Optimizing FTY720 Intermediate Production: A Technical Analysis of the Novel 5-Step Route
The pharmaceutical landscape for immunosuppressants has been significantly reshaped by the introduction of efficient synthetic pathways for critical intermediates. Patent CN1814583A details a groundbreaking methodology for the preparation of 2-p-octyl-phenethyl-2-amino-propanediol hydrochloride, a pivotal precursor in the manufacturing of FTY720 (Fingolimod). This compound has demonstrated exceptional efficacy in prolonging graft survival in renal, hepatic, and intestinal transplantation models, often exhibiting synergistic effects when combined with agents like CsA or FK506. The technical breakthrough presented in this patent addresses the longstanding inefficiencies of prior art, offering a robust solution that aligns with modern green chemistry principles while maintaining rigorous purity standards required for clinical applications. By transitioning from cumbersome multi-step sequences to a streamlined five-step protocol, this innovation not only enhances chemical efficiency but also provides a scalable framework for industrial production.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Prior to the advancements detailed in CN1814583A, the synthesis of this vital immunosuppressant intermediate was plagued by severe operational and economic inefficiencies. The earliest disclosed methods, such as those found in PCT/JP93/0515, relied on phenethyl alcohol as the starting material, necessitating a tedious ten-step reaction sequence that resulted in a dismal total recovery rate of approximately 4%. Alternative approaches attempted to utilize chloroethyl benzene; however, this required preliminary synthesis from phenethyl alcohol using hazardous reagents like thionyl chloride or phosphorus trichloride. These traditional routes were not only chemically inefficient but also environmentally detrimental, generating large quantities of toxic exhaust gases and requiring high-pressure reaction vessels that imposed stringent safety requirements and inflated capital expenditure for manufacturing facilities.
The Novel Approach
The innovative strategy outlined in the patent fundamentally reengineers the synthetic logic by utilizing styrene and capryloyl chloride as the primary building blocks. This novel approach condenses the entire synthesis into just five distinct chemical transformations, drastically reducing the production cycle and minimizing waste generation. The process initiates with a Friedel-Crafts acylation to establish the carbon skeleton, followed by a Michael addition to introduce the nitrogen functionality, and concludes with selective reductions and salt formation. As illustrated in the reaction scheme below, this pathway eliminates the need for high-pressure equipment and toxic halogenating agents, thereby creating a safer and more economically viable manufacturing environment. The cumulative effect of these improvements is a dramatic increase in total yield, reaching between 32% and 40%, which represents an order-of-magnitude improvement over legacy technologies.
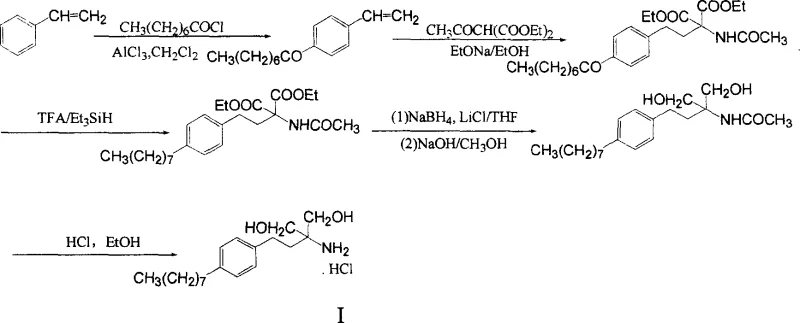
Mechanistic Insights into the Five-Step Synthetic Cascade
The core of this technological advancement lies in the precise control of regioselectivity and functional group tolerance throughout the reaction cascade. The initial Friedel-Crafts acylation between styrene and capryloyl chloride, catalyzed by aluminum chloride in dichloromethane, is meticulously controlled at low temperatures (0-5°C) to ensure para-substitution, yielding p-octanoyl styrene with a remarkable 90% isolated yield. This step is critical as it establishes the lipophilic octyl chain essential for the biological activity of the final molecule. Subsequently, the Michael addition reaction employs sodium ethoxide to facilitate the conjugate addition of acetamido diethyl malonate to the alpha,beta-unsaturated ketone system. This transformation is conducted under reflux in ethanol for an extended period to drive the equilibrium towards the desired adduct, achieving a 60% yield while maintaining the integrity of the sensitive ester groups.
Following the construction of the carbon-nitrogen backbone, the process employs a sophisticated reduction strategy to convert the ketone and ester functionalities into the final amino-diol structure. The ketone moiety is selectively reduced using triethyl silane in the presence of trifluoroacetic acid, a method chosen for its mildness and compatibility with the acetamido group, resulting in an 85% yield. The final transformation involves a dual reduction and hydrolysis step using sodium borohydride and lithium chloride in tetrahydrofuran, followed by alkaline hydrolysis. This specific reagent combination is crucial for reducing the esters to primary alcohols without affecting the amine protection prematurely, ultimately delivering the target amino-propanediol with an 87% yield before the final salt formation step secures the product stability.
