Scalable Total Synthesis of (-)-Newbouldine: A Breakthrough in Green Catalytic N-N Bond Formation
The pharmaceutical and fine chemical industries are constantly seeking more efficient pathways to access complex natural products, particularly those exhibiting unique biological activities. Patent CN109956948B, published in mid-2021, introduces a groundbreaking synthetic methodology for the total synthesis of (-)-newbouldine, a rare alkaloid containing a distinctive N-N bond within a penta-bicyclic skeleton. This natural product, originally isolated from Newbouldia laevis, has demonstrated significant neurophysiological activity, making it a high-value target for drug discovery programs. The disclosed technology represents a paradigm shift from traditional stoichiometric reductions to a modern, catalytic approach, specifically utilizing iron-mediated cyclization. For R&D directors and procurement managers alike, this patent offers a compelling value proposition: a concise five-step sequence that bypasses the severe safety hazards and environmental burdens associated with prior art, thereby establishing a new benchmark for the reliable (-)-newbouldine supplier market.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the total synthesis of (-)-newbouldine has been plagued by significant operational and safety challenges that hinder commercial viability. The pioneering work by the Trauner research group in 2010, while chemically elegant, relies heavily on the use of cuprous cyanide (CuCN) complexed with lithium chloride to facilitate conjugate addition. The preparation of this reagent alone requires harsh conditions, including heating at 100°C under high vacuum for 24 hours, posing substantial risks due to the extreme toxicity of cyanide species. Furthermore, the final ring-closing step in this legacy route demands ten equivalents of titanium trichloride (TiCl3) and three equivalents of sodium methoxide. This stoichiometric reliance on titanium generates massive amounts of metal salt waste, complicating downstream purification and wastewater treatment. Similarly, the racemic synthesis reported by Pla and Tan in 2014 utilizes high-energy ultraviolet light (254 nm) for photocyclization and requires refluxing in concentrated hydrochloric acid. These conditions are not only energy-intensive but also corrosive to standard reactor equipment, leading to increased maintenance costs and potential supply chain disruptions due to the handling of hazardous materials.
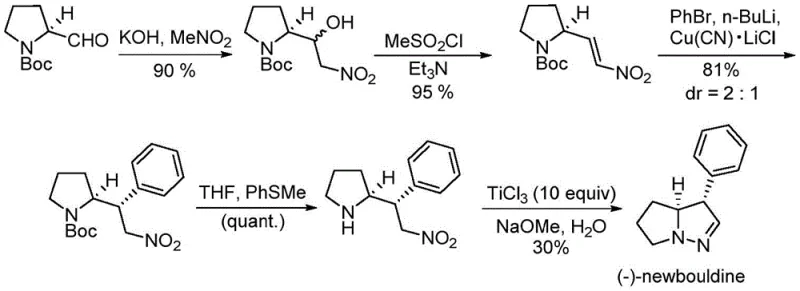
The Novel Approach
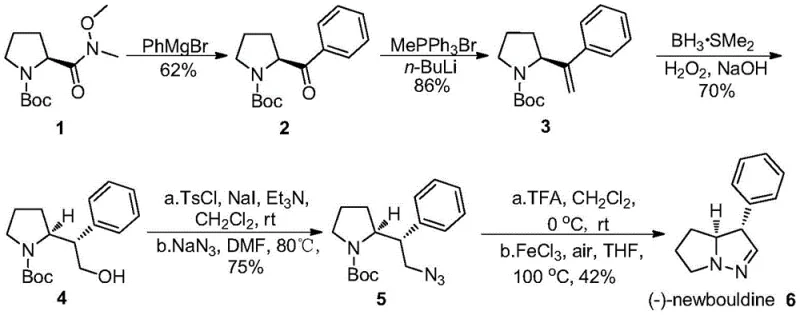
In stark contrast to these cumbersome legacy methods, the invention disclosed in CN109956948B presents a streamlined, five-step linear synthesis that prioritizes atom economy and operator safety. Starting from commercially available, optically active Boc-protected pyrrolidinecarboxamide, the route employs a logical sequence of Grignard addition, Wittig olefination, hydroboration-oxidation, azidation, and finally, an iron-catalyzed N-N bond formation. The most transformative aspect of this novel approach is the final cyclization step, which replaces the ten equivalents of toxic TiCl3 with a mere catalytic loading (0.02 to 0.05 equivalents) of anhydrous ferric chloride (FeCl3). This drastic reduction in reagent loading not only simplifies the workup procedure—eliminating the need for extensive filtration of titanium sludge—but also aligns with green chemistry principles by minimizing heavy metal contamination. The entire pathway operates under relatively mild thermal conditions, avoiding the need for cryogenic temperatures or high-energy photon sources, thus offering a robust platform for the cost reduction in pharmaceutical intermediate manufacturing.
