Advanced Manufacturing of 3-Sulfonyl Coumarins via Radical Cascade Cyclization for Global Supply Chains
Advanced Manufacturing of 3-Sulfonyl Coumarins via Radical Cascade Cyclization for Global Supply Chains
The pharmaceutical and fine chemical industries are constantly seeking robust synthetic pathways that balance molecular complexity with manufacturing efficiency, particularly for heterocyclic scaffolds that serve as critical building blocks in drug discovery. Patent CN106045955B introduces a transformative methodology for the preparation of 3-sulfonyl coumarin compounds, addressing long-standing challenges associated with traditional sulfonylation techniques. This innovation leverages a radical cascade cyclization strategy utilizing DABCO.(SO2)2 as a stable sulfur dioxide surrogate, enabling the direct construction of the coumarin skeleton under remarkably mild conditions. For R&D Directors and Procurement Managers, this represents a significant shift away from hazardous reagents towards a more sustainable and operationally simple process. The ability to synthesize these high-value intermediates without relying on corrosive sulfonyl chlorides or strong oxidants not only enhances laboratory safety but also streamlines the supply chain by reducing the dependency on specialized, difficult-to-handle reagents. As we analyze the technical depth of this patent, it becomes clear that this methodology offers a viable route for the commercial scale-up of complex pharmaceutical intermediates, ensuring consistent quality and supply continuity for global markets.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of sulfone-containing compounds has relied heavily on two primary strategies, both of which present substantial drawbacks for large-scale industrial application. The first conventional method involves the oxidation of sulfur-containing precursors such as thiophenols, thiols, or thioethers, which frequently necessitates the use of strong oxidizing agents that can be dangerous to handle and generate significant amounts of chemical waste. Furthermore, sulfur-containing reagents often possess foul odors and require specialized containment systems to prevent environmental contamination and operator exposure. The second traditional approach utilizes the substitution reaction between sulfinates and electrophiles, where the sulfinates are typically derived from sulfonyl chlorides. The preparation of sulfonyl chlorides itself is a hazardous process involving strong chlorinating reagents, creating a supply chain bottleneck due to the instability and corrosivity of these intermediates. These legacy methods impose high costs on waste treatment, require expensive corrosion-resistant equipment, and introduce significant safety risks that can disrupt production schedules. For supply chain heads, the reliance on such volatile chemistry translates to higher insurance costs, stricter regulatory compliance burdens, and potential delays in raw material sourcing, making the conventional routes increasingly untenable for modern, efficiency-driven manufacturing environments.
The Novel Approach
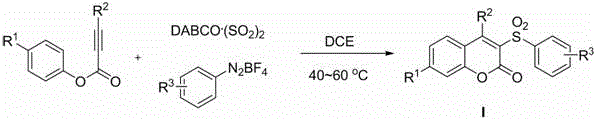
In stark contrast to these legacy techniques, the novel approach detailed in the patent utilizes aryl diazonium salts and DABCO.(SO2)2 to generate aryl sulfonyl radicals in situ, which then undergo a cascade reaction with phenyl phenylpropiolate. This method operates in common organic solvents such as 1,2-dichloroethane (DCE), 1,4-dioxane, or toluene at a mild temperature range of 40-60°C, eliminating the need for extreme thermal conditions or high-pressure equipment. By bypassing the use of sulfonyl chloride intermediates entirely, this process removes a major safety hazard and simplifies the raw material profile to stable, commercially available solids and liquids. The reaction proceeds efficiently within 0.5 to 1.0 hours, as monitored by TLC, indicating a rapid kinetic profile that enhances throughput potential. For procurement teams, this shift means sourcing simpler, less regulated chemicals that are less prone to supply disruptions compared to specialized chlorinating agents. The operational simplicity extends to the workup procedure, where the reaction mixture can be directly concentrated and purified via column chromatography, reducing the number of unit operations required. This streamlined workflow not only lowers labor costs but also minimizes the footprint of the manufacturing process, aligning perfectly with the industry's drive towards greener and more cost-effective chemical production methodologies.
