Advanced Organocatalytic Strategy for High-Purity Chiral Hydrazone Manufacturing and Commercial Scale-Up
Advanced Organocatalytic Strategy for High-Purity Chiral Hydrazone Manufacturing and Commercial Scale-Up
The pharmaceutical and agrochemical industries are constantly seeking robust, scalable, and environmentally benign synthetic routes for complex chiral intermediates. A significant breakthrough in this domain is detailed in patent CN115433108A, which discloses a novel method for synthesizing chiral hydrazone compounds containing trifluoromethyl groups. This technology represents a paradigm shift from traditional metal-dependent catalysis to efficient organocatalysis, utilizing N-ester acylhydrazones and beta-trifluoromethyl-alpha,beta-unsaturated ketones as readily available starting materials. The core innovation lies in the use of a urea-tertiary amine catalyst derived from L-tert-leucine, which facilitates the reaction under remarkably mild conditions at 30°C. For R&D directors and procurement managers alike, this development signals a new era of cost reduction in pharmaceutical intermediate manufacturing, offering a pathway to high-value chiral building blocks without the burden of heavy metal residues or harsh reaction environments.
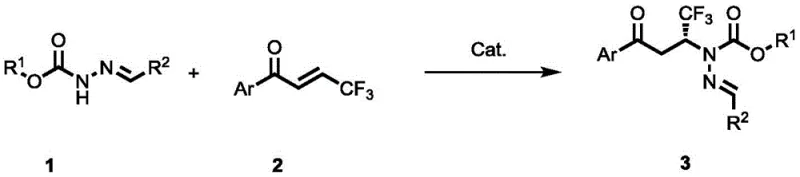
The strategic importance of this patent cannot be overstated for companies aiming to secure a reliable pharmaceutical intermediate supplier status. The introduction of the trifluoromethyl group into organic molecules is known to significantly enhance metabolic stability, lipophilicity, and bioavailability, making these motifs highly desirable in modern drug design. However, controlling the stereochemistry during the formation of the hydrazone linkage has historically been challenging. The method described in CN115433108A overcomes these hurdles by employing a bifunctional organocatalyst that activates both the nucleophile and the electrophile through a network of hydrogen bonds. This dual activation strategy ensures high regioselectivity and exceptional enantiocontrol, yielding products with enantiomeric excess values reaching up to 98% ee. Such high purity is critical for meeting the stringent regulatory requirements of global health authorities and minimizing the risk of toxic impurities in the final active pharmaceutical ingredients.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of chiral hydrazones and related heterocyclic precursors often relied heavily on transition metal catalysis or the use of stoichiometric chiral auxiliaries. These conventional approaches frequently suffer from significant drawbacks that impact both the economic viability and the environmental footprint of the manufacturing process. Transition metal catalysts, while effective, introduce the risk of heavy metal contamination, necessitating expensive and time-consuming purification steps such as scavenging or recrystallization to meet ppm-level limits. Furthermore, many traditional protocols require harsh reaction conditions, including elevated temperatures, strong bases, or inert atmospheres, which increase energy consumption and operational complexity. The use of stoichiometric oxidants also generates substantial chemical waste, complicating waste management and increasing the overall cost of goods sold. For supply chain heads, these factors translate into longer lead times, higher raw material costs, and increased vulnerability to supply disruptions of specialized metal catalysts.
The Novel Approach
In stark contrast, the novel approach outlined in the patent utilizes a metal-free organocatalytic system that operates efficiently under air atmosphere at a mild temperature of 30°C. By leveraging a chiral urea-tertiary amine catalyst, specifically derivative C5, the reaction achieves high conversion rates and excellent stereoselectivity without the need for external oxidants or sensitive metal complexes. This methodology drastically simplifies the post-treatment process; since no metals are involved, the purification workflow is streamlined to basic column chromatography or crystallization, significantly reducing processing time and solvent usage. The robustness of the catalyst allows the reaction to proceed in common organic solvents like toluene, which are inexpensive and easy to recover. This shift towards organocatalysis not only aligns with green chemistry principles but also offers a tangible competitive advantage in terms of production speed and cost efficiency, making it an ideal candidate for the commercial scale-up of complex pharmaceutical intermediates.
