Scalable Electrooxidative Preparation of Spiro[5.5] Molecular Skeletons for Advanced Pharmaceutical Intermediates
The landscape of organic synthesis for complex molecular scaffolds is undergoing a significant transformation, driven by the urgent need for greener and more economically viable manufacturing processes. Patent CN113005473B, published in March 2022, introduces a groundbreaking methodology for the preparation of spiro[5.5] molecular skeleton compounds via electrooxidation. This technology represents a paradigm shift from traditional transition-metal-catalyzed reactions to a sustainable, electricity-driven protocol that eliminates the need for expensive metal catalysts. For R&D directors and procurement specialists in the pharmaceutical and fine chemical sectors, this innovation offers a compelling alternative for constructing highly functionalized spirocyclic cores, which are increasingly recognized as privileged structures in drug discovery. By leveraging simple electrolytes and commercially available fluorine-containing sodium methylsulfinate compounds, this method not only simplifies the reaction setup but also drastically reduces the environmental footprint associated with heavy metal waste disposal.
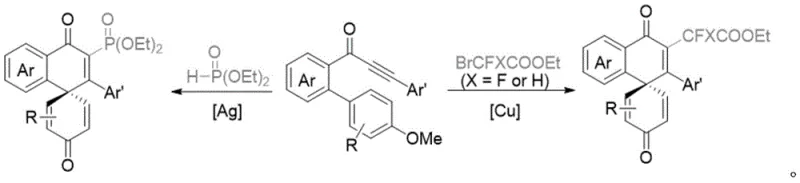
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of spiro[5.5] frameworks containing fluorine motifs has relied heavily on transition metal catalysis, typically employing salts such as silver nitrate (AgNO3) or copper(I) oxide (Cu2O) to initiate radical generation. As illustrated in the prior art reaction schemes, these conventional pathways often suffer from significant drawbacks that hinder their industrial applicability. The primary concern for supply chain managers is the high cost and volatility associated with precious metal catalysts, which can fluctuate wildly based on global market demands. Furthermore, the presence of these metals in the final product necessitates rigorous and costly purification steps to meet stringent pharmaceutical purity specifications, particularly regarding residual heavy metal limits. From an environmental compliance perspective, the generation of metal-contaminated waste streams poses a substantial liability, requiring specialized treatment protocols that increase the overall operational expenditure of the manufacturing facility.
The Novel Approach
In stark contrast to the metal-dependent legacy methods, the novel electrooxidative approach detailed in the patent utilizes electricity as the primary oxidant to drive the dearomatization of biphenyls. This catalyst-free strategy employs inexpensive fluorine-containing sodium methylsulfinate compounds as the radical source, which are oxidized at the anode to generate reactive fluorinated radicals in situ. The reaction proceeds smoothly under constant current conditions in a mixed solvent system of dichloroethane and acetonitrile, achieving yields of up to 60% without the need for external chemical oxidants or metal promoters. This fundamental change in the reaction mechanism not only lowers the raw material costs significantly but also simplifies the downstream processing workflow, as there is no requirement for metal scavenging agents. The ability to operate in an open system further enhances safety profiles by preventing the accumulation of hydrogen gas, making this a robust candidate for commercial scale-up of complex pharmaceutical intermediates.
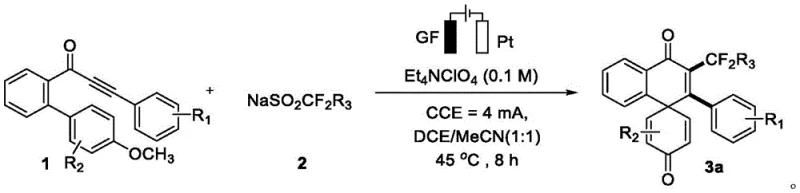
Mechanistic Insights into Electrooxidative Radical Cyclization
The mechanistic pathway of this transformation is a fascinating display of anodic oxidation driving complex bond formation. The process initiates at the anode surface, where fluorine-containing methylsulfinate ions undergo a single-electron oxidation to lose an electron, thereby generating a highly reactive fluorine-containing methyl radical species. This radical intermediate then selectively attacks the carbon atom at the alpha-position of the carbonyl alkynyl group on the biphenyl substrate, forming a thermodynamically stable vinyl radical intermediate. Subsequently, this vinyl radical undergoes a rapid 6-exo-trig cyclization onto the adjacent aromatic ring, creating a new carbon-carbon bond and establishing the spirocyclic core. The resulting cyclohexadienyl radical is then oxidized at the anode to form a carbocation, which finally undergoes deprotonation and demethylation at the cathode to restore aromaticity in the newly formed ring system, yielding the final spiro[5.5] product. This elegant cascade demonstrates how electrochemical potential can precisely control radical reactivity to achieve structural complexity that is difficult to access via thermal methods.
