Advanced One-Step Synthesis of Polysubstituted Pyridine Derivatives for Commercial Scale-up
Advanced One-Step Synthesis of Polysubstituted Pyridine Derivatives for Commercial Scale-up
The pharmaceutical and fine chemical industries are constantly seeking more efficient routes to access privileged heterocyclic scaffolds, particularly pyridine derivatives which serve as critical building blocks for numerous active pharmaceutical ingredients (APIs). A significant technological breakthrough in this domain is detailed in Chinese Patent CN112920111B, which discloses a novel, copper-catalyzed one-step cyclization method. This innovation allows for the direct construction of polysubstituted pyridine rings from alpha-thiocarbonyl-N,S-ketals and isocyanoacetates. By leveraging this advanced synthetic strategy, manufacturers can overcome the limitations of classical approaches, achieving higher efficiency and better control over substitution patterns. The versatility of this method supports the generation of diverse structures, making it an invaluable tool for modern drug discovery and process chemistry teams aiming to accelerate their pipelines.
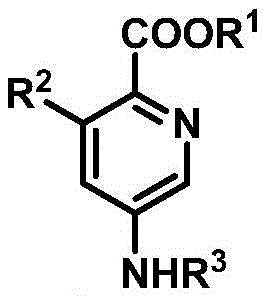
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of substituted pyridines has relied heavily on classical condensation reactions such as the Hantsch dihydropyridine synthesis or Chichibabin pyridine synthesis. These traditional pathways often suffer from significant drawbacks that hinder their utility in modern, high-throughput process development. Typically, they require multi-step sequences involving the preparation of highly functionalized precursors, which increases both the time and cost of production. Furthermore, these methods frequently necessitate harsh reaction conditions, including high temperatures and strong acids or bases, which can be incompatible with sensitive functional groups often present in complex drug candidates. The lack of regioselectivity in many conventional routes also leads to the formation of difficult-to-separate isomeric mixtures, complicating purification and reducing overall yield, thereby impacting the economic feasibility of large-scale manufacturing.
The Novel Approach
In stark contrast to these legacy methods, the technology described in patent CN112920111B introduces a streamlined, one-pot cyclization strategy that fundamentally reshapes the synthetic landscape for pyridine derivatives. This novel approach utilizes alpha-thiocarbonyl-N,S-ketals as versatile synthons which react directly with isocyanoacetates in the presence of a copper salt promoter. The reaction proceeds under remarkably mild conditions, typically in toluene at temperatures ranging from 80-110°C, avoiding the extreme thermal stress associated with older techniques. This methodology not only simplifies the operational workflow by eliminating intermediate isolation steps but also offers exceptional control over the substitution pattern on the pyridine ring. The ability to construct the core heterocycle in a single step from readily available starting materials represents a paradigm shift towards more sustainable and cost-effective chemical manufacturing.
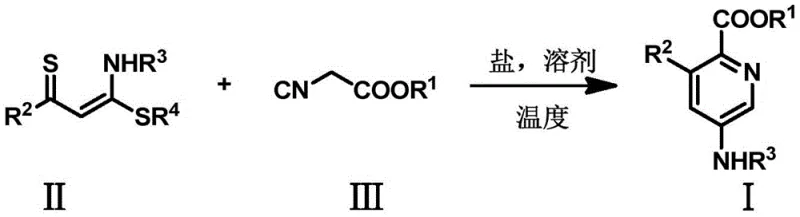
Mechanistic Insights into Copper-Catalyzed Cyclization
The efficacy of this synthetic route relies on the unique reactivity of the alpha-thiocarbonyl-N,S-ketal substrate in conjunction with the copper catalyst system. Mechanistically, the copper salt, preferably copper(II) bromide (CuBr2), acts as a Lewis acid promoter that likely activates the nitrile group of the isocyanoacetate or facilitates the cleavage of the carbon-sulfur bond in the ketal. This activation enables a nucleophilic attack that initiates the ring-closing sequence. The presence of a strong base, such as lithium tert-butoxide (tBuOLi), is crucial for deprotonating the active methylene species, generating a nucleophile capable of attacking the electrophilic centers. This synergistic interaction between the copper promoter and the base ensures a smooth progression through the transition states required for aromatization, ultimately yielding the stable pyridine core with high fidelity and minimal side reactions.
From an impurity control perspective, this mechanism offers distinct advantages for producing high-purity pharmaceutical intermediates. The specificity of the copper-catalyzed pathway minimizes the formation of polymeric byproducts or alternative cyclization isomers that often plague non-catalytic thermal reactions. The mild basic conditions prevent the hydrolysis of sensitive ester groups (such as the ethyl ester moiety shown in the examples), preserving the integrity of the molecule throughout the synthesis. Additionally, the reaction demonstrates broad functional group tolerance, accommodating various aryl and heteroaryl substituents without significant degradation. This robustness translates to a cleaner reaction profile, reducing the burden on downstream purification processes like column chromatography or recrystallization, which is essential for maintaining cost efficiency in commercial production.