Mechanistic Insights into FeCl3-Catalyzed N-N Cyclization
The core innovation of this patent lies in the mechanistic elegance of the final ring-closure transformation. Unlike the reductive cyclization mechanisms seen in prior art which consume vast amounts of reducing agents, the iron-catalyzed process likely proceeds via a single-electron transfer (SET) radical mechanism. Upon deprotection of the Boc group using trifluoroacetic acid, the free amine and the pendant azide moiety are positioned for intramolecular interaction. The catalytic FeCl3 acts as a Lewis acid and potentially a single-electron oxidant, facilitating the activation of the azide group and promoting the formation of the nitrogen-nitrogen bond to close the pyrazoline ring. This catalytic cycle allows the iron species to turnover multiple times, explaining the high efficiency observed with loadings as low as 2 mol%. From a purity perspective, this mechanism is superior because it avoids the generation of stoichiometric byproducts that often co-elute with the product or form difficult-to-remove complexes. The use of a chiral pool starting material ensures that the stereochemistry at the C-3 position is preserved throughout the sequence, while the hydroboration step installs the second chiral center with high diastereocontrol, resulting in a final product with defined optical rotation ([α]D 20: -52).
Furthermore, the impurity profile of this process is significantly cleaner compared to the TiCl3-mediated route. In the conventional method, the excess titanium reagents often lead to over-reduction or non-specific degradation of the sensitive hydrazone functionality. By utilizing a mild iron catalyst in tetrahydrofuran at moderate temperatures (90-110°C in a sealed tube), the reaction specificity is enhanced. The patent data indicates that side reactions are minimized, allowing for straightforward purification via standard silica gel chromatography. This level of control is critical for producing high-purity (-)-newbouldine suitable for biological testing, where trace metal impurities could confound assay results. The robustness of the FeCl3 system also suggests a wider operating window for scale-up, as the reaction is less sensitive to minor fluctuations in temperature or reagent addition rates compared to the highly exothermic and moisture-sensitive TiCl3 reductions.
How to Synthesize (-)-Newbouldine Efficiently
The synthesis of (-)-newbouldine described in this patent offers a practical blueprint for laboratory and pilot-scale production. The process begins with the conversion of a Weinreb amide derivative to a ketone, followed by chain extension and functional group manipulation to set up the cyclization precursor. Each step has been optimized for yield and ease of handling, utilizing common reagents such as phenylmagnesium bromide, borane-dimethyl sulfide, and sodium azide. The detailed standardized synthetic steps below outline the specific molar ratios, solvent choices, and workup procedures required to achieve the reported yields of 62% to 86% in the intermediate steps and 42% in the final cyclization. This protocol is designed to be reproducible and scalable, providing a clear path from raw materials to the final active alkaloid.
- Perform a Grignard reaction on Boc-protected pyrrolidine Weinreb amide with PhMgBr to form the ketone intermediate.
- Execute a Wittig olefination using methyltriphenylphosphonium bromide and n-BuLi to generate the terminal alkene.
- Conduct stereoselective hydroboration-oxidation with BH3*SMe2 followed by H2O2/NaOH to install the primary alcohol.
- Convert the alcohol to an azide via tosylation and subsequent nucleophilic substitution with sodium azide.
- Deprotect the Boc group and perform intramolecular N-N cyclization using catalytic anhydrous FeCl3 to yield (-)-newbouldine.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this synthetic route translates directly into tangible operational benefits and risk mitigation. The most immediate advantage is the elimination of cuprous cyanide from the supply chain. Sourcing and handling cyanide salts require specialized permits, dedicated storage facilities, and rigorous safety protocols, all of which add significant overhead costs. By removing this bottleneck, manufacturers can streamline their raw material sourcing and reduce regulatory compliance burdens. Additionally, the shift from stoichiometric titanium trichloride to catalytic iron chloride drastically reduces the volume of hazardous waste generated per kilogram of product. This reduction in waste disposal costs, combined with the simplified aqueous workups that replace complex metal filtrations, contributes to substantial cost savings in the overall manufacturing process. The mild reaction conditions also imply lower energy consumption, as there is no need for prolonged heating under high vacuum or the operation of high-power UV lamps.
