Advanced Synthesis of Oxiracetam via Silyl Protection for Commercial Scale-up
Advanced Synthesis of Oxiracetam via Silyl Protection for Commercial Scale-up
The pharmaceutical industry continuously seeks robust manufacturing pathways for nootropic agents like Oxiracetam, where purity and process efficiency are paramount. Patent CN102746207B, published in late 2013, introduces a transformative synthetic methodology that addresses critical bottlenecks in existing production technologies. This innovation leverages advanced silyl protection strategies, specifically utilizing tert-butyldimethylsilyl (TBDMS) or tert-butyl diphenylsilyl groups, to stabilize the hydroxyl functionality of the pyrrolidone core. By shifting away from unstable trimethylsilyl protections and direct alkylation methods that suffer from severe side reactions, this patent outlines a route capable of delivering product purity exceeding 99% without the need for complex ion-exchange resin purification. For global procurement teams and R&D directors, this represents a significant opportunity to optimize the supply chain for high-purity pharmaceutical intermediates, ensuring consistent quality while drastically simplifying downstream processing requirements.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of Oxiracetam has been plagued by inefficient purification steps and low overall yields, primarily due to the physicochemical properties of the intermediates involved. As illustrated in earlier patents such as US4173569A, the use of trimethylsilyl (TMS) protecting groups results in intermediates that are liquids and highly water-soluble, making isolation extremely difficult. These liquid intermediates necessitate the removal of large volumes of water under reduced pressure, placing high demands on equipment and increasing energy consumption. Furthermore, the acidic deprotection conditions required for TMS groups often induce hydrolysis of the lactam ring or the amide side chain, generating acidic impurities that are notoriously difficult to separate. Consequently, manufacturers are forced to rely on repeated passage through ion-exchange resin columns and extensive concentration of aqueous solutions, a process that not only lowers yield but also risks thermal degradation of the sensitive Oxiracetam molecule.
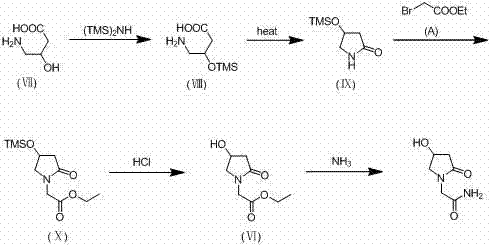
The Novel Approach
In stark contrast to these legacy methods, the methodology disclosed in CN102746207B employs bulkier silyl protecting groups, specifically TBDMS or TBDPS, which fundamentally alter the physical state of the reaction intermediates. This strategic modification ensures that the key amide intermediate precipitates as a solid directly from the reaction mixture upon aminolysis, allowing for the removal of the majority of impurities through simple filtration rather than chromatography or resin treatment. The subsequent deprotection step utilizes an alcoholic solvent under acidic conditions, which cleverly esterifies any trace carboxylic acid byproducts formed during the reaction, keeping them in solution while the target Oxiracetam crystallizes out. This elegant solution eliminates the need for water concentration and resin purification, resulting in a process that is not only operationally simpler but also delivers significantly higher purity and yield suitable for GMP manufacturing environments.
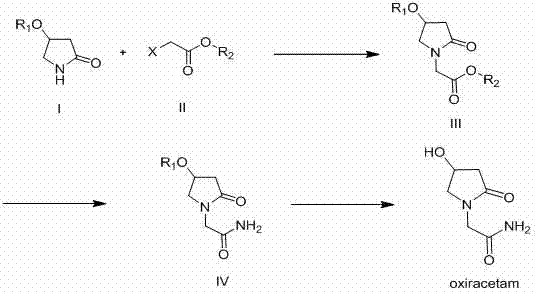
Mechanistic Insights into Silyl-Protection Mediated Alkylation
The core of this technological advancement lies in the enhanced stability and steric bulk provided by the tert-butyldimethylsilyl ether moiety. In the initial alkylation step, the nitrogen atom of the protected 4-hydroxy-2-pyrrolidinone acts as a nucleophile, attacking the electrophilic carbon of a low-molecular-weight halogenated acetic acid ester, such as ethyl bromoacetate. This reaction is conducted in non-protonated solvents like tetrahydrofuran (THF) or dimethylformamide (DMF) at controlled low temperatures between -5°C and 10°C to suppress potential O-alkylation or elimination side reactions. The use of strong, non-nucleophilic bases such as lithium hexamethyldisilazide (HMDSLi) ensures quantitative deprotonation of the lactam nitrogen, driving the formation of the N-alkylated ester intermediate with high regioselectivity. Unlike the fragile TMS ether, the TBDMS ether remains intact under these basic conditions, preserving the integrity of the hydroxyl group for the final deprotection step.
Following alkylation, the ester intermediate undergoes aminolysis with ammonia to form the corresponding acetamide. It is at this stage that the superiority of the TBDMS group becomes most apparent; the resulting acetamide derivative possesses low solubility in the reaction medium and precipitates as a high-purity solid, typically exceeding 99% purity by HPLC analysis. This phase separation is a critical process control point, as it physically excludes soluble organic impurities and salts from the product stream. The final step involves acid-catalyzed cleavage of the silyl ether using hydrochloric acid in methanol or ethanol. The alcoholic solvent serves a dual purpose: it solubilizes the silyl byproducts and simultaneously esterifies any free carboxylic acids generated by minor hydrolysis, preventing them from contaminating the final zwitterionic Oxiracetam product which precipitates upon completion.