Mechanistic Insights into DABCO.(SO2)2-Mediated Radical Cascade Cyclization
The core of this technological breakthrough lies in the unique reactivity of DABCO.(SO2)2, which serves as a solid, stable source of sulfur dioxide that releases the gas under mild thermal conditions to participate in radical chemistry. Upon heating in the presence of aryl diazonium salts, the system generates aryl sulfonyl radicals through a single-electron transfer process, initiating a cascade that is both highly selective and efficient. These radicals subsequently add to the triple bond of the phenyl phenylpropiolate, forming a vinyl radical intermediate that is poised for intramolecular cyclization. This cyclization step constructs the coumarin core with high fidelity, followed by an ester group migration and aromatization sequence that finalizes the formation of the 3-sulfonyl coumarin structure. The elegance of this mechanism is that it combines multiple bond-forming events into a single operational step, drastically reducing the step count compared to linear synthetic routes. For R&D directors, understanding this mechanism is crucial as it highlights the tolerance of the reaction to various electronic substituents, allowing for the synthesis of a diverse library of derivatives without modifying the core conditions. The radical nature of the transformation ensures that even sterically hindered substrates can be accommodated, providing a versatile platform for medicinal chemistry optimization.
Furthermore, the impurity profile of this reaction is significantly cleaner than traditional methods due to the absence of side reactions commonly associated with strong oxidants or chlorinating agents. The mild conditions prevent the decomposition of sensitive functional groups that might be present on the aryl diazonium salt or the alkyne substrate, thereby preserving the integrity of complex molecular architectures. The use of inert gas protection, such as nitrogen or argon, ensures that the radical intermediates are not quenched by oxygen, maintaining high conversion rates and consistent yields ranging from 50% to 88% as reported in the patent data. This level of control over the reaction environment is essential for maintaining batch-to-batch consistency, a key metric for quality assurance in pharmaceutical manufacturing. By avoiding the formation of sulfonic acid byproducts or chlorinated waste streams, the downstream purification process is simplified, leading to higher overall recovery of the target API intermediate. The mechanistic robustness of this pathway provides a solid foundation for process validation, giving supply chain stakeholders confidence in the reliability of the production method for long-term commercial contracts.
How to Synthesize 3-Sulfonyl Coumarin Efficiently
The implementation of this synthesis route in a production setting requires careful attention to the stoichiometry and addition sequence of the reagents to maximize efficiency and safety. The patent outlines a straightforward protocol where DABCO.(SO2)2, phenyl phenylpropiolate, and the aryl diazonium salt are combined in an organic solvent, typically using a molar ratio that favors the sulfur dioxide surrogate to ensure complete conversion of the alkyne. Detailed standardized synthesis steps are provided in the guide below to ensure reproducibility across different scales of operation.
- Charge the reactor with DABCO.(SO2)2, phenyl phenylpropiolate, and aryl diazonium salt in an organic solvent such as 1,2-dichloroethane.
- Maintain the reaction mixture under an inert atmosphere of nitrogen or argon while heating to a temperature range of 40-60°C.
- Stir the mixture for 0.5 to 1.0 hours until TLC indicates completion, then concentrate and purify via column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this patented methodology offers profound advantages that extend beyond mere chemical efficiency, directly impacting the bottom line and operational resilience of the manufacturing organization. The elimination of sulfonyl chloride intermediates removes a significant cost center associated with the handling, storage, and disposal of hazardous corrosive materials, leading to substantial cost savings in waste management and safety compliance. Additionally, the mild reaction conditions reduce energy consumption compared to high-temperature processes, contributing to a lower carbon footprint and reduced utility costs. For procurement managers, the ability to source stable, non-hazardous raw materials like DABCO.(SO2)2 and simple aryl diazonium salts mitigates the risk of supply chain disruptions caused by regulatory restrictions on dangerous goods. This stability ensures a continuous flow of materials into the production line, preventing costly downtime and enabling more accurate forecasting of production output. The simplified workup procedure further reduces labor hours and solvent usage, enhancing the overall economic viability of the process for large-scale manufacturing.
