Advanced Synthesis of C-Acyl Azlactones with Alpha-Quaternary Centers for Global Pharmaceutical Supply Chains
Advanced Synthesis of C-Acyl Azlactones with Alpha-Quaternary Centers for Global Pharmaceutical Supply Chains
The construction of quaternary carbon chiral centers remains one of the most formidable challenges in modern organic synthesis, particularly within the realm of bioactive drug development. Patent CN102329281A introduces a groundbreaking methodology that addresses this synthetic bottleneck through the utilization of a novel chiral bicyclic imidazole nucleophilic catalyst. This technology facilitates the asymmetric Steglich rearrangement of O-acyl azlactones to generate C-acyl azlactones bearing alpha-quaternary stereocenters with exceptional precision. For R&D directors and process chemists seeking reliable pharmaceutical intermediate supplier partnerships, this patent represents a significant leap forward in accessing complex molecular architectures that were previously difficult or economically unviable to produce. The innovation lies not just in the final product structure, but in the elegant simplicity of the catalytic system which replaces costly and cumbersome prior art methods.
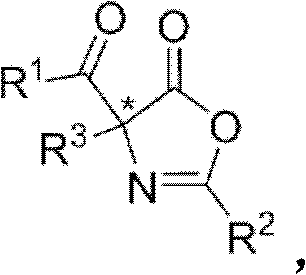
Traditionally, the establishment of quaternary carbon stereocenters has relied heavily on specialized reagents that often suffer from limited availability and exorbitant costs. Prior art methods frequently employed planar chiral 4-dimethylaminopyridine (DMAP) derivatives or central chirality DMAP catalysts, which, while effective, impose severe limitations on industrial applicability. These conventional approaches typically necessitate harsh reaction conditions, including cryogenic temperatures that demand significant energy infrastructure and specialized equipment. Furthermore, the synthesis of these legacy catalysts often involves multi-step transformations that degrade overall process efficiency and introduce impurities that are difficult to remove. For procurement managers focused on cost reduction in API manufacturing, these factors translate directly into inflated raw material costs and extended lead times, creating a supply chain vulnerability that this new technology effectively mitigates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
The reliance on traditional nucleophilic catalysts for Steglich rearrangements has long been a pain point for process development teams aiming to scale up chiral synthesis. Existing literature highlights that successful examples in this field are scarce, and those that exist often require the use of expensive chiral auxiliaries or transition metals that pose contamination risks. The necessity for low-temperature operations, often below -20°C, creates a substantial barrier to entry for standard manufacturing facilities not equipped with advanced cooling systems. Additionally, the enantioselectivity achieved by older catalyst generations was frequently insufficient for stringent pharmaceutical standards, requiring additional purification steps such as recrystallization or chiral HPLC that drastically reduce overall yield. These inefficiencies compound to create a process that is neither economically sustainable nor environmentally friendly, failing to meet the rigorous demands of modern green chemistry initiatives.
The Novel Approach
In stark contrast, the methodology disclosed in CN102329281A leverages a uniquely designed chiral bicyclic imidazole catalyst that operates under remarkably mild conditions. By utilizing a nucleophilic catalysis mechanism, this approach eliminates the need for expensive stoichiometric reagents and allows the reaction to proceed efficiently at room temperature (20°C). The catalyst itself is derived from readily available imidazole and acrolein, undergoing a straightforward addition and cyclization followed by optical resolution, which significantly lowers the barrier to entry for catalyst production. This shift from harsh, resource-intensive protocols to a streamlined, ambient-temperature process offers a compelling value proposition for supply chain heads concerned with operational continuity. The ability to achieve high enantiomeric excess (ee) values, reaching up to 95% in optimized conditions, ensures that the resulting C-acyl azlactones meet the high-purity specifications required for downstream drug synthesis without excessive waste.