Mechanistic Insights into Urea-Tertiary Amine Catalyzed Michael Addition
The success of this synthetic route hinges on the sophisticated design of the L-tert-leucine derived urea-tertiary amine catalyst, which acts as a bifunctional activator. Mechanistically, the tertiary amine moiety of the catalyst deprotonates the N-ester acylhydrazone to generate a reactive nucleophilic species, while the urea functionality simultaneously activates the beta-trifluoromethyl-alpha,beta-unsaturated ketone through double hydrogen bonding with the carbonyl oxygen. This precise spatial arrangement brings the reactants into close proximity within a well-defined chiral environment created by the bulky tert-butyl group of the leucine backbone. The rigid chiral pocket effectively shields one face of the electrophile, forcing the nucleophilic attack to occur from the less hindered side, thereby dictating the absolute configuration of the newly formed stereocenter. This synergistic activation lowers the activation energy of the Michael addition, allowing the reaction to proceed rapidly even at ambient temperatures.
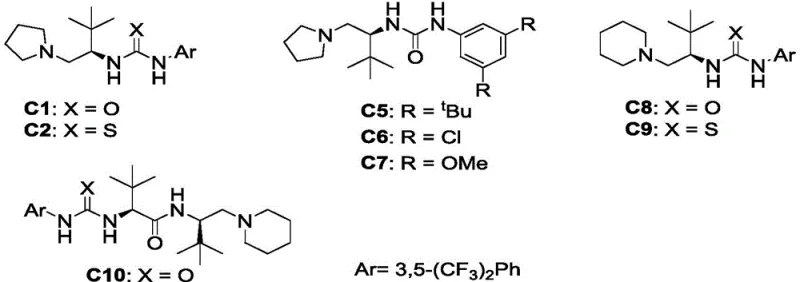
From an impurity control perspective, this mechanism offers distinct advantages over radical-based or metal-mediated pathways. The absence of single-electron transfer processes minimizes the formation of radical coupling byproducts, which are often difficult to separate. Additionally, the high specificity of the hydrogen-bonding network reduces the likelihood of non-selective background reactions that could lead to racemic mixtures. The patent data demonstrates that varying the substituents on the catalyst aromatic rings (such as the 3,5-bis(trifluoromethyl)phenyl group in C5) further tunes the acidity of the urea protons and the steric bulk, optimizing the balance between reactivity and selectivity. For R&D teams, understanding this mechanistic nuance is vital for troubleshooting and adapting the process to diverse substrate scopes, ensuring consistent quality across different batches of high-purity OLED material or pharmaceutical precursors.
How to Synthesize Chiral Hydrazone Compounds Efficiently
The practical implementation of this technology is straightforward and designed for ease of adoption in standard chemical manufacturing facilities. The protocol involves dissolving the N-ester acylhydrazone and the optimized catalyst C5 in a suitable solvent such as toluene, followed by the addition of the trifluoromethylated enone. The reaction mixture is then stirred at a controlled temperature of 30°C under air, monitored by TLC until the starting material is fully consumed. This operational simplicity reduces the need for specialized equipment like gloveboxes or high-pressure reactors, lowering the barrier to entry for contract manufacturing organizations. The detailed standardized synthesis steps, including specific molar ratios and workup procedures, are provided below to facilitate immediate process validation.
- Dissolve N-ester acylhydrazone and the L-tert-leucine derived urea-tertiary amine catalyst (such as C5) in an organic solvent like toluene.
- Add beta-trifluoromethyl-alpha,beta-unsaturated ketone to the reaction mixture under air atmosphere.