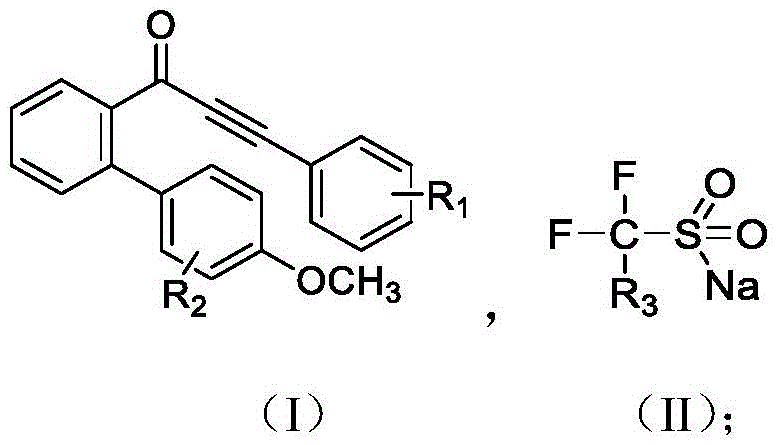
Understanding the substrate scope is critical for R&D teams evaluating the versatility of this platform. The patent specifies that the ortho-alkynylbenzoyl biphenyl substrate can tolerate a wide array of electronic environments, with R1 groups ranging from electron-withdrawing functionalities like nitro and halogens to electron-donating groups such as amino and methoxy moieties. Similarly, the fluorine source can be varied between trifluoromethyl and difluoromethyl sodium sulfinates, allowing for the modular introduction of different fluorinated motifs which are crucial for modulating the metabolic stability and lipophilicity of drug candidates. The tolerance of these diverse functional groups under mild electrochemical conditions (45°C) suggests that this method is compatible with sensitive substrates that might decompose under the harsh thermal conditions often required for traditional radical cyclizations. This broad compatibility ensures that the process can be adapted for the synthesis of a wide library of bioactive molecules without extensive re-optimization.
How to Synthesize Spiro[5.5] Compounds Efficiently
The operational simplicity of this electrochemical protocol makes it highly attractive for process chemists looking to implement new synthetic routes. The standard procedure involves dissolving the biphenyl substrate and the sodium sulfinate salt in a mixture of dichloroethane and acetonitrile containing tetraethylammonium perchlorate as the supporting electrolyte. The reaction is conducted in an undivided cell equipped with a graphite felt anode and a platinum sheet cathode, maintaining a constant current of 4 mA at 45°C for approximately 8 hours. Following the electrolysis, the workup is straightforward, involving solvent evaporation and purification via standard silica gel column chromatography.
- Combine ortho-alkynylbenzoyl biphenyl and fluorine-containing sodium methylsulfinate in a mixed solvent of dichloroethane and acetonitrile with tetraethylammonium perchlorate electrolyte.
- Perform electrooxidation using a graphite felt anode and platinum cathode at a constant current of 4 mA and temperature of 45°C for 8 hours.