How to Synthesize Oxiracetam Efficiently
The synthesis protocol detailed in the patent provides a clear, scalable pathway for producing Oxiracetam with minimal equipment requirements. The process begins with the protection of commercially available 4-hydroxy-2-pyrrolidinone, followed by N-alkylation and amidation, and concludes with a mild acidic deprotection. This sequence avoids the harsh conditions and complex workups associated with previous generations of synthesis technology. For process chemists looking to implement this route, the key lies in maintaining strict temperature control during the alkylation phase and leveraging the crystallization behavior of the intermediates for purification. Detailed standardized synthesis steps are provided in the guide below to ensure reproducibility and safety.
- React 4-tert-butyldimethylsilyloxy-2-oxo-pyrrolidine with low-molecular-weight halogenated acetic acid ester in a non-protonated solvent under basic conditions at -5 to 10°C.
- Perform aminolysis on the resulting ester intermediate using ammonia to form the solid amide derivative.
- Remove the silyl protecting group under acidic conditions in an alcoholic solvent to isolate high-purity Oxiracetam.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this synthesis route offers profound benefits for cost reduction in pharmaceutical intermediates manufacturing and supply chain reliability. By eliminating the need for ion-exchange resin columns and the associated regeneration chemicals, the process significantly reduces both material costs and waste disposal expenses. The ability to purify intermediates via crystallization and filtration rather than chromatography or extensive aqueous workups translates to shorter cycle times and reduced solvent consumption. Furthermore, the use of stable, solid intermediates mitigates the risks associated with handling hygroscopic liquids, thereby enhancing operational safety and reducing the potential for batch-to-batch variability caused by moisture sensitivity.
- Cost Reduction in Manufacturing: The elimination of expensive purification resins and the reduction in solvent usage for extraction and concentration directly lower the variable cost of goods sold. Since the intermediates precipitate as solids, the energy-intensive process of evaporating large volumes of water is removed from the workflow, leading to substantial utility savings. Additionally, the high yield of the final deprotection step minimizes raw material waste, ensuring that the expensive starting materials are converted efficiently into the final API intermediate.
- Enhanced Supply Chain Reliability: The reagents required for this synthesis, such as TBDMSCl and ethyl bromoacetate, are commodity chemicals with robust global supply chains, reducing the risk of raw material shortages. The simplicity of the equipment requirements—standard reactors and filtration units—means that production can be easily scaled or transferred between manufacturing sites without the need for specialized resin columns or complex distillation setups. This flexibility ensures consistent supply continuity even during periods of high market demand.
- Scalability and Environmental Compliance: The process generates significantly less aqueous waste compared to traditional methods that rely on resin desalting and water concentration. By avoiding the generation of large volumes of saline wastewater, the environmental footprint of the manufacturing process is drastically reduced, simplifying compliance with increasingly stringent environmental regulations. The solid-state handling of intermediates also reduces the risk of spills and exposure, contributing to a safer and more sustainable manufacturing environment.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation and advantages of this specific Oxiracetam synthesis pathway. These insights are derived directly from the experimental data and comparative analysis presented in the patent documentation, providing clarity on why this method is preferred for modern industrial applications. Understanding these nuances is essential for technical teams evaluating process transfers or vendor qualifications.
Q: Why is the TBDMS protecting group superior to TMS in Oxiracetam synthesis?
A: The tert-butyldimethylsilyl (TBDMS) group offers greater stability compared to the trimethylsilyl (TMS) group used in older methods. This stability prevents premature hydrolysis during the aminolysis step, allowing the intermediate to precipitate as a solid with purity greater than 99%, whereas TMS intermediates often remain liquid and water-soluble, complicating purification.
Q: How does this method improve product purity compared to prior art?
A: Traditional methods often require ion-exchange resin purification and extensive water concentration, which can lead to hydrolysis byproducts. This novel method allows the key intermediate to crystallize directly from the reaction mixture, removing impurities via simple filtration. The final deprotection in alcohol further esterifies trace acid byproducts, preventing them from co-precipitating with the target Oxiracetam.
Q: What are the typical reaction conditions for the alkylation step?
A: The alkylation is conducted in non-protonated solvents such as THF or DMF at low temperatures ranging from -5°C to 10°C. Strong bases like lithium hexamethyldisilazide (HMDSLi) or sodium hydride are used to facilitate the nucleophilic substitution of the halogenated acetate onto the pyrrolidone nitrogen.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Oxiracetam Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of adopting advanced synthetic methodologies to meet the rigorous demands of the global pharmaceutical market. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of patents like CN102746207B are fully realized in practical, large-scale operations. We maintain stringent purity specifications and operate rigorous QC labs to guarantee that every batch of Oxiracetam meets the highest international standards, providing our partners with the confidence they need to advance their own drug development pipelines.
We invite you to engage with our technical procurement team to discuss how this optimized synthesis route can benefit your specific project requirements. By requesting a Customized Cost-Saving Analysis, you can gain a deeper understanding of the economic advantages of switching to this superior manufacturing process. We encourage potential partners to contact us directly to obtain specific COA data and route feasibility assessments, allowing us to demonstrate our commitment to quality and innovation as your trusted long-term supplier.