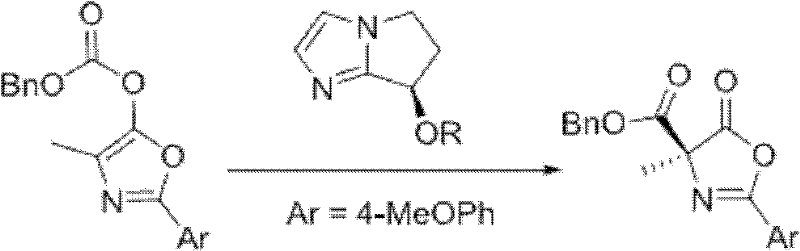
Mechanistic Insights into Chiral Bicyclic Imidazole Catalyzed Asymmetric Steglich Rearrangement
The core of this technological advancement lies in the unique structural features of the chiral bicyclic imidazole catalyst, which creates a highly defined chiral environment around the reactive center. During the asymmetric Steglich rearrangement, the nucleophilic nitrogen of the imidazole ring attacks the carbonyl carbon of the O-acyl azlactone substrate, forming a transient acyl-ammonium intermediate. This key intermediate is stabilized by the rigid bicyclic framework of the catalyst, which restricts conformational freedom and directs the subsequent migration of the acyl group to the alpha-carbon with high stereocontrol. The steric bulk provided by the substituents on the catalyst (such as the benzyloxy or naphthylmethoxy groups) plays a critical role in differentiating the enantiotopic faces of the substrate, thereby ensuring the formation of the desired enantiomer over its mirror image. Understanding this mechanistic pathway is crucial for R&D teams looking to adapt this chemistry for diverse substrate scopes, as it highlights the tunability of the catalyst for specific steric and electronic requirements.
Furthermore, the mechanism inherently suppresses the formation of common byproducts that plague traditional acylation reactions. The intramolecular nature of the rearrangement, once the acyl-ammonium species is formed, minimizes intermolecular side reactions that could lead to oligomerization or hydrolysis. This intrinsic selectivity translates to a cleaner crude reaction profile, simplifying the downstream purification process. For quality control laboratories, this means fewer impurities to track and validate, accelerating the release of materials for clinical trials. The robustness of the catalytic cycle allows for the turnover of multiple substrate equivalents without significant degradation of the catalyst's performance, a feature that is essential for maintaining consistent batch-to-batch quality in a commercial setting. The patent data indicates that varying the solvent from non-polar hexane to polar THF or toluene can fine-tune the reaction kinetics, offering process chemists additional levers to optimize yield and selectivity for specific batches.
How to Synthesize C-Acyl Azlactone Efficiently
The practical implementation of this synthesis route is designed to be accessible to standard organic synthesis laboratories while retaining the scalability required for industrial production. The process begins with the preparation of the chiral catalyst, followed by the mixing of the O-acyl azlactone substrate in a suitable organic solvent such as toluene. The reaction is conducted under an inert atmosphere to prevent moisture interference, which is a standard precaution in nucleophilic catalysis. Detailed operational parameters, including specific molar ratios and workup procedures, are critical for reproducing the high enantioselectivity reported in the patent literature. The following guide outlines the standardized synthesis steps derived from the experimental embodiments to ensure reproducibility and safety.
- Preparation of the chiral bicyclic imidazole catalyst involves the addition and cyclization of imidazole with acrolein, followed by optical resolution using (+)-tartaric acid and subsequent alkylation.
- Mix O-acyl azlactone substrate and the chiral catalyst (5-20 mol%) in an organic solvent such as toluene under inert gas protection.
- Stir the reaction mixture at 0-40°C for 2-10 days, then quench with dilute acid, extract with organic solvent, and purify via column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain strategists, the adoption of this catalytic technology offers profound advantages that extend beyond mere chemical yield. The shift away from cryogenic conditions and expensive chiral reagents fundamentally alters the cost structure of producing these valuable intermediates. By enabling reactions to run at ambient temperatures, manufacturers can significantly reduce energy consumption associated with cooling systems, leading to a lower carbon footprint and reduced utility costs. The simplicity of the catalyst synthesis, which avoids precious metals or rare earth elements, insulates the supply chain from the volatility often seen in the market for specialized catalytic materials. This stability is crucial for long-term planning and ensures that production schedules are not disrupted by the sudden unavailability of key reagents.
