-oxidizing other sensitive functional groups on the aromatic ring.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-Alkyl-4-Trifluoromethyl-3-Alkylsulfonylbenzoic Acid Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of efficient and scalable synthetic routes in the competitive agrochemical landscape. Our team of expert chemists possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that complex molecules like 2-alkyl-4-trifluoromethyl-3-alkylsulfonylbenzoic acids are delivered with consistent quality. We adhere to stringent purity specifications and operate rigorous QC labs to guarantee that every batch meets the exacting standards required for active pharmaceutical and agrochemical ingredient synthesis. Our commitment to technological excellence allows us to adopt advanced methodologies, such as the nickel-catalyzed processes described in CN108349883B, to provide our clients with a distinct competitive edge in terms of cost and supply security.
We invite you to engage with our technical procurement team to discuss how this innovative synthesis route can be tailored to your specific volume requirements. By requesting a Customized Cost-Saving Analysis, you can gain a clear understanding of the economic benefits of switching to this optimized process. We encourage potential partners to contact us for specific COA data and route feasibility assessments, ensuring that your supply chain is built on a foundation of scientific rigor and commercial viability. Let us be your trusted partner in navigating the complexities of fine chemical manufacturing.