- Stir the mixture at 30°C until the starting material is consumed, then purify via column chromatography to isolate the high-purity product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain strategists, the adoption of this organocatalytic method presents a compelling value proposition centered on cost optimization and supply security. The elimination of transition metals removes a major cost driver associated with catalyst procurement and, more importantly, the downstream removal of metal traces. This simplification of the purification train leads to substantial cost savings in terms of reduced solvent consumption, lower energy usage for heating and cooling, and decreased labor hours for quality control testing. Furthermore, the use of commercially available starting materials and a robust catalyst that functions under air atmosphere enhances supply chain reliability by reducing dependency on scarce or geopolitically sensitive metal resources. The mild reaction conditions also extend the lifespan of reactor equipment and reduce safety risks associated with exothermic runaway reactions.
- Cost Reduction in Manufacturing: The primary economic benefit stems from the metal-free nature of the catalyst, which eradicates the need for expensive metal scavengers and extensive analytical testing for heavy metal residues. This streamlining of the post-reaction workflow significantly lowers the operational expenditure per kilogram of product. Additionally, the high catalytic efficiency allows for low catalyst loading (10 mol%), minimizing the cost contribution of the chiral inducer itself. The ability to run the reaction at 30°C rather than requiring cryogenic conditions or high heat further reduces utility costs, contributing to a leaner and more profitable manufacturing process.
- Enhanced Supply Chain Reliability: By utilizing stable organocatalysts derived from abundant amino acids like L-tert-leucine, the process mitigates risks associated with the supply volatility of precious metals such as palladium or rhodium. The reaction's tolerance to air and moisture means that strict anhydrous or anaerobic conditions are not required, simplifying logistics and storage requirements for raw materials. This robustness ensures consistent production schedules and reduces the likelihood of batch failures due to environmental fluctuations, thereby securing a steady flow of high-purity pharmaceutical intermediates to downstream customers.
- Scalability and Environmental Compliance: The process is inherently scalable due to its mild thermal profile and lack of hazardous oxidants, making it suitable for large-scale production from 100 kgs to multi-ton quantities. The reduced generation of heavy metal waste aligns with increasingly stringent environmental regulations, facilitating easier permitting and waste disposal. This eco-friendly profile not only enhances the corporate sustainability image but also future-proofs the manufacturing site against tightening regulatory frameworks regarding chemical emissions and effluent treatment.
Frequently Asked Questions (FAQ)
To address common technical and commercial inquiries regarding this synthesis method, we have compiled a set of answers based on the experimental data and mechanistic understanding provided in the patent literature. These insights are intended to clarify the operational parameters and potential applications for partners considering this technology for their supply chains. Understanding these details is crucial for assessing the feasibility of integrating this route into existing production lines for specialty chemicals.
Q: What are the key advantages of this organocatalytic method over traditional metal-catalyzed routes?
A: This method eliminates the need for transition metals and stoichiometric oxidants, significantly simplifying downstream purification and reducing heavy metal contamination risks in pharmaceutical intermediates.
Q: What level of enantioselectivity can be achieved with catalyst C5?
A: Experimental data indicates that using catalyst C5 in toluene at 30°C can achieve enantiomeric excess (ee) values up to 98%, ensuring high optical purity for chiral drug synthesis.
Q: Is this process scalable for industrial production of agrochemical intermediates?
A: Yes, the reaction operates under mild conditions (30°C) and air atmosphere without sensitive reagents, making it highly suitable for commercial scale-up and robust supply chain integration.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Chiral Hydrazone Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of this organocatalytic technology in delivering high-value chiral intermediates to the global market. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory discovery to industrial reality is seamless and efficient. Our state-of-the-art facilities are equipped with rigorous QC labs capable of verifying stringent purity specifications, including chiral HPLC analysis to guarantee enantiomeric excess levels that meet or exceed the 98% ee benchmark demonstrated in the patent. We are committed to leveraging our technical expertise to optimize this route for your specific needs, ensuring consistent quality and supply continuity.
We invite you to collaborate with us to explore how this advanced synthesis method can enhance your product portfolio and reduce your overall manufacturing costs. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your volume requirements and quality standards. Please contact us today to request specific COA data for our chiral hydrazone intermediates and to discuss route feasibility assessments for your upcoming projects. Let us be your trusted partner in navigating the complexities of modern chemical synthesis and securing a competitive edge in the marketplace.