- Cost Reduction in Manufacturing: The economic impact of this technology is driven by the replacement of expensive and hazardous reagents with commodity chemicals. Titanium trichloride is not only costly but generates large volumes of solid waste that require expensive disposal. In contrast, ferric chloride is an abundant, low-cost industrial chemical. Using it in catalytic quantities (0.02-0.05 equiv) means the reagent cost for the key step becomes negligible. Furthermore, the high yields in the early steps (e.g., 86% for the Wittig reaction) ensure that valuable chiral starting materials are not wasted, maximizing the return on investment for every batch produced. The simplified purification processes also reduce the consumption of solvents and silica gel, further driving down the cost of goods sold (COGS).
- Enhanced Supply Chain Reliability: Reliance on specialty reagents like freshly prepared CuCN/LiCl complexes introduces fragility into the supply chain. Any disruption in the availability of these specific catalysts can halt production entirely. The new route relies on stable, shelf-stable reagents such as PhMgBr, BH3*SMe2, and TsCl, which are readily available from multiple global suppliers. This diversification of the supply base ensures continuity of supply and protects against price volatility. Moreover, the avoidance of corrosive concentrated hydrochloric acid reflux conditions extends the lifespan of reactor vessels and piping, reducing capital expenditure on equipment maintenance and replacement. This reliability makes the process ideal for long-term commercial contracts where consistent delivery is paramount.
- Scalability and Environmental Compliance: Scaling chemical processes often exposes hidden safety and environmental issues, but this route is inherently designed for expansion. The absence of high-energy photochemistry removes the scaling limitations associated with light penetration depth in large reactors. Similarly, the exothermic nature of the Grignard and hydroboration steps is manageable with standard cooling systems, unlike the violent reactions sometimes seen with bulk TiCl3. From an environmental standpoint, the process aligns with increasingly strict global regulations on heavy metal discharge. By minimizing titanium and eliminating copper/cyanide waste, the facility's environmental footprint is significantly reduced, facilitating easier permitting and fostering a positive corporate sustainability profile. This 'green' credential is increasingly valued by downstream pharmaceutical partners who are auditing their suppliers for ESG compliance.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production of (-)-newbouldine using this patented methodology. These answers are derived directly from the experimental data and comparative analysis provided in the patent documentation, offering clarity on the process capabilities and limitations. Understanding these details is essential for technical teams evaluating the feasibility of integrating this intermediate into their broader drug development pipelines.
Q: How does this new synthesis method improve safety compared to previous routes?
A: Unlike the Trauner route which requires highly toxic cuprous cyanide (CuCN) and stoichiometric titanium trichloride, this novel method utilizes a catalytic amount of iron(III) chloride (0.02-0.05 equiv). This significantly reduces heavy metal waste and eliminates the need for handling extremely hazardous cyanide reagents, enhancing overall process safety.
Q: What is the stereochemical outcome of this synthetic route?
A: The synthesis starts from optically active Boc-protected pyrrolidinecarboxamide, ensuring high stereocontrol throughout the sequence. The hydroboration-oxidation step creates a new chiral center with high diastereoselectivity, ultimately yielding the levorotatory (-)-newbouldine enantiomer, avoiding the racemic mixtures produced by photochemical methods.
Q: Is this process suitable for large-scale commercial manufacturing?
A: Yes, the process is designed for scalability. It avoids harsh conditions such as high-vacuum preparation of catalysts or high-energy UV irradiation (254 nm). The reactions proceed under mild temperatures (mostly 0°C to 100°C) and utilize standard workup procedures like aqueous quenching and column chromatography, making it viable for kilogram-to-ton scale production.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable (-)-Newbouldine Supplier
At NINGBO INNO PHARMCHEM, we recognize the strategic importance of accessing complex natural product scaffolds like (-)-newbouldine for advanced medicinal chemistry programs. As a premier CDMO partner, we possess the technical expertise to translate the innovative FeCl3-catalyzed route described in CN109956948B from benchtop discovery to commercial reality. Our facilities are equipped to handle the specific requirements of this synthesis, including inert atmosphere operations for Grignard reactions and sealed tube heating for the final cyclization. We bring extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with consistency and precision. Our commitment to quality is underpinned by stringent purity specifications and rigorous QC labs that utilize state-of-the-art analytical instrumentation to verify the stereochemical integrity and chemical purity of every batch.
We invite you to collaborate with us to leverage this cutting-edge synthetic technology for your projects. By partnering with NINGBO INNO PHARMCHEM, you gain access to a Customized Cost-Saving Analysis tailored to your specific volume requirements, demonstrating how this greener route can optimize your budget. We encourage potential partners to contact our technical procurement team to request specific COA data from our pilot runs and to discuss detailed route feasibility assessments. Let us help you secure a sustainable and cost-effective supply of high-purity (-)-newbouldine, accelerating your path from research to market.