- Cost Reduction in Manufacturing: The primary driver for cost reduction in this process is the strategic avoidance of expensive and hazardous sulfonyl chloride intermediates, which traditionally require specialized storage facilities and generate significant acidic waste streams that are costly to neutralize. By utilizing DABCO.(SO2)2 as a stable solid surrogate, the process eliminates the need for corrosion-resistant reactors and extensive scrubbing systems, thereby lowering capital expenditure and maintenance costs. Furthermore, the high atom economy of the cascade reaction minimizes the amount of raw material wasted in side products, ensuring that a greater proportion of the input mass is converted into valuable product. This efficiency translates directly into a lower cost of goods sold (COGS), allowing for more competitive pricing in the global market while maintaining healthy profit margins. The reduction in solvent volume and purification steps also contributes to significant operational savings, making this route economically superior to conventional multi-step syntheses.
- Enhanced Supply Chain Reliability: Supply chain reliability is significantly bolstered by the use of commercially available and stable starting materials that are not subject to the same stringent transportation regulations as corrosive sulfonyl chlorides or strong oxidants. This ease of logistics allows for broader sourcing options and larger inventory buffers without incurring excessive safety risks or storage costs. The robustness of the reaction conditions, operating at mild temperatures and atmospheric pressure, reduces the likelihood of equipment failure or process deviations that could lead to batch losses. For supply chain heads, this means a more predictable production schedule and the ability to meet tight delivery deadlines with greater confidence. The simplified process flow also reduces the dependency on specialized contract manufacturing organizations that possess specific hazard handling capabilities, giving the company more flexibility in choosing production partners and locations.
- Scalability and Environmental Compliance: The scalability of this method is evidenced by its simple operational parameters and the absence of exothermic hazards associated with strong oxidants, making it safe to transition from laboratory benchtop to multi-ton production scales. The environmental compliance aspect is particularly strong, as the process avoids the generation of chlorinated waste and sulfur-containing odors, aligning with increasingly strict global environmental regulations. This green chemistry profile facilitates easier permitting for new production facilities and reduces the risk of regulatory fines or shutdowns due to emissions violations. The ability to scale without compromising safety or quality ensures that the supply of high-purity pharmaceutical intermediates can grow in tandem with market demand. This long-term viability makes the technology a strategic asset for companies looking to secure their position in the fine chemical market through sustainable and compliant manufacturing practices.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology, providing clarity for stakeholders evaluating its adoption. These answers are derived directly from the patent specifications and practical considerations for industrial application, ensuring that all information is accurate and relevant to decision-making processes.
Q: What are the primary advantages of using DABCO.(SO2)2 over traditional sulfonyl chlorides?
A: The use of DABCO.(SO2)2 eliminates the need for hazardous sulfonyl chloride intermediates, which are often corrosive, unstable, and generate significant acidic waste. This solid sulfur dioxide surrogate is stable, easy to handle, and releases SO2 in situ under mild conditions, significantly improving operational safety and reducing waste treatment costs in large-scale manufacturing.
Q: What is the typical yield range for this radical cascade cyclization process?
A: According to the patented data, the reaction consistently achieves yields ranging from 50% to 88% across various substrate derivatives. This robust yield profile is maintained under relatively mild thermal conditions (40-60°C), indicating a high level of process reliability suitable for commercial production.
Q: Can this synthesis method be scaled for industrial production of pharmaceutical intermediates?
A: Yes, the process is explicitly designed for scalability. The avoidance of strong oxidants and the use of simple, commercially available starting materials like phenyl phenylpropiolate facilitate safe scale-up. The mild temperature requirements also reduce energy consumption and equipment stress, making it highly viable for multi-kilogram to ton-scale manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3-Sulfonyl Coumarin Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of adopting advanced synthetic technologies to maintain a competitive edge in the global fine chemical market. Our team of expert chemists has thoroughly evaluated the pathway described in CN106045955B and confirmed its potential for robust commercial production. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from lab-scale innovation to industrial reality is seamless and efficient. Our facilities are equipped with stringent purity specifications and rigorous QC labs to guarantee that every batch of 3-sulfonyl coumarin meets the highest standards required by the pharmaceutical industry. We understand that consistency is key for our clients, and our commitment to quality assurance ensures that the impurity profiles remain within tight limits, supporting your regulatory filings and product safety.
We invite you to collaborate with us to optimize your supply chain for these valuable intermediates. By leveraging our technical expertise, we can provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality targets. We encourage you to contact our technical procurement team to request specific COA data and route feasibility assessments for your projects. Our goal is to be more than just a supplier; we aim to be a strategic partner in your development process, offering the reliability and technical support needed to bring your products to market faster and more efficiently.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