- Cost Reduction in Manufacturing: The elimination of expensive stoichiometric chiral auxiliaries and the replacement of complex multi-step catalyst syntheses with a streamlined route results in substantial cost savings. The use of common solvents like toluene and the ability to operate at room temperature removes the need for specialized low-temperature reactors, allowing existing infrastructure to be utilized more effectively. This reduction in capital expenditure and operational complexity directly contributes to a more competitive pricing model for the final pharmaceutical intermediate, enhancing margins for downstream partners.
- Enhanced Supply Chain Reliability: The robustness of the chiral bicyclic imidazole catalyst ensures consistent performance across different batches, reducing the risk of failed runs that can delay product delivery. Since the catalyst components are derived from commodity chemicals like imidazole and acrolein, the supply chain is less susceptible to the bottlenecks that affect niche reagent markets. This reliability allows for more accurate forecasting and inventory management, ensuring that critical API intermediates are available when needed for formulation and clinical testing phases.
- Scalability and Environmental Compliance: The process generates minimal hazardous waste compared to traditional methods that rely on heavy metal catalysts or toxic solvents. The high atom economy of the rearrangement reaction means that a larger proportion of the starting material ends up in the final product, reducing the volume of waste that requires treatment and disposal. This alignment with green chemistry principles not only simplifies regulatory compliance but also enhances the corporate sustainability profile of the manufacturing entity, a factor increasingly weighted in vendor selection processes by major pharmaceutical companies.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this asymmetric Steglich rearrangement technology. These answers are derived directly from the experimental data and technical specifications outlined in the patent documentation, providing a clear understanding of the method's capabilities and limitations. Understanding these details is essential for technical teams evaluating the feasibility of integrating this route into their existing manufacturing pipelines.
Q: What are the primary advantages of this chiral bicyclic imidazole catalyst over traditional DMAP derivatives?
A: Unlike traditional planar chiral DMAP catalysts which often require harsh cryogenic conditions and complex multi-step syntheses, this novel bicyclic imidazole catalyst operates efficiently at room temperature (20°C) and offers superior enantioselectivity (up to 95% ee) with a simpler preparation route.
Q: Is this process suitable for large-scale commercial production of pharmaceutical intermediates?
A: Yes, the process utilizes common organic solvents like toluene and avoids extreme temperatures, making it highly scalable. The elimination of expensive reagents and the robustness of the catalyst support reliable commercial scale-up for complex API intermediates.
Q: What types of substrates are compatible with this asymmetric Steglich rearrangement?
A: The method demonstrates broad substrate scope, successfully converting various O-acyl azlactones with different aryl (e.g., 4-MeOPh, Ph) and alkyl groups (e.g., Methyl, Benzyl, Ethyl, Isobutyl) into C-acyl isomers with high yields and excellent stereocontrol.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable C-Acyl Azlactone Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of high-quality chiral intermediates in the development of next-generation therapeutics. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory discovery to full-scale manufacturing is seamless and efficient. We adhere to stringent purity specifications and utilize rigorous QC labs to guarantee that every batch of C-acyl azlactone meets the exacting standards required by global regulatory bodies. Our commitment to technical excellence allows us to navigate the complexities of chiral synthesis, delivering products that empower our clients to accelerate their drug development timelines with confidence.
We invite potential partners to engage with our technical procurement team to discuss how this innovative catalytic technology can be tailored to your specific project needs. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into how optimizing your synthesis route can improve your bottom line. We encourage you to contact us today to obtain specific COA data and route feasibility assessments, ensuring that your supply chain is built on a foundation of reliability, quality, and scientific innovation.