- Evaporate solvents under vacuum and purify the crude residue via silica gel column chromatography to isolate the target spiro compound.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the transition from metal-catalyzed to electrochemical synthesis offers tangible strategic benefits beyond mere technical novelty. The elimination of transition metal catalysts directly translates to a reduction in raw material procurement costs, as expensive silver or copper salts are replaced by commodity chemicals and electricity. Moreover, the removal of metal residues from the final product stream significantly reduces the cost of goods sold (COGS) by eliminating the need for specialized metal scavengers and the associated validation testing for heavy metals. This streamlined purification process not only accelerates the production timeline but also minimizes the generation of hazardous solid waste, aligning with modern green chemistry principles and reducing disposal fees. The use of simple electrode materials like graphite felt and platinum sheets ensures that the capital expenditure for reactor setup remains low, while the open-system operation mitigates safety risks associated with pressurized hydrogen buildup.
- Cost Reduction in Manufacturing: The most significant economic driver for adopting this technology is the complete removal of precious metal catalysts from the bill of materials. In traditional processes, the cost of silver or copper salts, combined with the expense of ligands and metal scavengers, can constitute a substantial portion of the total production cost. By replacing these with electricity and inexpensive sodium sulfinate salts, manufacturers can achieve substantial cost savings per kilogram of product. Additionally, the simplified workup procedure reduces solvent consumption and labor hours required for purification, further enhancing the overall economic efficiency of the manufacturing process without compromising on yield or purity.
- Enhanced Supply Chain Reliability: Relying on transition metals exposes supply chains to geopolitical risks and market volatility, as seen in recent years with the fluctuation of rare earth and precious metal prices. This electrochemical method diversifies the supply risk by utilizing widely available organic salts and standard electrical infrastructure, which are far less susceptible to supply disruptions. The robustness of the reaction conditions, which tolerate a wide range of substituents and function well in common organic solvents, ensures consistent production output. This reliability is crucial for maintaining continuous supply lines for critical pharmaceutical intermediates, preventing delays that could impact downstream drug formulation and market availability.
- Scalability and Environmental Compliance: Scaling electrochemical reactions is inherently linear; increasing the electrode surface area or running multiple cells in parallel allows for seamless transition from gram-scale laboratory synthesis to multi-ton commercial production. The process operates at mild temperatures (45°C) and atmospheric pressure, reducing the energy load on heating and cooling systems compared to high-temperature thermal cyclizations. From a regulatory standpoint, the absence of heavy metals simplifies the environmental impact assessment and permits easier compliance with strict discharge regulations. This eco-friendly profile not only future-proofs the manufacturing site against tightening environmental laws but also enhances the brand value of the final product as a sustainably produced chemical entity.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this electrooxidative technology. These answers are derived directly from the experimental data and specifications outlined in the patent documentation, providing a clear overview of the method's capabilities and limitations for potential adopters. Understanding these details is essential for assessing the feasibility of integrating this route into existing manufacturing portfolios.
Q: Does this electrooxidative method require transition metal catalysts?
A: No, the process described in patent CN113005473B operates without transition metal catalysts like silver or copper, utilizing electricity to generate radicals directly from inexpensive sodium methylsulfinate salts.
Q: What is the typical yield for this spiro[5.5] synthesis?
A: The patent reports reaction yields reaching up to 60% for various substituted biphenyl substrates under optimized constant current conditions of 4 mA.
Q: Can this method introduce trifluoromethyl groups?
A: Yes, unlike some prior art methods limited to difluoro groups, this electrochemical approach successfully introduces both trifluoromethyl and difluoromethyl groups into the spiro skeleton.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Spiro[5.5] Compounds Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of electroorganic synthesis in the production of high-value pharmaceutical intermediates. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovative laboratory methods like the one described in CN113005473B can be successfully translated into robust industrial processes. Our state-of-the-art facilities are equipped with advanced electrochemical reactors and stringent purity specifications, supported by rigorous QC labs that guarantee the highest quality standards for every batch. We are committed to helping our partners navigate the complexities of modern chemical synthesis, delivering cost-effective and sustainable solutions that meet the evolving demands of the global healthcare industry.
We invite you to explore how this catalyst-free electrooxidative technology can optimize your supply chain and reduce manufacturing costs for your key intermediates. Our technical team is ready to provide a Customized Cost-Saving Analysis tailored to your specific project requirements. Please contact our technical procurement team today to request specific COA data and route feasibility assessments, and let us demonstrate how our expertise in electrochemical synthesis can add value to your product portfolio.