How to Synthesize 2-p-octyl-phenethyl-2-amino-propanediol hydrochloride Efficiently
Implementing this synthesis requires strict adherence to the reaction parameters defined in the patent to maximize yield and purity. The process is designed to be operationally simple, avoiding the complexities of cryogenic conditions or inert atmosphere requirements beyond standard laboratory practices. Each intermediate can be purified through straightforward crystallization or extraction techniques, ensuring that impurities do not carry over to subsequent steps. For R&D teams looking to replicate or scale this process, the following standardized guide outlines the critical operational phases derived directly from the patent embodiments.
- Perform Friedel-Crafts acylation between styrene and capryloyl chloride using AlCl3 to generate p-octanoyl styrene.
- Execute Michael addition with acetamido diethyl malonate under basic conditions (EtONa) to form the diethyl malonate derivative.
- Reduce the ketone group using triethyl silane and trifluoroacetic acid (TFA) to obtain the octyl styryl intermediate.
- Conduct reduction and hydrolysis using NaBH4 and LiCl in THF, followed by base treatment to yield the amino-propanediol.
- Finalize the process by acidifying with hydrochloric acid in ethanol to crystallize the pure hydrochloride salt.
Commercial Advantages for Procurement and Supply Chain Teams
From a strategic procurement perspective, the adoption of this synthetic route offers profound advantages in terms of cost structure and supply chain resilience. The shift from expensive, multi-step precursors to commodity chemicals like styrene significantly lowers the raw material entry cost, which is a primary driver of the overall cost of goods sold (COGS). Furthermore, the reduction in reaction steps from ten to five inherently reduces the consumption of solvents, reagents, and energy, leading to a leaner manufacturing footprint. This efficiency translates directly into improved margin potential for downstream API manufacturers, making the sourcing of intermediates produced via this method highly attractive for long-term supply agreements focused on cost optimization.
- Cost Reduction in Manufacturing: The elimination of high-pressure reactors and the avoidance of toxic reagents like thionyl chloride remove significant capital and operational expenditures associated with specialized equipment maintenance and hazardous waste disposal. By simplifying the process to ambient or mild thermal conditions, the energy intensity of the production is drastically lowered, contributing to substantial cost savings in utility consumption. Additionally, the high yield of each individual step minimizes the loss of valuable intermediates, ensuring that the theoretical mass balance is closely approached in practical production scenarios.
- Enhanced Supply Chain Reliability: Utilizing widely available starting materials such as styrene and capryloyl chloride mitigates the risk of supply disruptions often associated with niche or custom-synthesized precursors. The robustness of the chemical transformations means that the process is less susceptible to minor fluctuations in reaction conditions, ensuring consistent batch-to-batch quality and reliable delivery schedules. This stability is crucial for pharmaceutical supply chains where continuity of supply is paramount to prevent production stoppages of the final active pharmaceutical ingredient.
- Scalability and Environmental Compliance: The process generates significantly fewer "three wastes" (waste water, waste gas, and waste residue) compared to conventional methods, simplifying the regulatory compliance burden for manufacturing sites. The absence of high-pressure steps and toxic gas evolution makes the technology easier to scale from pilot plant to commercial tonnage without requiring extensive retrofitting of existing facilities. This environmental friendliness not only reduces disposal costs but also aligns with the increasingly stringent sustainability mandates imposed by global regulatory bodies and corporate responsibility initiatives.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented synthesis method. These insights are derived from the specific experimental data and comparative analysis provided in the patent documentation, offering clarity on yield expectations, safety profiles, and raw material sourcing. Understanding these details is essential for technical teams evaluating the feasibility of integrating this route into their existing manufacturing portfolios.
Q: What is the total yield advantage of this new synthesis method compared to conventional routes?
A: The novel 5-step process described in patent CN1814583A achieves a total yield of 32% to 40%, which is a substantial improvement over the conventional 10-step route that typically yields only around 4%.
Q: Does this manufacturing process involve high-pressure reactions or toxic gases?
A: No, unlike previous methods requiring thionyl chloride or phosphorus trichloride which generate large quantities of exhaust gas, this route utilizes mild conditions and avoids high-pressure equipment, significantly enhancing operational safety.
Q: What are the primary raw materials used in this optimized pathway?
A: The synthesis begins with commercially available and cost-effective raw materials, specifically styrene and capryloyl chloride, replacing the more expensive and difficult-to-handle phenethyl alcohol derivatives used in older methods.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable FTY720 Intermediate Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of efficient synthetic routes in the competitive landscape of immunosuppressant manufacturing. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory bench to industrial reactor is seamless and robust. We are committed to delivering high-purity pharmaceutical intermediates that meet stringent purity specifications, supported by our rigorous QC labs equipped with state-of-the-art analytical instrumentation to verify every batch against the highest industry standards.
We invite procurement leaders and R&D directors to engage with us for a Customized Cost-Saving Analysis tailored to your specific volume requirements. By leveraging our expertise in this optimized 5-step synthesis, we can help you achieve significant reductions in manufacturing costs while securing a stable supply of high-quality intermediates. Please contact our technical procurement team today to request specific COA data and comprehensive route feasibility assessments that demonstrate how our capabilities can support your long-term production goals.