How to Synthesize Polysubstituted Pyridine Derivatives Efficiently
Implementing this synthesis in a laboratory or pilot plant setting requires careful attention to reagent stoichiometry and atmospheric conditions to maximize yield and reproducibility. The protocol generally involves charging a reaction vessel with the alpha-thiocarbonyl-N,S-ketal and the isocyanoacetate in an aprotic solvent, with toluene being the preferred medium due to its boiling point and solubility profile. The addition of the copper catalyst and base must be managed under an inert atmosphere, such as nitrogen or argon, to prevent oxidation of sensitive intermediates. While the patent outlines specific molar ratios, optimizing these parameters for specific substrates can further enhance performance. For a comprehensive guide on the exact operational parameters and safety considerations, please refer to the standardized synthesis steps provided below.
- Prepare the reaction mixture by combining alpha-thiocarbonyl-N,S-ketal and isocyanoacetate in an aprotic solvent like toluene.
- Add a copper salt promoter such as CuBr2 and a strong base like lithium tert-butoxide under an inert atmosphere.
- Heat the mixture to 80-110°C for 5-12 hours to facilitate cyclization, followed by standard workup and purification.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this copper-catalyzed methodology presents a compelling value proposition centered on cost optimization and supply reliability. The primary starting materials, specifically the alpha-thiocarbonyl-N,S-ketals, are structurally diverse yet commercially accessible, reducing the risk of raw material bottlenecks. Unlike processes dependent on scarce precious metals like palladium or rhodium, this method utilizes abundant and inexpensive copper salts, which drastically lowers the bill of materials. Furthermore, the simplified one-step nature of the reaction reduces labor hours, energy consumption, and solvent usage, contributing to a leaner manufacturing footprint. These factors collectively drive down the cost of goods sold (COGS), allowing for more competitive pricing strategies in the global market for pharmaceutical intermediates.
- Cost Reduction in Manufacturing: The elimination of multi-step sequences and the use of low-cost copper catalysts significantly reduce production expenses. By avoiding expensive transition metals and complex purification protocols associated with traditional methods, manufacturers can achieve substantial cost savings. The high atom economy of the cyclization reaction ensures that a greater proportion of raw materials are converted into the final product, minimizing waste disposal costs and maximizing resource efficiency in API manufacturing.
- Enhanced Supply Chain Reliability: The robustness of this synthetic route enhances supply chain stability by reducing dependency on specialized reagents that may have long lead times. The reaction conditions are mild and tolerant to variations, which ensures consistent batch-to-batch quality even when scaling up. This reliability is critical for maintaining continuous production schedules and meeting the stringent delivery deadlines required by downstream pharmaceutical clients, effectively reducing lead time for high-purity intermediates.
- Scalability and Environmental Compliance: The process is inherently scalable, having been demonstrated to work efficiently in standard organic solvents like toluene which are well-understood in industrial settings. The absence of hazardous reagents and the generation of minimal toxic byproducts align with green chemistry principles, simplifying environmental compliance and waste treatment. This ease of scale-up facilitates the commercial production of complex heterocycles from kilogram to tonne scales without requiring significant capital investment in new reactor infrastructure.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented synthesis technology. Understanding these details is crucial for R&D teams evaluating process feasibility and procurement officers assessing supplier capabilities. The answers are derived directly from the experimental data and technical specifications outlined in the patent documentation, ensuring accuracy and relevance for decision-making purposes.
Q: What are the key advantages of this copper-catalyzed method over traditional Hantsch synthesis?
A: Unlike the traditional Hantsch method which often requires multi-step sequences and harsh conditions, this patented approach constructs the pyridine ring in a single step. It utilizes readily available alpha-thiocarbonyl-N,S-ketals, operates under milder temperatures (80-110°C), and demonstrates superior functional group tolerance, significantly simplifying the process for complex pharmaceutical intermediates.
Q: Is this synthesis method suitable for large-scale industrial production?
A: Yes, the method is highly amenable to scale-up. The use of inexpensive copper salts instead of precious metal catalysts reduces raw material costs. Furthermore, the reaction employs common solvents like toluene and achieves high yields (up to 90%), making it economically viable for commercial scale-up of complex heterocycles without requiring specialized high-pressure equipment.
Q: How does this method impact the purity and impurity profile of the final pyridine derivative?
A: The reaction exhibits excellent stereoselectivity and regioselectivity, which inherently limits the formation of structural isomers and side products. The mild reaction conditions prevent the degradation of sensitive functional groups, resulting in a cleaner crude reaction mixture. This simplifies downstream purification, ensuring the delivery of high-purity pyridine derivatives essential for drug development.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Polysubstituted Pyridine Derivative Supplier
At NINGBO INNO PHARMCHEM, we recognize the strategic importance of efficient synthetic routes like the one described in CN112920111B for accelerating drug development timelines. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project transitions smoothly from benchtop discovery to full-scale manufacturing. Our state-of-the-art facilities are equipped with rigorous QC labs capable of meeting stringent purity specifications, guaranteeing that every batch of polysubstituted pyridine derivative delivered meets the highest industry standards for quality and consistency.
We invite you to collaborate with us to leverage this advanced technology for your specific project needs. Our technical team is ready to provide a Customized Cost-Saving Analysis tailored to your target molecule, demonstrating how this copper-catalyzed route can optimize your budget. Please contact our technical procurement team today to request specific COA data and route feasibility assessments, and let us help you secure a reliable supply chain for your critical pharmaceutical intermediates.
